CHEM 237 EXAM
1/120
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
121 Terms
Enzyme Classifications
Class | Rxn Type |
Oxidoreductases | Redox rxn |
Transferases | Transfer of functional group |
Hydrolases | Hydrolysis rxn |
Lyases (synthase) | Breaking and forming bonds |
Isomerases | Isomerization rxns |
Ligases (synthetase) | Bond formation typically coupled w/ ATP hydrolysis |
Translocase | Movement of molecules across/within membranes |
Michaelis-Menten Kinetics Assumptions:
We examine early times (beginning) in the rxn when [P] is low. So, we can ignore the reverse rxn (ignore k2)
[S] >> [E], sp we can assume the [ES] doesn’t alter [S]
A steady state exist such that the rate of ES formation = rate of ES consumption
Step 1 is faster than step 2, k1 >> k2, and k-1 >> k2, so step 2 is rate limiting, rate = k2[ES]
[ES] is at a steady state, d[ES]/dt = 0
[E]t = [E] + [ES], there are no alternate forms of the enzyme
Double-Displacement (Ping Pong) Bisubstrate Reaction
A functional group X of the first substrate A (= P-X) us displaced from the substrate by the enzyme E to yield the first product P and a stable enzyme form F (= E-X) in which X is tightly (often covalently) bound to the enzyme (Ping).
In the second stage of the rxn, X is displaced from the enzyme by the second substrate B to yield the second product Q (= B – X), thereby regenerating the original form of the enzyme, E (Pong). (e.g. transaminase).
Oseltamivir (Tamiflu)
The anti-influenza drug oseltamivir (Tamiflu) is hydrolyzed int he liver to yield oseltamivir carboxylate.
Oseltamivir carboxylate is a competitive inhibitor of influenza neuraminidase that binds to the enzyme’s active site.
How are Enzyme Mechanisms Determined?
Analysis of a mechanism of non-enzymatic rxn. Investigation of pH dependences of enzyme-catalyzed rxn. Analysis of stable intermediates or of kinetic data w/ multiple substrates.
Isotope shift experiment: e.g. replace H w/ D, and if rxn slows down then cleavage of that bond is part of the mechanism.
Example of enzyme pH dependency: pH affects charge of any ionizable group participating in catalysis and the overall stability of the enzyme (tertiary structure).
Enzyme Reaction Mechanisms
Acid-base catalysis: specific residues act as proton donors or acceptors to facilitate bond breaking/forming
Covalent catalysis: a nucleophilic residue on the enzyme forms a temporary, transient covalent bond w/ the substrate
Metal ion catalysis: metal ions in the active site help orient the substrate, stabilize negative charges, or participate in oxidation-reduction.
Proximity and orientation effects: the enzyme binds substrates in a specific, close, arrangement, increasing the reaction rate by reducing entropic effects.
Electrostatic catalysis: active site residues stabilize the charged transition state more than the substrate, reducing the activation energy.
Note: enzymes can use more than 1 type of catalytic mechanisms
RNase A: Concerted Acid-Base Catalysis
Bovine pancreatic RNase A catalyzes endonucleolytic cleavage of 3’-end of pyrimidine nucleotides: (Cytosine – C, Uracil – U).
Concerted acid-base catalysis is a mechanism where an acid and a base act simultaneously on a substrate in the same transition state, often described as a “push-pull” mechanism – one pushes and the other pulls a proton.
Mechanism of Uncatalyzed Hydrolysis of RNA Phosphodiester Bond
In basic (alkaline) conditions (pH > 7), RNA is rapidly (degraded) hydrolyzed via 2 steps intramolecular transesterification rxn:
2’-O- acts as a nucleophile attacking phosphodiester bond producing a 2’-3’ cyclic nucleotide intermediate
The 2’,3’-cyclic phosphate intermediate is hydrolyzed to form either a 2’-phosphate or a 3’-phosphate product
Mechanism of RNase A Catalyzed Hydrolysis of RNA Phosphodiester Bond
It is a 2-step concerted general acid base catalysis. The enzyme catalyzed rxn also goes through 2’-3’ cyclic nucleotide intermediate. The enzyme catalyzed hydrolysis always produces only the 3’ phosphate product.
RNase A is pH dependent. pKa values are roughly 6. Histidine side chains are catalyzed in this reaction.
The RNase A Mechanism
Step 1: His12 acts as a general base and His119 acts as a general acid. The 2’ O- does an SN2 attack on the phosphodiester bond forming 2’,3’-cyclic nucleotide intermediate.
Substrate: RNA
Products: 2’,3’-cyclic nucleotide intermediate & 5’-OH RNA fragment
Note: H+ from His110 becomes 5’-OH in the product
Step 2: His12 acts as a general base, and His119 acts as a general acid. The OH- of water does SN2 attack to promote hydrolysis of 2’,3’-cyclic nucleotide intermediate.
Substrate: 2’,3’ - cyclic nucleotide intermediate & H2O
Product: 3’ - phosphate RNA fragment
Note: OH- from H2O becomes part of the product, H+ from H2O is on His 119
Metal Ions Participate in Catalysis in 3 Ways
By binding to substrates to orient them properly for reaction
By mediating oxidation-reduction rxn through reversible changes in the metal ion’s oxidation state
By electrostatically stabilizing or shielding negative charges
Mechanism of Carbonic Anhydrase
Zn2+ is coordinated in the active sites by His residues. Chelating agents that bind tightly to metal ions diminish carbonic anhydrase activity. Zn2+ polarizes H2O creating OH- that acts as a nucleophile. The enzyme’s catalytic site is regenerated by the binding of second H2O to the Zn2+ ion.
The proton produced in the reaction is shuttled to the enzymes surface through base catalysis facilitated by a fourth His residue (His 64)
Lysozyme & Structure
Lysozyme is an antimicrobial enzyme that destroys bacterial cell walls. It hydrolyzes B (1 --> 4) glycosidic linkages from NAM to NAG in bacterial cell wall polysaccharides called peptidoglycans.
Lysozyme's catalytic site was identified through X-ray crystallography. Its active site cleft is long enough to accommodate an oligosaccharide of 6 residues.
Identification of Lysozyme Cleavage Site
The product of the hydrolysis reaction had 18O bonded to the C1 atom of its newly liberates reducing terminus, thereby demonstrating that bond cleavage occurs b/w C1 and O1.
Lysozyme-Substrate Interactions
The only functional groups int he immediate vicinity of Lysozyme’s reactive center that have the required catalytic properties are the side chains of Asp52 & Glu35. These side chains, which are disposed to either side of the glycosidic linkage to be cleaved have markedly diff environments.
Diff in Properties of Asp52 & Glu35
Asp52 is in a polar environment and has a normal pKa value. Glu35 is a non-polar environment and has an abnormally high pKa value, remains protonated even at hgiher pH values.
Microenvironment can radically alter pKa values.
Mechanisms of Nonenzymatic Acid-Catalyzed Acetal Hydrolysis
Nonenzymatic acetal hydrolysis is an acid-catalyzed reaction that involves the protonation of a reactant O followed by cleavage of its C–O bond. Mechanism involves formation of positively charged oxonium intermediate.
Mechanism of Lysozyme Reaction
Based on the structure the mechanism was proposed in which Glu 35 acts as a general acid catalyst and Asp 52 stabilizes the charged intermediate and then forms a covalent intermediate. The structure of the sugars and steric interference and a network of hydrogen bonds stabilize the D ring in the half chair configuration
STUDY DOC
Experimental Evidence of Lysozyme Mechanism
The double-displacement mechanism allows the incoming water molecule to attach to the same face of the D residue as the E residue it replaces. Consequently, the configuration of the D residue is retained. A single-displacement reaction, in which water directly displaces the leaving group, would invert the configuration at C1 of the D ring between the substrate and product, a result that is not observed experimentally.
X-ray studies confirm that the δ -lactone analog of (NAG)4 binds to lysozyme in such a way that the lactone ring occupies the D subsite in a half-chair-like conformation.
The X-ray structure of lysozyme bound to NAG2FClcF reveals a long covalent bond between C1 of the D ring and a side chain carboxyl O of Asp52.
Serine Proteases
Examples are trypsin, chymotrypsin, and elastase.
Chymotrypsin: the 3 invariant resides (Asp-His-Ser) form a HB constellation referred to as the catalytic triad.
Specificity Pockets of Serine Proteases
The X-ray structures of the 3 enzymes suggest the bases for their differing specificities. Chymotrypsin has large hydrophobic pockets – binds AA w/ large hydrophobic side chains. Trypsin has Asp at the bottom of the pocket – binds positively charged AA (Lys, Arg). Elastase has narrow binding pocket – binds small hydrophobic AA (Ala, Gly, sometimes Val)
Determining Serine Mechanism
DIPF Irreversibly Inactivates Serine Proteases: DIPF reacts only w/ Ser195 of chymotrypsin, but not w/ any other Serine in the protein.
Chymotrypsin has High Affinity for TPCK: Chymotrypsin specifically binds tosyl-L phenylalanine chloromethylketone (TPCK) b/c of its resemblance to a Phe residue.
pH dependence: There is a group w/ a pKa of ~7 that mut be deprotonated for activity. It is His57.
Overview Mechanism of Serine Proteases
The O of Ser attacks carbonyl C of peptide bond (nucleophilic attack), cleaving peptide bond and forming covalently attached acyl-enzyme intermediate
The acyl-enzyme intermediate is cleaved by water to generate the new carboxyl group and regenerate the free enzyme
Mechanism of Serine Proteases
Step 1: Ser195 nucleophilically attacks the scissile peptide’s carbonyl group to form the reaction’s tetrahedral intermediate (covalent catalysis). The resulting intermediate has a well-defined, although brief, existence. Much of chymotrypsin’s catalytic power derives from its preferential binding of the transition state leading to this intermediate (transition state binding catalysis).
Step 2: The tetrahedral intermediate decomposes to the acyl–enzyme intermediate under the driving force of proton donation from N3 of His57 (general acid catalysis).
Step 3: The amine leaving group (R′NH2, the new N-terminal portion of the cleaved polypeptide chain) is released from the enzyme and replaced by water from the solvent.
Step 4: The acyl–enzyme intermediate is highly susceptible to hydrolytic cleavage as His57 withdraws a proton from water (general base catalysis), yielding a hydroxide ion. That ion attacks the carbonyl of the acyl group, yielding a second tetrahedral intermediate.
Step 5: Reformation of the carbonyl group yields the carboxylate product (the new C-terminal portion of the cleaved polypeptide chain), which dissociates from the enzyme, thereby returning it to its initial state. In this process, water is the attacking nucleophile and Ser195 is the leaving group.
What Makes Ser195 a Good Nucleophile?
X-ray studies indicate that Ser195 is ideally positioned to carry out this nucleophilic attack (proximity and orientation effects).
Interaction of Ser195 with His57 polarizes the Ser hydroxyl group and orients the side chain.
When substrate binds, Ser is deprotonated by His57 (general base catalysis), which generates an alkoxide ion (RO−), a powerful nucleophile.
This process is aided by Asp102, which is hydrogen bonded to His57 and both orients and electrostatically stabilizes His to act as a proton acceptor in the direction of Ser (electrostatic catalysis).
Asp102, His57 and Ser195 together are called a catalytic triad. The pKa of His 57 is ~12
Transition State Stabilization in Serine Proteases
Detailed comparisons of the X-ray structures of several serine protease–inhibitor complexes have revealed that the active site of chymotrypsin has groups to stabilize the CO- intermediate in the transition state: the oxyanion hole
Trypsin-BPTI Complex
The portion of BPTI (bovine pancreatic trypsin inhibitor) in contact w/ the trypsin active site resembles a bound substrate. A specific Lys side chain of BPTI occupies the trypsin specificity pocket.
Activation of Trypsinogen to Trypsin
Enteropeptidase excises the N terminal hexapeptide from trypsinogen (zymogen – inactive form of an enzyme) by specifically cleaving its Lys15 – Ile16 peptide bond.
The Aspartic Proteases
Have 2 catalytic Asp residues in the active site acting as general acid-base catalysts. Most aspartic proteases have a tertiary structure consisting of 2 lobes (N-terminal and C-terminal) w/ approximate 2-fold symmetry.
Example: pepsin (stomach protease active at low pH) and HIV-1 protease
HIV Protease & Inhibitors
HIV-1 protease cleaves the polyprotein products of the HIV genome. HIV-1 protease is a homodimer. Active site is 2 folded symmetric.
HIV protease inhibitors work by specifically binding to the active site by mimicking the tetrahedral intermediate.
Catalytic Mechanism of HIV Protease
The difference in pKa values of the Asp residues is crucial for catalysis.
One Asp (right side on the slide) has a relatively low pKa and acts as a general base, accepting a proton from water and forming OH- that acts as a nucleophile.
Second Asp (left side on the slide) has a relatively high pKa and acts as a general acid, donating a proton.
Control of Enzyme Activity
Enzyme activity may be controlled by covalent modification. Control by covalent modification usually involves protein phosphorylation or dephosphorylation.
Example: the enzymatic activity of glycogen phosphorylase is controlled by its phosphorylation/dephosphorylation as well as by the influence of allosteric effectors.
Control by Covalent Modification: Phosphorylation
In eukaryotes, the most common covalent modifications are phosphorylation and dephosphorylation of the OH group of a Ser, Thr, and Tyr residue. Phosphorylated enzymes can either have higher or lower activity. Enzymes can have multiple phosphorylation sites.
Example of enzyme regulation by phosphorylation: glycogen phosphorylase
Glycogen phosphorylase or simply phosphorylase catalyzes the phosphorolysis (breakdown) of glycogen during the fasting to free up glucose.
Conformational Changes: Glycogen Phosphorylase
Glycogen phosphorylase has 2 conformational states, the enzymatically active R state and the enzymatically inactive T state
Control by phosphorylation: the phosphorylation of Ser 14 promotes phosphorylases.
T (inactive) --> R (active) conformational change.
Monosaccharides (Simple Sugars)
Class of organic compounds with the formula (CH2O)n. More specifically, they are classified as polyhydroxy aldehydes (aldoses) or polyhydroxy ketones (ketoses).
Monosaccharides can be classified by the # of C: triose (3), tetrose (4), pentose (5), hexose (6), heptase(7).
Monosaccharides have chiral C, so they have different enantiomers (non-superimposable mirror images of each other).
In the Fischer projection of a sugar, if the chiral center furthest away from the carbonyl C has –OH group on the right then this is a D form. If it is on the left then this is an L form. D-sugars are the predominant form found in nature.
Sugars with 3+ C have multiple chiral centers and can exists as diastereomers (stereoisomers that are not mirror images of each other). Diastereomers that diff in configuration at exactly one chiral center are known as epimers.
D-Glucose (CH2O)6
C2, C3, C4 & C5 are chiral centers
- D-Glucose is one of 24 = 16 possible stereoisomers for 6 C aldose
- D-Ketoses with 3-6 C have less chiral C
Epimers of D-Glucose: D-Mannose and D-Galactose
Chemical Rxns of Monosaccharides:
Alcohol + Aldehyde <=> Hemiacetal
Alcohol + Ketone <=> Hemiketal
Aldehydes and ketones react with alcohols via nucleophilic addition mechanisms forming hemiacetals and hemiketals.
Cyclization of Glucose & Fructose
Aldohexose can exist in 6 membered cyclic form called pyranose when C5-OH attacks C1-carbony C.
Ketohexose can also exists in pyranose form when C6-OH attacks C2-carbonyl C, but it also can form a 5-membered cyclic form known as furanose when C5-OH attacks C2-carbonyl C. 5 or 6 membered ring are the most stable.
NOTE: sugars are named after their parent ring structure
a and B Anomers can Interconvert
a-D-glucopyranose and B-D-glucopyranose are anomers of each other b/c they diff only in their configuration about the anomeric carbon C1 (new chiral center present only in cyclic form).
When C1-OH group is “below” the ring, than these are a-anomers, when it is above than they are B-anomers. Conversion of one anomer to another is known as mutarotation.
Chair Conformations of B-D-Glucose
Equatorial positions are when functional groups are extended alternately above or below the ring.
Axial positions are when the groups occupy more crowded vertical positions.
Reactions of Monosaccharides
Tollen’s Test (Silver Mirror): aldehyde reduces Ag+ to Ag
2 [Ag (NH3)2 ]+ + RCHO + H2O --> 2 Ag(s) + 4 NH3 + RCO2H + 2H+
Fehling’s test: aldehyde reduces Cu2+ to Cu+
Aldehyde + Fehling’s solution --> Carboxylic acid + red precipitate
These reactions are used to detect presence of reducing sugars which are sugars that have a free aldehyde. Rings have no free aldehyde but they can open up to form a free aldehyde group.
Glucose Coloimetric Assay
Enzymatic methods are used to quantify reducing sugars such as glucose. The enzymes glucose oxidase catalyzes the conversion of glucose to glucono-d-lactone and hydrogen peroxide. Hydrogen peroxide oxidizes organic molecules into highly colored compounds. Concentrations of such compounds is measured colorimetrically.
Electrochemical detection is used in portable glucose sensors.
Sugar Modifications: Oxidation of Aldehyde Yields Aldonic Acids
Aldonic acids are named by appending the suffix –onic acid to the root name of the parent aldose, glucose. Gluconic acid occurs naturally in fruit, honey, and wine. Used as food additive (E574) for acidity regulation.
The gluconate anion chelates Ca2+, Fe2+, and other metals. It is used in cleaning produces, where it dissolves mineral deposits.
Glucose Oxidation: Electrochemical Assays to Measure Glucose
Glucose is oxidized to gluconic acid. Ferricyanide is reduced to ferrocyanide. At the electrode surface ferrocyanide is oxidized back to ferricyanide.
The current required to oxidize ferrocyanide is proportional to the glucose [ ]. used in portable glucose sensors and monitors.
Sugar Modifications: Oxidation of Primary Alcohol Yields Uronic Acids
Uronic acids, are named by appending –uronic acid to the root name of the parent aldose, for example, D-glucuronic acid.
D-glucuronic acid is involved in detoxification in the liver, where it conjugates toxins, drugs, bilirubin, and steroids, making them more water-soluble for excretion.
Sugar Modifications: Reduction of Aldehyde Yields Alditols (Sugar Alcohols)
Ribitol is a component of flavin coenzymes. Xylitol is a sweetener that is used in “sugarless” gum and candies. Glycerol is a component of storage lipids triacylglycerols. Cyclic polyhydroxy alcohol myo-Inositol is membrane lipid component.
Sugar Modifications: Reduction of Alcohol Yields Deoxy Sugars
B-D-deoxyribose is the sugar component of DNA’s sugar-phosphate backbone.
L-Fucose is one of the few L-sugar components of polysaccharides.
Sugar Modifications: Amino Sugars
Examples include glucosamine and N-acetyl glucosamine.
Found in human cartilage, synovial fluid, and in the covering of shellfish. It is a building block for glycosaminoglycans and proteoglycans, which are crucial structural components of articular cartilage.
Sugar Modifications: Sialic Sugars
N-Acetylneuraminic acid is an important constituent of glycoproteins and glycolipids.
N-acetylneuraminic acid and its derivates are often referred to as sialic acids.
The bond connecting the anomeric C to the alcohol O is termed a glycosidic bond.
Formation of O-Glycosides
The anomeric group of a sugar can condense with an alcohol to form a- and B-glycosides.
O-Glycosides can no longer open and the stereochemistry of the anomeric C is fixed and the compound is no longer a reducing sugar. The reverse rxn of hydrolysis of the glycosidic bond happens during complex carbohydrate breakdown.
Disaccharides
Lactose has a free anomeric C on its glucose residue and is therefore a reducing sugar.
Maltose, or malt sugar, is a disaccharide of 2 glucose molecules. It is an important sugar in germinating seeds and is a reducing sugar.
Sucrose is a table sugar. The anomeric C of each sugar (C1 in glucose and C2 in fructose) participates in the glycosidic bond, and hence sucrose is a nonreducing sugar.
Note: 2 anomeric C = unable to reopen ring for free ketone or aldehyde = non-reducing sugar
Polysaccharides
The principal storage polysaccharides are amylose & amylopectin, which together constitute starch in plants, and glycogen, which is stored in animal and microbial cells. Both starch and glycogen are stored in granules within cells.
Glycogen is deposited in the liver, which acts as a central energy storage organ in many animals. It is also abundant in muscle tissue, where it is more immediately available for energy release. It and the components of starch – amylose and amylopectin – are storage polysaccharides.
Amylose is linear: amylopectin and glycogen are branched.
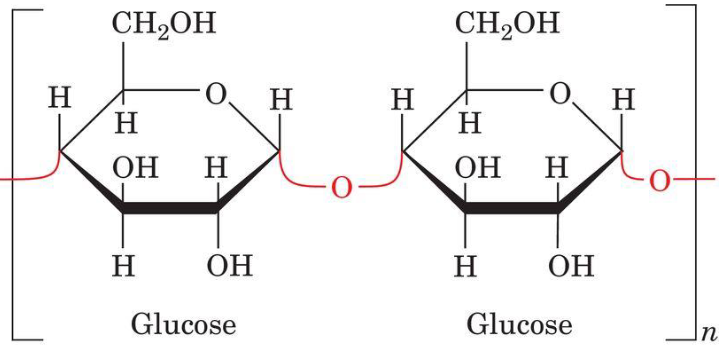
a-Amylose: a (1 --> 4) linked D-Glucose
a (1 --> 4) linked D-Glucose
It is a linear polymer of several thousand glucose residues linked by a (1 --> 4) bonds. There are irregularly aggregating helically coiled conformations
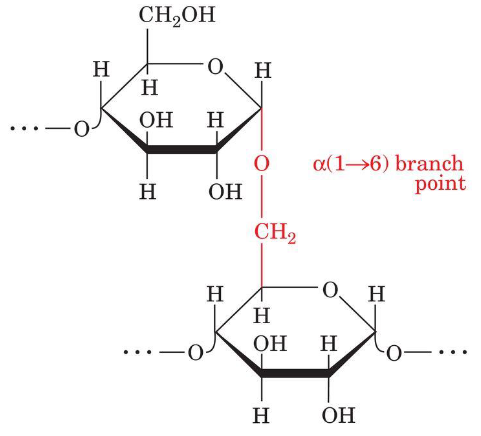
Amylopectin: a (1 --> 6) - branches
It consists mainly of a (1 --> 4) - linked glucose residues but is a branched molecule with a (1 --> 6) branch, points every 24 to 30 glucose residues on average.
Glycogen Granules in Liver Cells
Glycogen is prevalent in skeletal muscle and in liver, where it occurs as cytoplasmic granules. Glycogen is more branched than starch, i.e. has more a (1 --> 6) bonds.
Cellulose Fibers
Cellulose, the primary structural component of plant cell walls accounts for over half of the C in the biosphere. It is entirely linear.
Cellulose is B (1 --> 4) - Linked D-Glucose
It is a linear polymer of up to 15,000 D-glucose residues linked by B (1 --> 4) glycosidic bonds. Humans lack an enzyme to break scuh bond.
Cellulose is tightly packed and has a fully extended conformation. Every C in glucose has an O & H, allowing for HB. Branching will break the HB in the polymer, but since Cellulose is linear, it can form many HB. Singular HB are weak but thousands made it strong.
Chitin
It is a B (1 --> 4) - Linked N-Acetyl-D-Glucosamine.
It is the principal structure component of the exoskeletons of invertebrates and is the second most abundant biomolecule.
Glycosaminoglycans: Repeating Disaccharide Units
Hyaluronic acid: acts as a lubricant and cushioning agent in the joints, eyes, and skins
Chondroitin sulfate: structural component of carilage and connective tissue
Dermatan sulfate: plays role in regulating growth factors, coagulation and wound repair in skin
Keratan sulfate: acts as a crucial hydrate buffer, maintaining tissue structure, transparency in the cornea
Heparin: produced by mast cells and basophils in all mammals and acts as a natural anticoagulant
Pectins: a (1 --> 4)-Linked Galacuronate Residues with Rhamnose
Pectins are major components of plant cell walls. It binds cells together, ensures structural integrity, and enables cell adhesion.
Proteoglycan: Central Strand with Numerous Projections
Proteins and glycosaminoglycans in the extracellular matrix aggregate covalently and noncovalently to form a diverse group of macromolecules known as proteoglycans.
Bacterial Peptidoglycans: Repeating Unit
The cell walls of bacteria consist of covalently linked polysaccharide and polypeptide chains, which form a cage like macromolecule that completely encases the cell.
Peptidoglycan-Specific Antibiotics
Penicillin contains a thiazolidine ring fused to a B-lactam ring. A variable R group is bonded to the B-lactam ring via a peptide link. It targets the synthesis of bacteria cell walls.
G+ & G- Bacterial Cell Wall
G+ bacteria have a thick peptidoglycan cell wall surrounding their plasma membrane
G- bacteria have a thin cell wall covered by a complex outer membrane
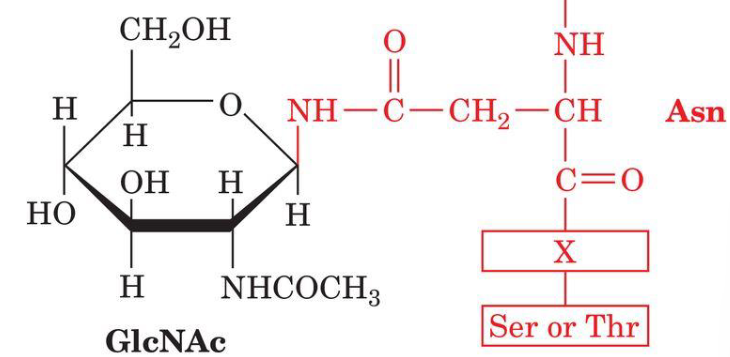
N-linked Oligosaccharides
N-linked glycosylation of proteins occur at Asn. X is any amino acid except P and only rarely Asp, Glu, Leu or Trp.
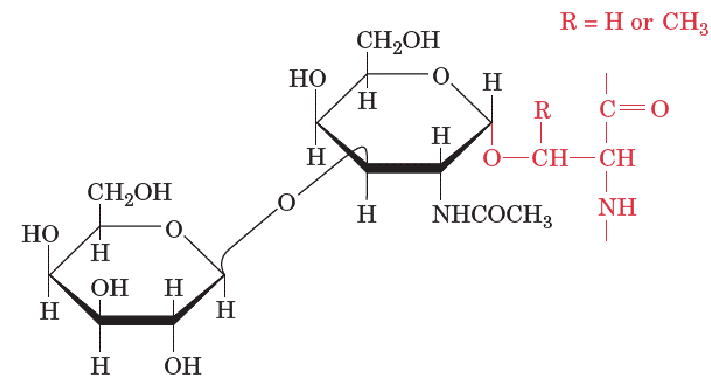
O-Linked Oligosaccharides
O-Linked glycosylation of proteins occur at Ser, Thr, and Tyr (all involved in phosphorylation).
They vary in size from a single galactose residue in collagen to the chains of up to 1000 disaccharide units in proteoglycans
Oligosaccharides are conformationally Mobile
Erthrocyte Glycocalyx
All cells are coated with sugars in the form of glycoconjugates such as glycoproteins and glycolipids. These sugars form glycocalyx – dynamic, gel-like, carbohydrate-rich layer covering the surface of cells, particularly vascular endothelium, composed of glycoproteins, proteoglycans, and glycolipids.
Lectins are a type of carbohydrate-binding protein.
Blood Types: Oligosaccharides found in the A, B, and H antigens
Type A has A-antigen. Type B has B-antigen. Type AB has both A and B-antigens. Type O has the H antigen which is the precursor oligosaccharide of A and B antigens.
Fatty Acids
Fatty acids are carboxylic acids with long-chain hydrocarbon side groups. In higher plants and animals, the main fatty acid residues are C16 and C18 types: palmitic, oleic, linoleic, and stearic acids.
They are amphipathic molecules: mainly hydrophobic with polar head (carboxylic group). Also, contains double bonds mostly in cis form.
In common saturated fatty acids, the solubility of the aqueous solution decreases as chain length increases. Longer tail = stronger LDF = higher MP
In common unsaturated fatty acids, double bonds are usually not adjacent to each other so there is no conjugation and MP decreases.
Geometry of Saturated vs Unsaturated Gats
Stearic acids are straight tails, but oleic acid has a bent tail due to the presence of double bonds.
Increasing # of double bonds lowers the MP, b/c double bond introduces a kink that prevents Van der Waals interactions.
Geometry of cis vs trans Unsaturated Fats
Trans fatty acids form by partial dehydrogenation of unsaturated fatty acids. They can pack more regularly and show higher melting points than cis forms. It occurs in small amounts naturally and stimulate cholesterol synthesis and lead to atherosclerosis.
Triacylglycerols: Fatty Acid Esters of Glycerol
Fats and oils in plants an animals are primarily mixtures of triacylglycerols, which are fatty acids triesters of glycerol.
Triacyglycerols are non-polar, water-insoluble substances that serve as energy reservoirs in animals.
They are the most abundant lipids in animals but do not form part of cellular membranes.
Triacyglycerols (TAG) Structure
Most triacylglycerol contain 2 or 3 different fatty acid residues and are named based on their position on the glycerol molecule, with the fatty acid name ending changing from –ate to –oyl in the ester form.
TAGs are more hydrophobic than free fatty acids – can be stored in lipid droplets without water. TAGs are less dense than water: fats and oils float.
Majority of dietary lipids are TAGs: solid TAGs – fat, liquid TAGs – oil.
Animals have Adipose Tissue to Store TAGs
Adipocytes containing fat globules of triacyclglycerols. Fatty acids in TAGs carry more energy per C than polysaccharides b/c they are more reduced. Fatty acids carry less water along b/c they are nonpolar.
Glucose and glycogen are for short-term quick energy. Fats are for long term (days to months) energy needs.
Structure of Glycerophospholipids
The main lipids in biological membranes consist of glycerol-3-phosphate esterified with fatty acids at C1 and C2, with the phosphoryl group linked to a polar group, X.
These amphiphilic molecules have nonpolar hydrocarbon “tails” and polar phosphoryl-X “heads”, crucial for membrane structure.
Glycerophospholipids play roles in signal transduction, membrane homeostasis, and metabolism, and are acted upon by various enzymes. The simplest glycerophospholipids, phosphatidic acids, contain X = H.
Common glycerophospholipids feature saturated C16 or C18 fatty acids as C1 and unsaturated C16 to C20 fatty acids at C2, with names based on fatty acid residues.
Common Glycerophospholipids Head Groups
Glycerophospholipids are named after their head groups:
Serine = phosphatidylserine
Ethanolamine = phosphatidylethanolamine
Choline = phosphatidylcholine
Glycerol = phosphatidylglycerol
Inositol = phosphatidylinositol
Plasmalogen
Plasmalogens are a type of glycerophospholipid with a unique a, B-unsaturated ether linkage at the C1 position of glycerol.
Common plasmalogen head groups include ethanolamine, choline, and serine.
They may react with O free radicals, thereby preventing free-radical damage to other cell constituents. Highly concentrated in the brain, heart, and immune cells, they act as crucial antioxidants, maintain membrane integrity, and modulate membrane dynamics and signaling.
Phospholipases Hydrolyze Glycerophospholipids
Hydrolysis of glycerophospholipids by phospholipases is a vital step in cell signaling, acting as a “molecular switch” that converts membrane phospholipids into potent, bioactive second messengers.
Phospholipases are activated by various stimuli – such as hormones or environmental stress – and their derived products control key cellular processes including growth, development, defense responses, and intracellular traffic.
Sphingolipids
Sphingolipids are major membrane components. Most sphingolipids are derivatives of the C18 amino alcohol sphingosine (not glycerol) which possesses a trans-alkene.
The N-acyl fatty acid (amide) derivatives of sphingosine are known as ceramides. A polar head gorup is connected to sphingosine by a glycosidic or phosphodiester linkage. The sugar containing glycosphingolipids are found largely in the outer face of plasma membrane.
Structure of Sphingolipids
Sphingomyelins are ceramides bearing either a phosphocholine or a phosphoethanolamine head group. It is abundant in myelin sheath that surrounds some nerve cells in animals.
They typically make up 10-20 mol % of plasma membrane lipids.
Myelinated Nerve Fibers
The myelin sheath insulating nerve cell axons is rich in sphingomyelins.
Gangliosides: Sialic acid-containing glycosphingolipids
It is abundant in the brain. Carbohydrate groups are added to the head group. Some act as receptors for glycoprotein hormones; cell-cell recognition.
Defects in processing enzymes cause Tay Sachs neurodegenerative disorder.
Steroids & Cholesterol
Steroids are derivatives of cyclopentanoperhydrophenanthrene consisting of 4 fused, nonplanar rings.
Cholesterol is classified as a sterol due to its C3-OH group. Polar OH group gives weak amphiphilic character, and fused ring system provide rigidity. Most abundant steroid in animals.
Constitues 30-40 mol% of animal plasma membrane lipds (plants have related sterols in their membranes). Cholesterol and related sterols modulate membrane fluidity in eukaryotes. Most bacteria lack sterols.
Cholesterol Esters
Cholesterol can be esterified with long-chain fatty acids, forming cholesteryl esters like cholesteryl stearate. Cholesteryl esters are more hydrophobic than cholesterol. It is used for transporting cholesterol in lipoprotein particles: e.g. LDL, HDL.
Sterol hormones are synthesized from cholesterol.
Forms of Vitamin D: Vitamin D2 and D3
Vitamin D is synthesized in the skin from cholesterol derivative 7-dehydrocholesterol upon UV radiation from sunlight.
Ubiquinone
Ubiquinone (also known as coenzyme Q (CoQ)) is reversibly reduced and oxidized in the mitochondrial membrane; mammalian ubiquinone consists of 10 isoprenoid units.
Eicosanoids
Eicosanoids include prostaglandins, prostacyclin's, thromboxane's, leukotrienes, and lipoxins, are all C20 compounds. They regulate various physiological processes.
Arachidonic acid is the key eicosanoid precursor, stored as phospholipid esters and released by phospholipase A2.
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
NSAIDs inhibit prostaglandin synthesis to relieve pain and fever. Salicylate is the first NSAID, obtained from the bark of the white willow tree. Ibuprofen is a widely used NSAID.
Aggregates of Single-Tailed Lipids
In aqueous solutions, amphiphilic molecules like soaps and detergents form micelles. The hydrophobic effect drives the close packing of lipid tails, as water molecules max their entropy.
Bilayer Formation by Phospholipids
The hydrocarbon tails of glycerophospholipids and sphingolipids give these amphiphiles a rectangular cross-section, leading to large, disklike micelles (extended bimolecular leaflets).
Liposome
Membrane permeability:
Gases (e.g. O2) and small hydrophobic molecule (e.g. steroid hormones) can cross the plasma membrane by themselves
Large (e.g. protein) or small polar molecules (e.g. glucose) can’t cross the plasma membrane and require special transporters
Diffusion in Phospholipid Bilayer
Proteins and lipids move laterally freely but rarely flip-flop from layer to layer (usually require help of flipase enzymes). Biological membranes has a “fluid mosaic” structure.
Transverse diffusion is flip flop and is very slow. Lateral diffusion is rapid.
Phase Transition in Lipid Bilayer
There is a proper fluidity for a membrane that need to be maintained at all times to ensure proper biological functions (e.g. receptor binding, enzyme activity, endocytosis, etc.)
Transition temp (aka melting temp Tm) - temp at which phase transition occurs
T < Tm: liquid ordered state (aka gel) - membrane is too rigid
T > Tm: liquid disordered state (aka liquid-crystalline) - membrane is too fluid.
Phase transition for a membrane has a sigmoidal shape. Proper membrane fluidity is around Tm.
Factors Influence Tm
Longer tail or less unsaturated= higher Tm
Shorter tail or more unsaturated = lower Tm
Organism can adapt to changes in environmental temp by diff mechanisms:
Bacteria change composition of membrane lipids
e.g. if T drop composition will change to more unsaturated fatty acids
Eukaryotes have sterol in their membranes (cholesterol in animals)
Membrane composition is altered to maintain fluidity at all temps.
Cholesterol & Phase Transition
Cholesterol helps maintain proper fluidity: If T < Tm cholesterol prevents tight packaging of FA tails – prevents transition to semi-solid state. If T > Tm – cholesterol will prevent free rotation of FA tails – prevent transition to fluid-like state.
As a result, phase transition in the presence of cholesterol is wider. The central point (Tm) does not change, but the range of temp at which membrane has perfect fluidity is broader.
Topology of Membrane-Associated Proteins
Membrane proteins catalyze rxns, mediate nutrient/waste flow, and help relay info about the extracellular environment to various intracellular components by interacting with the lipid bilayer.
Membrane proteins can be integral (transmembrane or lipid-anchored) or peripheral.
Types of Membrane Anchored Proteins
N-myristoylation (C4) at N-terminal Gly (amide)
S-palmitoylation (C16) at Cys-residue near C-terminus (thioester)
S-prenylation at Cys residue of C-terminus (thioether)
Glycosylphosphatidylinositol (GPI)-anchored proteins (C-terminus)
Structure of Prenylated Proteins
The common prenylations site is the C-terminal tetrapeptide C – X – X –Y, where C is Cys and X is an aliphatic AA. Farnesylation occurs if Y is Ala, Met, or Ser, and geranylgeranylation if Y is Leu.
After prenylation, the X – X – Y tripeptide is excised, and the terminal carboxyl group is methylated.
Structure of GPI-Anchored Proteins
GPI anchor is added to proteins after translation. Treatment of GPI anchored protein with phospholipase releases them from the membrane.
Membrane Proteins
Peripheral proteins can be removed with change in salt [ ] , chelating agents or change in pH
Integral Anchored proteins can be removed with enzymes that cleave that specific bond anchoring the protein in the membrane.
Integral Transmembrane Proteins can only be removed with detergents (non-biological amphipathic molecules)
Structure of Integral Membrane Proteins
Integral membrane proteins have diff distribution of hydrophobic AA. Hydrophobic AA are on the outside of the protein structure where they interact with hydrocarbon chains of the membrane lipids.