3.2 physical chemistry
1/55
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
what does system mean in a chemical reaction?
the atoms and bonds involved in the chemical reaction
explain the law of conservation
the amount of energy in an isolated system remains the same
energy cannot be created or destroyed, only transferred from one form to another
what energy change occurs in breaking bonds?
endothermic (energy is taken in)
what energy change occurs in making bonds?
exothermic (energy is released)
what is an endothermic reaction?
a reaction with an overall positive enthalpy change (+ΔH)
enthalpy of product is more than enthalpy of reactants)
what is an exothermic reaction?
a reaction with an overall negative enthalpy change (-ΔH)
enthalpy of products is less than enthalpy of reactants
show an enthalpy change diagram for an exothermic and endothermic reaction
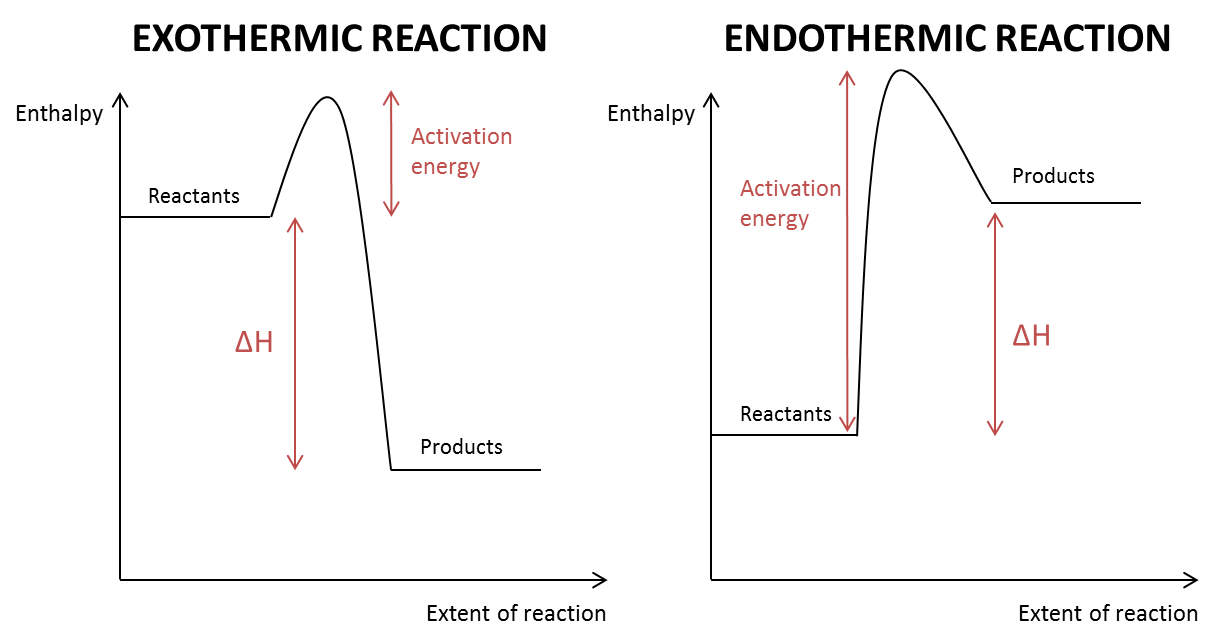
what does activation energy mean?
minimum energy needed for a reaction to take place
which way does the arrow for activation energy point on an enthalpy profile diagram?
upwards ↑
what are the standard conditions?
100kPA
298 K
what does in standard state mean?
the state an element/compound exists in at standard conditions (110kPA, 298 K)
define enthalpy change of formation
energy change that takes place when 1 mole of a compound is formed from its elements in their standard state under standard conditions
H₂(g) + ½ O₂(g) → H₂O(l)
define enthalpy change of combustion
energy change that takes place when 1 mole of a substance is completely combusted
C(s) + O₂(g) → CO₂(g)
define enthalpy change of neutralisation
enthalpy change when 1 mole of water is formed from a neutralisation reaction
what does enthalpy change of reaction mean?
energy change associated with a given reaction
how can you calculate enthalpy change from experimental data?
Q=mcΔT
enthalpy change = mass of substance being heated x specific heat capacity x change in temperature
draw a simple calorimeter
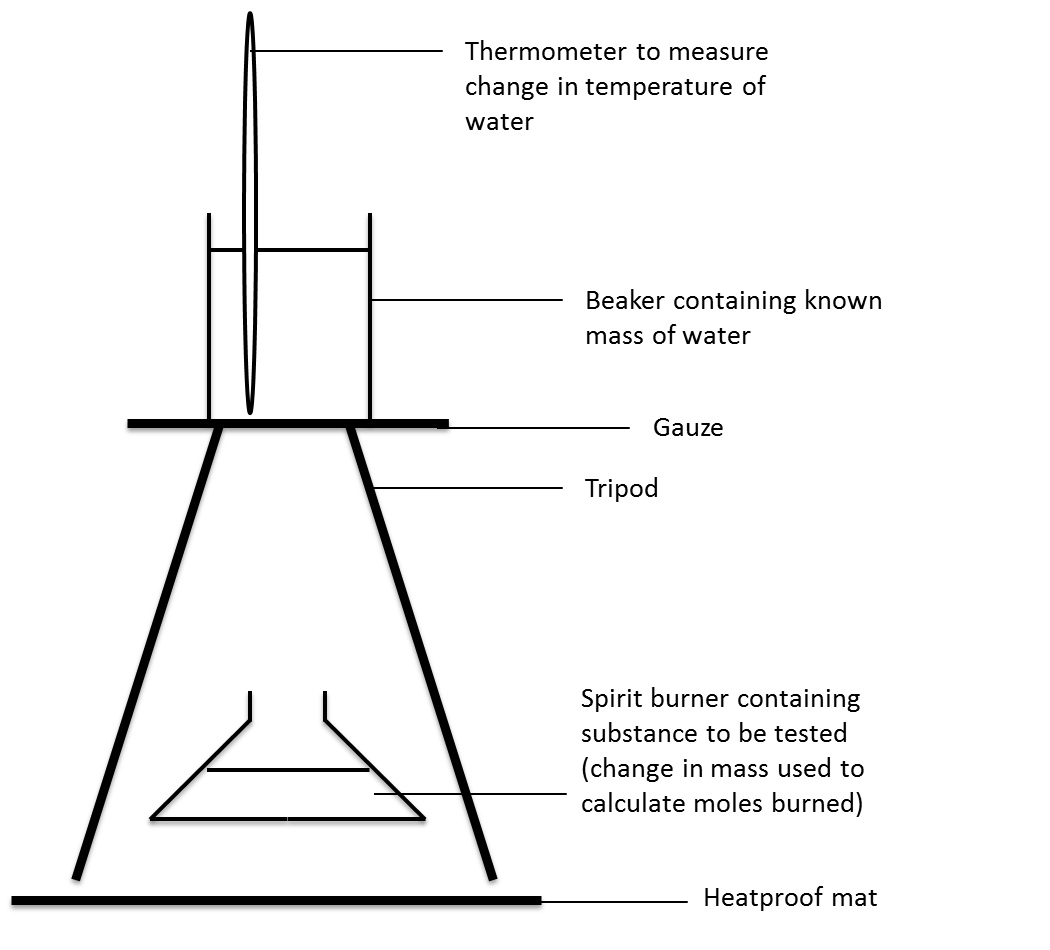
what are the advantages of using a bomb calorimeter?
minimise heat loss
pure oxygen used to ensure complete combustion
why might experimental methods for enthalpy determination not be accurate?
heat is lost to surroundings
not in standard conditions
reaction may not go to completion
what does average bond enthalpy mean?
the mean energy required to break 1 moles of bonds in gaseous molecules
why will using bond enthalpies not be as accurate as using standard enthalpy of combustion/formation?
bond enthalpies are a mean for the same bond across different molecules whereas standard enthalpy of combustion and formation apply just to that molecule therefore they are more accurate
how to calculate enthalpy change of reaction using average bond enthalpies?
Δ/H = bond enthalpies of reaction - bond enthalpies of products)
what equation is used to calculate rate?
rate = change in concentration / time
rate = (volume/mass of product/reactant formed) / time
what is the unit?
mol dm ⁻³s⁻¹
cm³s⁻¹
g s⁻¹
mol s⁻¹
what must particles do in order to react?
collide with sufficient energy (activation energy) and the correct orientation
do most collisions result in a reaction?
no
what factors affect rate of reaction?
temperature
pressure
concentration
surface area
catalyst
why does increasing temperature increase the rate of reaction?
increases rate of reaction
higher proportion of particles have energy greater than the activation energy
more successful collisions per second
increased rate
why does increasing concentration/pressure increase the rate of reaction?
increases rate of reaction
more particles in a given volume
more frequent successful collisions
increased rate
what are variables in an experiment that can be monitored to calculate the rate of reaction?
concentration of reactants or product
gas volume of products
mass of substances formed
how to calculate rate from a concentration time graph?
draw a tangent
work out gradient of tangent
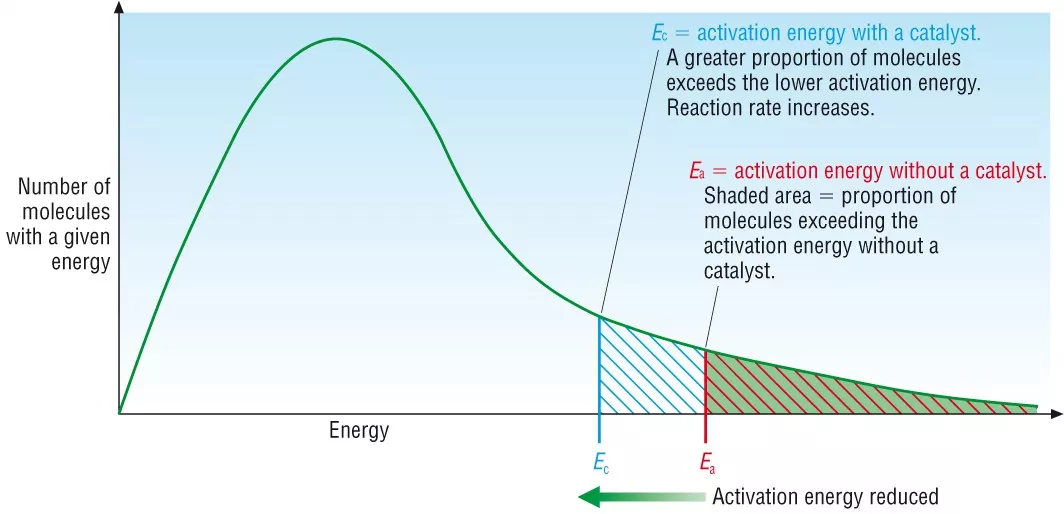
what is a catalyst?
a substance that increases rate of reaction without being used up
how do catalyst increases rate of reaction
they provide an alternate reaction pathway with a lower activation energy
due to lower activation energy, more particles have energy higher than the activation energy so more frequent successful collisions so increased rate
what does homogeneous catalyst mean?
a catalyst that is in the same phase as the reactants
(e.g. liquid catalyst with liquid reactants)
what does heterogeneous catalyst mean?
catalyst used in reaction is in different phase to the reactants
(e.g. gaseous reactants passed over a solid catalyst)
what are catalytic convertors?
they are present in vehicles to reduce toxic emission and prevent photochemical smog
define activation energy
minimum energy particles must collide with for a reaction to occur
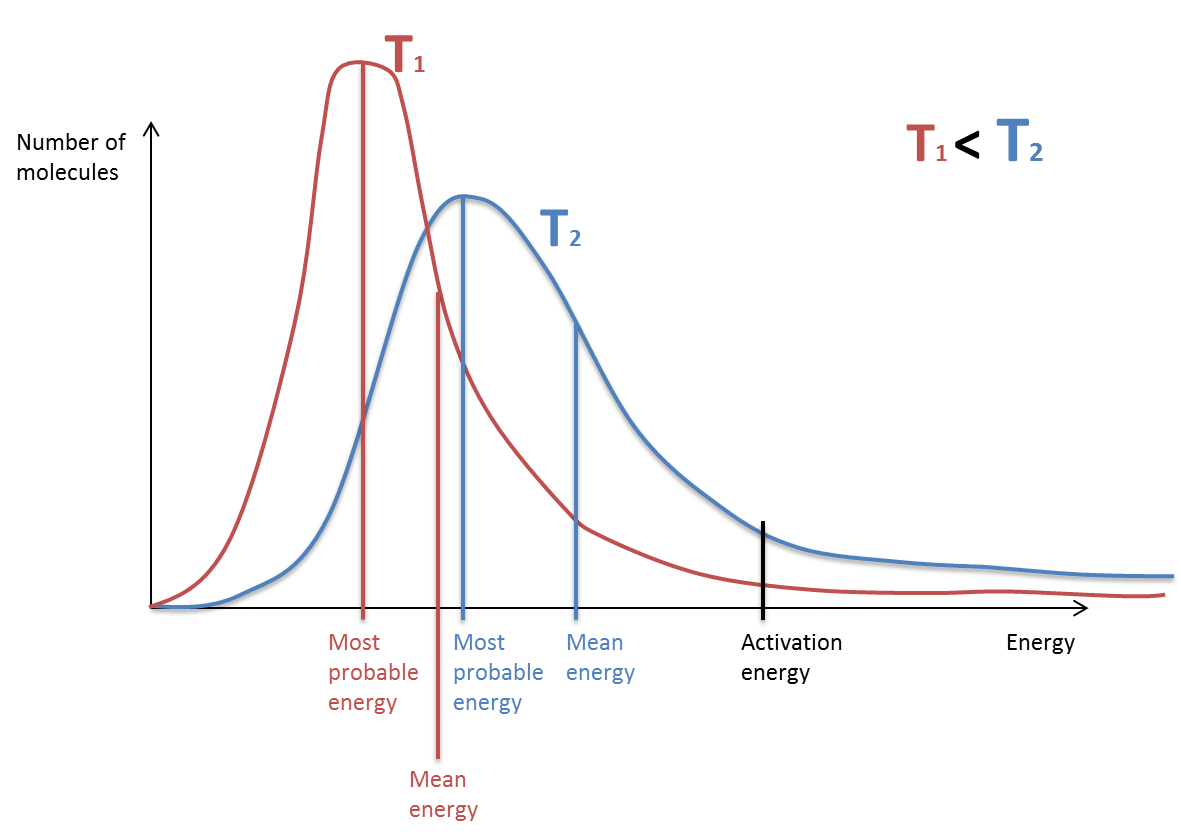
name important features of Boltzmann distribution
area under curve = total number of molecules
area under curve does not change when conditions alter
curve does not start at origin
curve does not touch or cross the energy axis
only the molecules with energy greater than activation energy can react
what are the axis in a Boltzmann distribution?
X-axis = energy
Y-axis = number of molecules with a given energy
Boltzmann curve:
Boltzmann curve with catalyst:
what does dynamic equilibrium mean?
it occurs when the rate of forward reaction equals the rate of reverse reaction and the concentration of reactants and products remain constant in a closed system
what factors can alter the position of equilibrium?
concentration of reactants or products
pressure
temperature
explain Le Chatelier's principle
if a system at equilibrium is disturbed, the equilibrium moves in the direction that tends to reduce the disturbance
if the forwards reaction is endothermic, what effect would increasing the temperature have?
equilibrium shifts to the right (endothermic direction) to decrease the temperature
if there are more molecules of products what effect would increasing the pressure have?
reduces yield of products becasue equilibrium shifts to the side with less gaseous molecules
why are the optimal conditions not always used?
because you need to reach a balance between optimum conditions and a fast rate of reaction
for example, low pressure and temperature will have a very low rate of reaction
high pressure and temperature may be dangerous and expensive to maintain
what effect does a catalyst have on the position of equilibrium?
no effect
(because catalyst affects rate of forward and reverse reactions equally)
what condition affects the value of K꜀
temperature
for the following reaction, deduce the expression for K꜀ and the units
2[A] + 3[B] + [C] ⇌[D] + 4[E]
K꜀ = [D] [E]⁴ / [A]² [B]³ [C]
K꜀ = 5(mol dm⁻³) / 6(mol dm⁻³)
= mol⁻¹ dm⁻³
what type of system is K꜀ relevant for?
homogeneous systems in equilibrium
what does K꜀ being greater or less than 1 suggest about the position of equilibrium?
greater than 1 = over to the right
less that 1 = over to the left
what effect does decreasing the temperature of an endothermic reaction have on K꜀?
K꜀ decreases
the equilibrium shifts in the exothermic direction (left) to increase the temperature
what effect does increasing the temperature of an endothermic reaction have on K꜀?
K꜀ increases
becasue equilibrium shifts in the edothermic direction (right) to decrease temperature
what effect does decreasing the temperature of an exothermic reaction have on K꜀?
K꜀ increases
equilibrium shifts in the exothermic direction (to the right) to increase temperature
what effect does increasing the temperature of an exothermic reaction have on K꜀?
K꜀ decreases
equilibrium shifts in the endothermic direction (left) to decrease temperature