ccp chem - kinetics chapter test
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
19 Terms
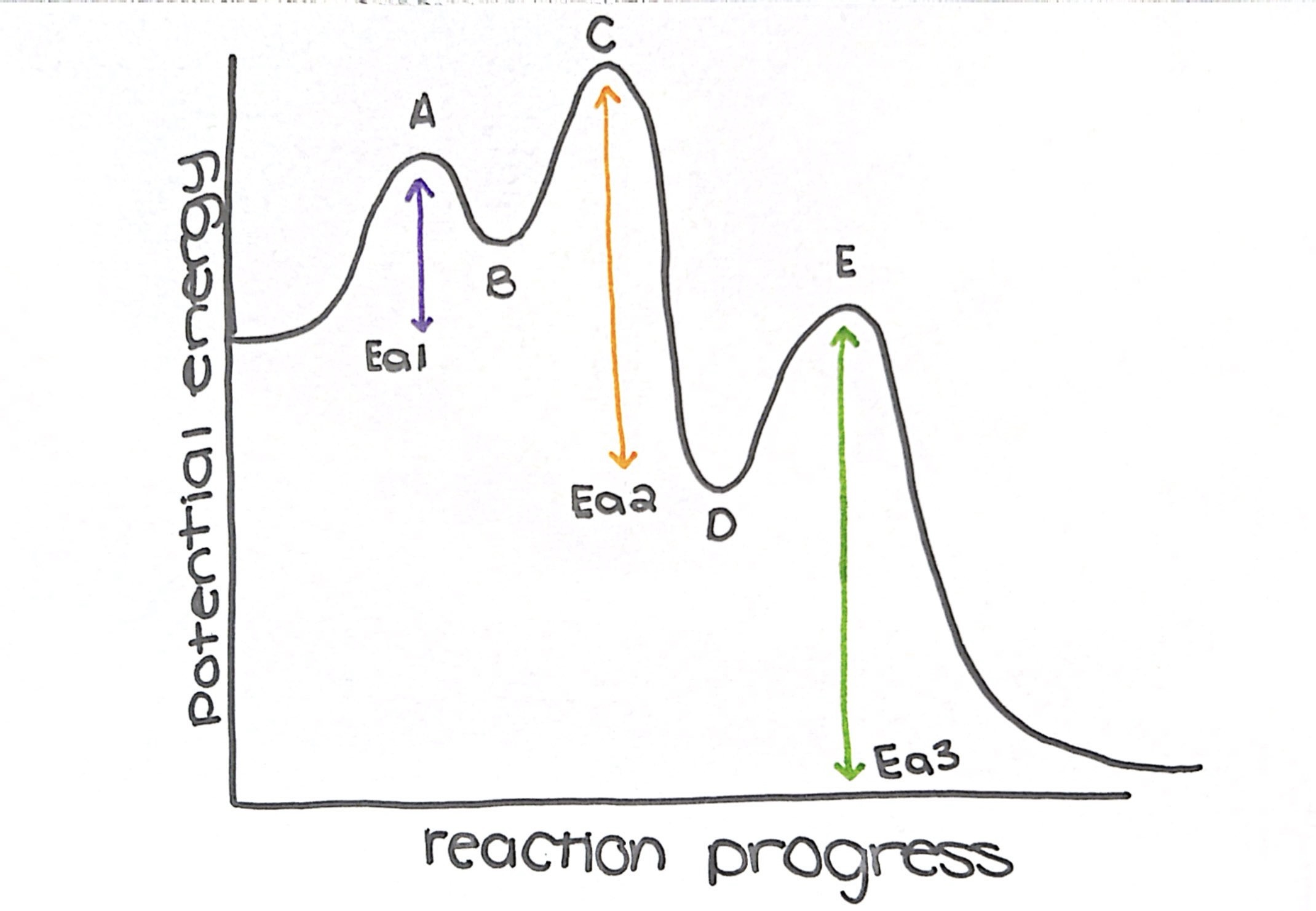
what type of reaction is this diagram
exothermic
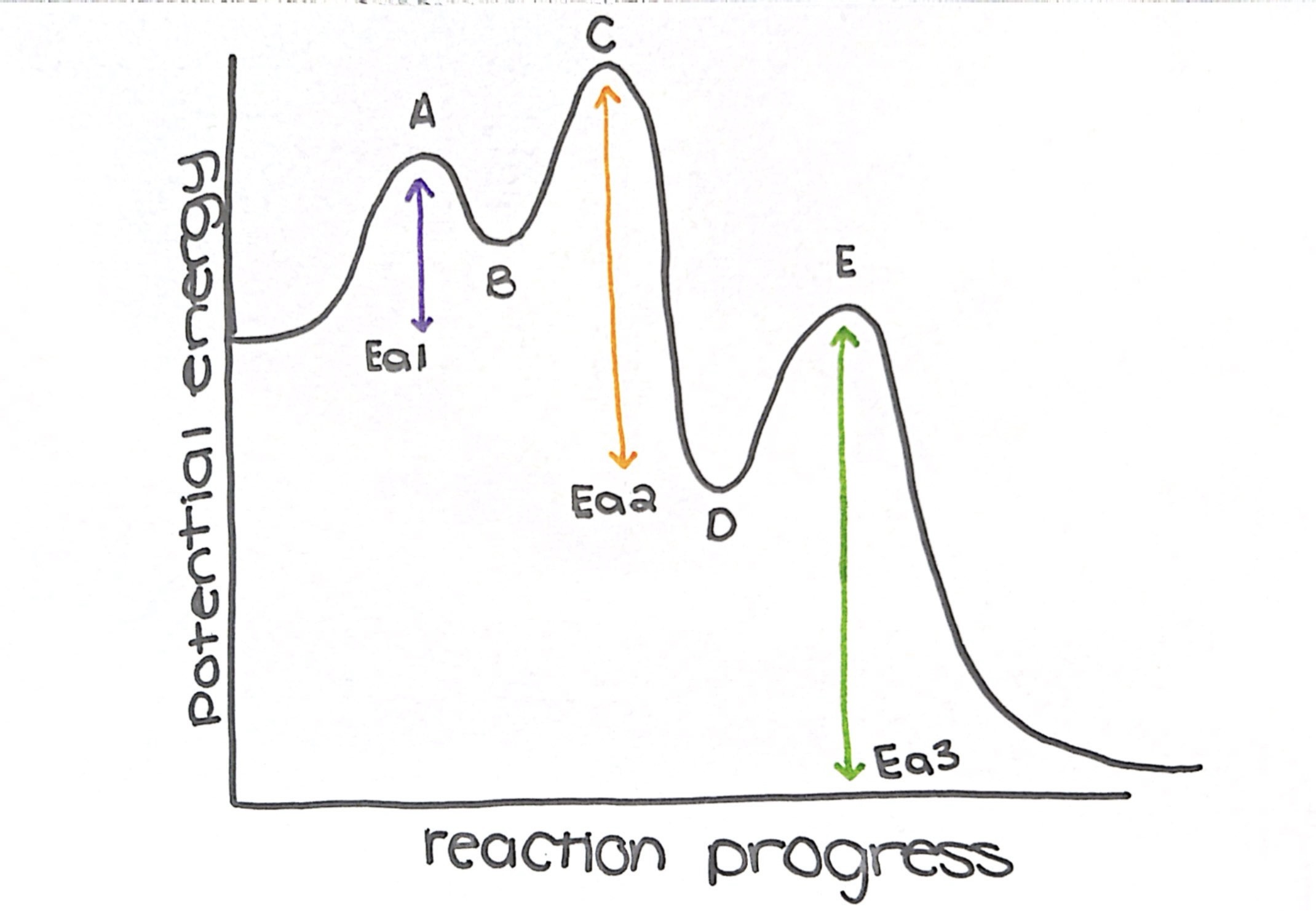
name the transition states
a, c, and e
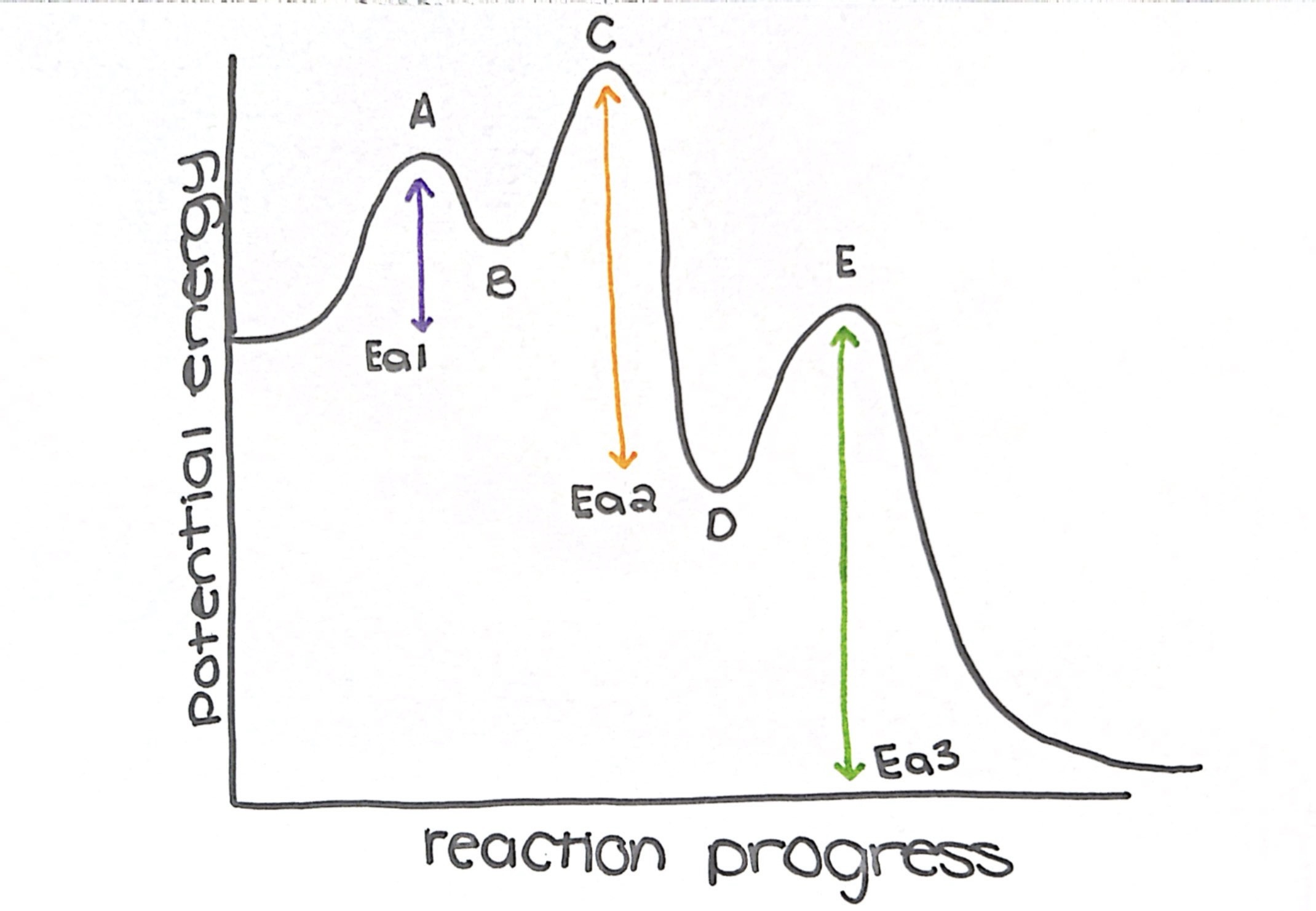
name the intermediate
b and d
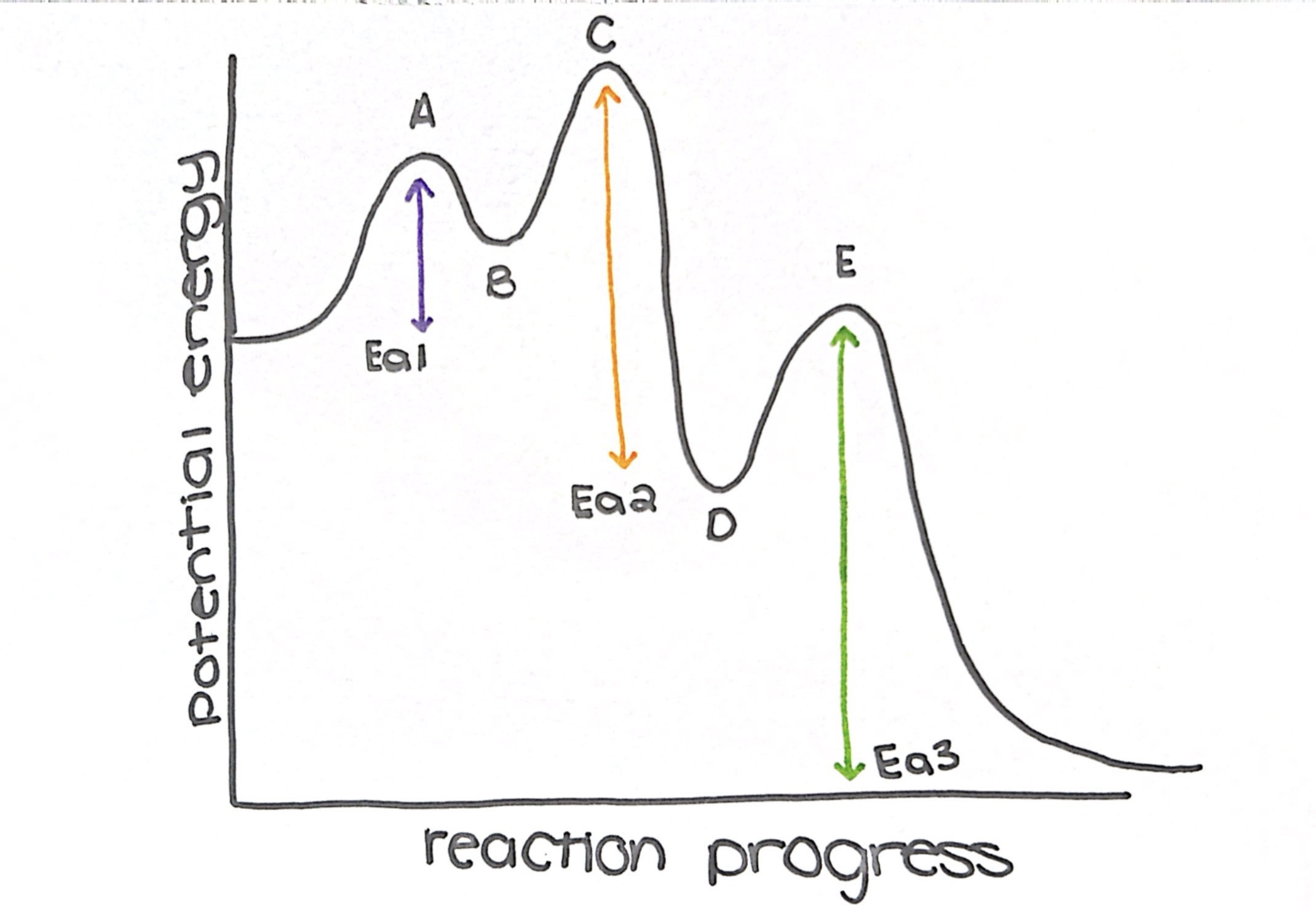
how many steps does this diagram have
3
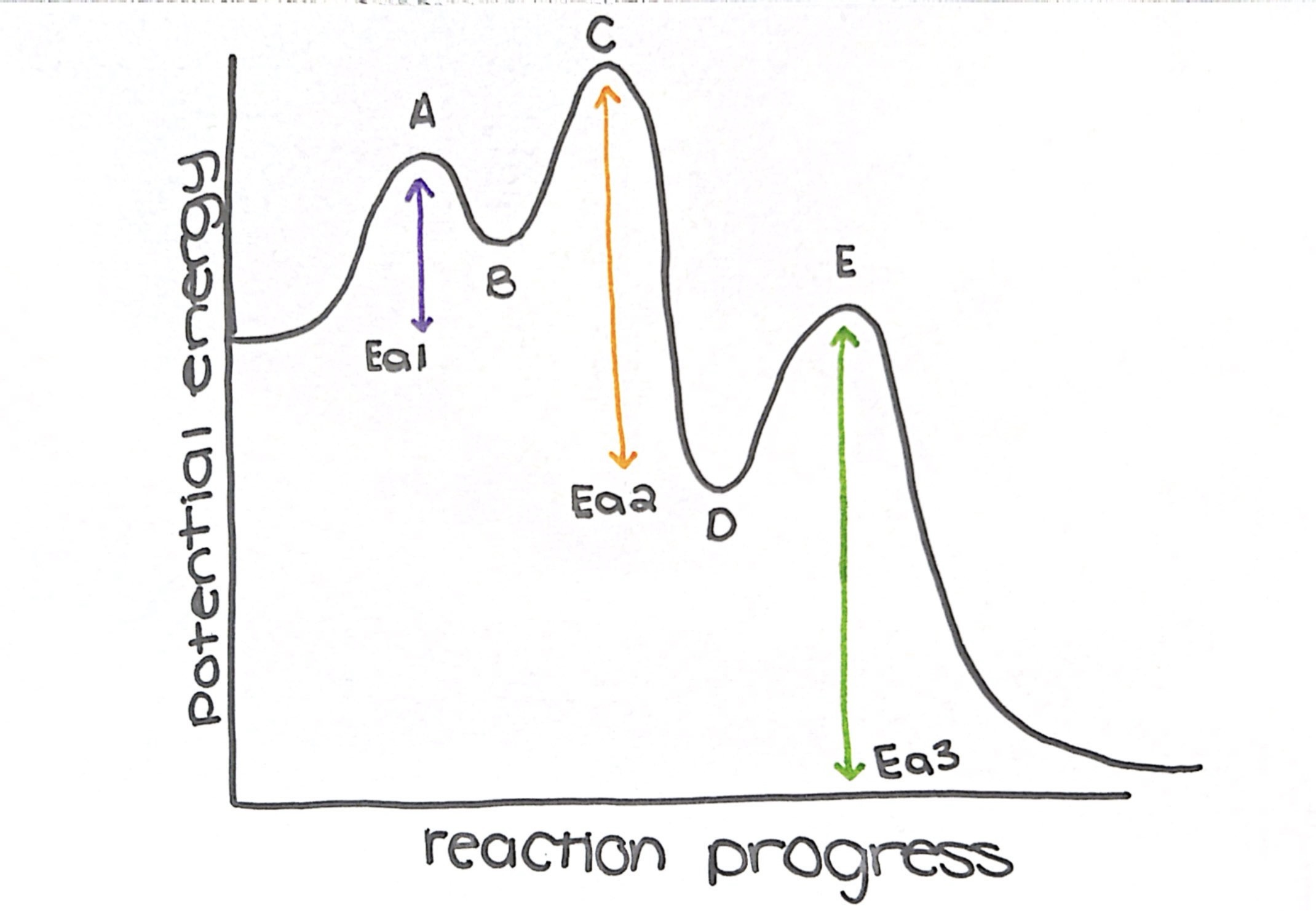
what is the rate determining step (slow step)
step 3
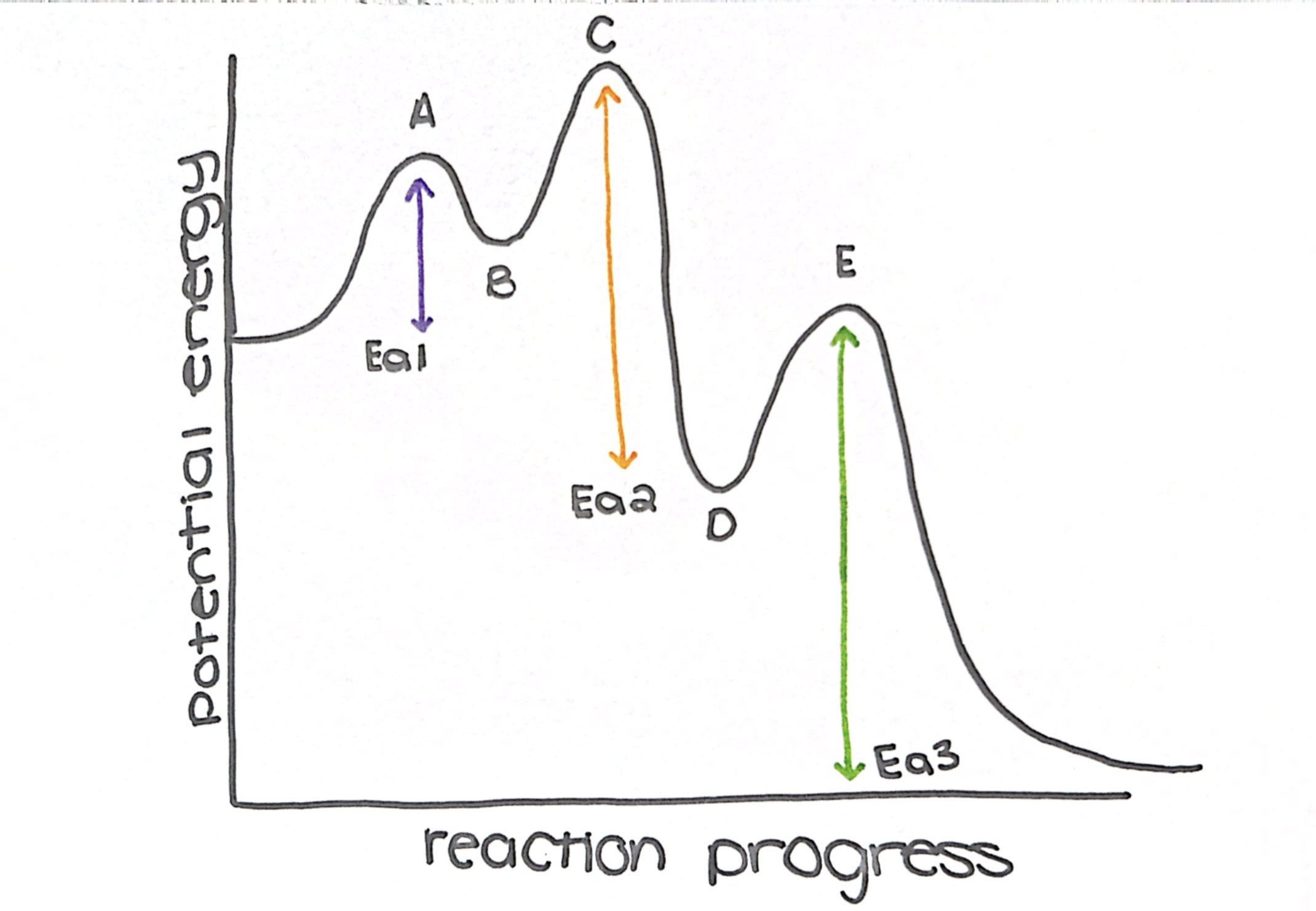
which step has the lowest Ea
step 1
when do coefficients matter
only when comparing the rates of 2 substances
when do you use stoich
to find the rate of one substance when compared to another
zero order
rate = k[A]^0 / rate = k
[A] = -kt + [A] sub 0
[A] vs t (slope = -k / k = - slope)
t1/2 = [A] sub 0 over 2k
first order
rate = k[A]
In[A] = -kt + In[A] sub 0
In[A] vs t (slope = -k / k = - slope)
t1/2 = .693 over k
second order
rate = k[A
[A] = -kt + [A] sub 0
[A] vs t (slope = -k / k = - slope)
t1/2 = [A] sub 0 over 2k
meaning of reaction order
zero - any change in concentration will not change the rate
first - change to the concentration will equally change the rate
second - the rate = (change)²
how does temp change the rate
increase in temp = increase in rate
decrease in temp = decrease in rate
how does volume change the rate
increased volume = decrease rate
decreased volume = increase rate
what is the collision theory
molecules of a concentration can only react if they collide at the correct orientation and with enough energy to cause the bond breakage and formation
average of reaction rate
rate = change in concentration / change in time
how does the addition of a catalyst change rate
the reaction increases due to the decrease of activation energy in the reaction
how does concentration change the rate
decrease in concentration = decrease in rate