chemistry module 6 benzene and aromatic compounds
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
8 Terms
what are the 2 models of benzene
Kekule, delocalised
describe the kekule model
a hexagonal ring of six carbon atoms with alternating single and double C-C bonds
It suggests that the π-electrons in benzene are localised within these three double bonds
describe the delocalised model of benzene and draw a diagram
It proposes that the p-orbitals of all six carbon atoms overlap sideways, both above and below the plane of the ring
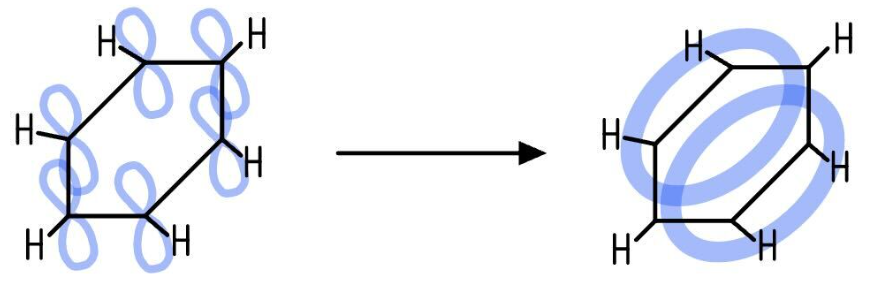
This overlap forms a continuous ring of electron density, creating a delocalised π-system where the six π-electrons are spread over the entire ring
how do bond lengths support the delocalised model over the kekule model
x-ray diffraction analysis shows that the bond lengths of all 6 carbon-carbon bonds are equal. whereas the kekule model would have alternating short double bonds and long single bonds
how do molecular shape & bond angles support the delocalised model over the kekule model
x-ray diffraction shows that benzene is a perfectly planar hexagon. the perfect hexagon only exists if all 6 bonds are equal in length
how does enthalpy of hydrogenation support the delocalised model over the kekule model
The hydrogenation of cyclohexene (one C=C bond) has an enthalpy change of -120 kJ mol-1
The Kekulé structure (with three C=C bonds) would therefore be expected to have an enthalpy of hydrogenation of -360 kJ mol-1
However, the experimental value for benzene is only -208 kJ mol-1
This means benzene is significantly more stable (less exothermic) than the Kekulé structure suggests, due to the energy of the delocalised π-system.
halogenation in benzene
In benzene, the electron density of the six π-electrons is delocalised and spread out over the entire ring
This means the electron density at any one point is lower than in an alkene's C=C bond
So, it is not strong enough to polarise the Br2 molecule
Therefore, benzene does not react with bromine under normal conditions
Benzene resists addition reactions as this would disrupt the very stable delocalised ring
Instead, it undergoes substitution reactions
This requires a halogen carrier catalyst, such as AlBr3