Nucleophilic substitution of haloalkanes (hydrolysis)
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
What is a nucleophile
An electron pair donor
What are common nucleophiles that react with haloalkanes
Hydroxide ions
Water molecules
Ammonia molecules
What happens when the nucleophile reacts with a haloalkane
The nucleophile replaces the halogen in a substitution reaction
Draw the mechanism of nucleophilic substitution
Products are a new alcohol and a halide ion as it keeps electrons
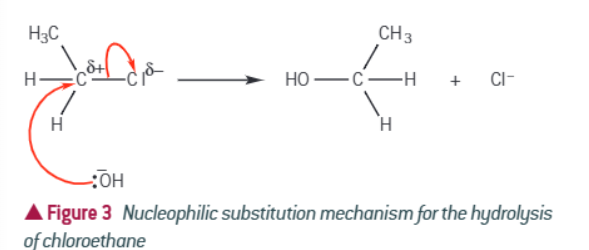
What does the rate of hydrolysis of a haloalkane depend on
The strength of the carbon-halogen bond in the haloalkane, the bond enthalpy
What is the order of the bond enthalpy of carbon-halogen bonds
C-F
C-Cl
C-Br
C-I
Why does C-I have the lowest bond enthalpy
Iodine has a larger atomic radius and more electron shielding, so the electrostatic attraction between the nucleus and the bonding electrons is weaker, giving a lower C–I bond enthalpy.