Core Concepts-L4-Inflammation and Cellular Recruitment
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
How is the early innate immune response initiated and how are neutrophils and monocytes recruited to infection sites?
resident cells are the first sensors- macrophages, DC, mast cells
detect infection by pattern recognition receptors- PRR’s and release inflammatory mediator: IL-1, TNF, C3a and C5a and induce vasodilation
2. blood vessel respond to this- up regulate adhesion molecules like E-selectin and P-selectin. lose tight junction integrity and allow leukocytes in to deliver immune cells and humeral factors(complement, Ab)
3. chemokine guide trafficking. neutrophils have CXCR1 and follow CXCL8 first. monocytes have CCR2 receptors and follow MCP-1/CCL2
where do immune cells come from?
bone marrow stem cells- self renewing pluripotent
stroll cells support hemopoiettic stem cells
hematopoietic stem cells give rise to all blood and immune cells and differentiate from lymphoid progenitor and myeloid progenitor(and erythrocytes)
how does hematopoiesis work?
regulated by growth factors and cytokines- CSF-1 or GM-CSF
lineage specific transcription factors- such as C-kit and Lineage markers
also depends on the niches and specific environment
what do phagocytes do?
destroy pathogens or debris to limit infection
create peptide antigens for T/B cells recognition
signal and modulate immune responses
explain the process of phagocytosis
recognition- detect danger signals- PAMPs and MAMPs
cell membrane rearranges its actin cytoskeleton and forms a phagosome.
phagosome then fuses with the lysosome to form a phagolysosome. has digestive enzymes and reactive oxygen species for killing microbes.
NADPH oxidase on phagosome membrane produces superoxide and kills microbes
what are phagocytic cells are there?
macrophages- professional phagocytes- engulf microbes and debris and antigen presentation
neutrophils- highly antimicrobial- engulf and kill microbes and degranulate and form nets
dendritic cells- phagocytose microbes and present antigens to T cells- perform macropinocytosis- always taking in its environment and sampling it
role of antigen presenting cells and how do they do this?
capture and process antigens on MHC molecules to antigen specific lymphocytes
MHC I- endogenous/intracellular pathogens- to D8 cells
MHC II- CD4 cells- exogenous-extracellular pathogens
steps of T/B cell activation
recognition- T/B cell receptor binds to the MHC
produce costimulatory cells from the APC- CD80
cytokine signalling- APC makes cytokines and helps with T cell differentiation
describe the difference between the induction vs effector phase and when does this happen
induction phase- in the lymph nodes- naive T cells encounter antigens from APCs and get activated and differentiate
effector phase- at the site of infection- activated T cells migrate to the infected tissue and recognise their specific antigen- and target elimination
induction- trains T cells
effector- action at the infection site
discuss resident phagocytic cells
macrophages
more frequent than DCs and are irregular shaped.
rich in lysosomes
from myeloid progenitor- common at mucosal surfaces
has PRR’s and can present antigens
name the type of innate immune cells
phagocytic cells- engulfs and destroy pathogens/debris. can also signal the immune system. macrophages/neutrophils/DC
signalling cells- detect pathogens/danger signals and release mediators. can also recruit other immune cells. certain DCs, innate lymphoid cells, mast cells
name phagocytic surface markers
CD11b- inter grin involved in adhesion and phagocytosis
CD14- coreceptor for LPS recognition(TLR4)
F4/80- classic macrophage marker found in the mouse
discuss tissue resident DC
has CD11c integrin, CD80/86- a costimulatory molecule for T cell activation
Has mHC antigen complexes- to present to T cells
has CCR7- a chemokine receptor to bring it to the lymph nodes
resident in tissues but always trafficking through lymphatics
discuss epithelial resident cells
express pattern recognition receptors and TLRs- regulated in the gut to stop overactivation
nn phagocytic- specialised transport for nutrient absorption but traffic molecules like innate cells
uses xenophagy to restrict infection- degrades damaged organelles in a double membrane vesicle. acidifies vesicle and fuses with lysosome to control cytoplasmic infection
discuss intestinal epithelial cells
IeCs- mucosal surface- senses via PRRs and degrades damaged organelles and cytoplasmic pathogens
paneth cells- small cells- produce AMPs
goblet cells- among epithelium- make mucin and forms a protective mucosal barrier
M-cells- transports antigens via transcytosis- deliver to underlying APCs
discuss mast resident cells
degranulates
releases histamine and other mediators
causes vasodilation and fluid release and stimulates local nerves, recruits immune cells to sites of inflammation
discuss specificity in immune cell recruitment- chemokines and their receptors on the cell
chemokine and ligands
CCL2 target monocytes via CCR2- follows chemokine gradient to site of infection
CCL21- lymphocytes- guides migration to lymph nodes for APC
neutrophils- CXCL8(IL-8) is produced and has CXCR1 detect gradient
discuss neutrophil recruitment
rolling: neutrophils slow down in the bloodstream near site of infection
selectin on activated endothelial cells- E-selectin/P-selectin are upregulated on inflamed endothelium and binds to carbohydrate ligands on neutrophils. stops neutrophils from flowing freely and ROLL instead
chemokine released from the tissue bind to chemokine receptors on neutrophils- CXCL8 binds to receptor CXCR1/CXCR2. activates intergrins(LFA-1/Mac-1)on neutrophils for firm adhesion
adhesion- integrins(LFA-1/Mac-1)- bind tightly to immoglobulin superfamily adhesion molecules on endothelial cells- ICAM-1 and ICAM-2. goes from rolling to firm arrest at site of inflammation
discuss macrophage recruitment
Rolling (P/E-selectin ↔ PSGL-1) → Chemokine activation (CCL2→CCR2) → Integrin activation → Firm adhesion(ICAM-1/VCAM-1) → Diapedesis (CCL2-guided).
monocytes- rolling and have loose interaction due to up regulation of selecting on activated endothelium slow down monocytes.
P selection and E selection on endothelium cells and monocyte P selection glycoprotein ligand 1 on the monocytes. binding occurs via carbohydrate monomers
chemokine from tissue bind to monocyte receptors CCL2 to CCR2 receptors- activates integrins for firm adhesion
Ig superfamily on endothelium interact with integrins on monocytes. ICAM-1(on endothelium) and VACM interact with mCR3 on the monocyte
MCP-1 and CCR-2 chemokine help the monocyte get under the endothelium
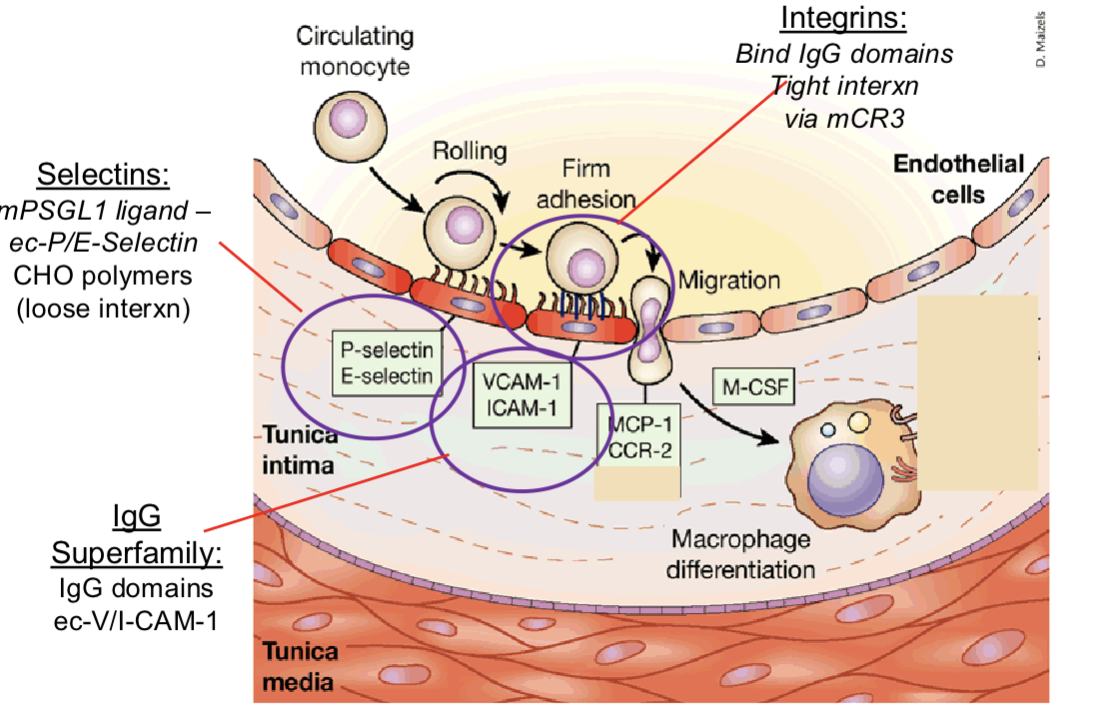

discuss macrophage differentiation
monocyte integrins CR3 are activated during firm adhesion which rearranges the actin cytoskeleton
once in the tissue- monocytes find the chemokine CCL2(CCR2 receptor)
maturation- entry into tissues triggers maturation. key signal CSF-1 promotes the phagocytic capacity and survival. makes large cytoplasm with lysosomes and the ability to secrete cytokines
become M1 macrophages by IFN-gamma, TNF- pro inflammatory or M2 macrophages- IL-4, IL-13 by dead cells and are anti inflammatory and secrete IL-10
difference between immune cell infilitration- neutrophils vs monocytes
timing- neutrophils are recruited first
chemokine in monocytes- CCL1 and responds by CCR2 receptor in monocytes
chemokine in neutrophils- CXCL8 responds by CCL2 in neutrophils.
integrins in neutrophils- LFA-1,Mac-1
integrins in monocytes- CR3/Mac-3
discuss similarities between cell infiltration between neutrophils and monocytes
rolling/firm adhesion/diapedesis
both depend on chemokine gradients(nt- CXCL8-CCL2, mono-CCL1 responds by CCR2)
interaction with firm adhesion- both use selectin P/E and integrins for firm adhesion- ICAM-1, VCAM-1