BIOC 4331 Lecture 21
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
What two major factors contribute to catalytic rate enhancement from binding energy?
Stabilization of the transition state and entropy reduction.

What do catalytic groups often form during enzyme reactions?
A covalent intermediate with the substrate or perform group transfer to or from a substrate.


What small groups can be transferred in enzyme catalysis?
Electrons (redox) or protons (acid–base catalysis).


What is specific acid–base catalysis?
Proton transfer between water and the intermediate that is faster than breakdown to reactants; additional H+ donors/acceptors do NOT increase rate.


What is general acid–base catalysis?
Proton transfer mediated by groups other than water (e.g., amino acid side chains).


When does general acid–base catalysis increase reaction rate?
When proton transfer from water is slower than intermediate breakdown, so additional H+ donors/acceptors increase the rate.


Why might enzymes require general acid–base catalysis?
The active site may exclude H2O.

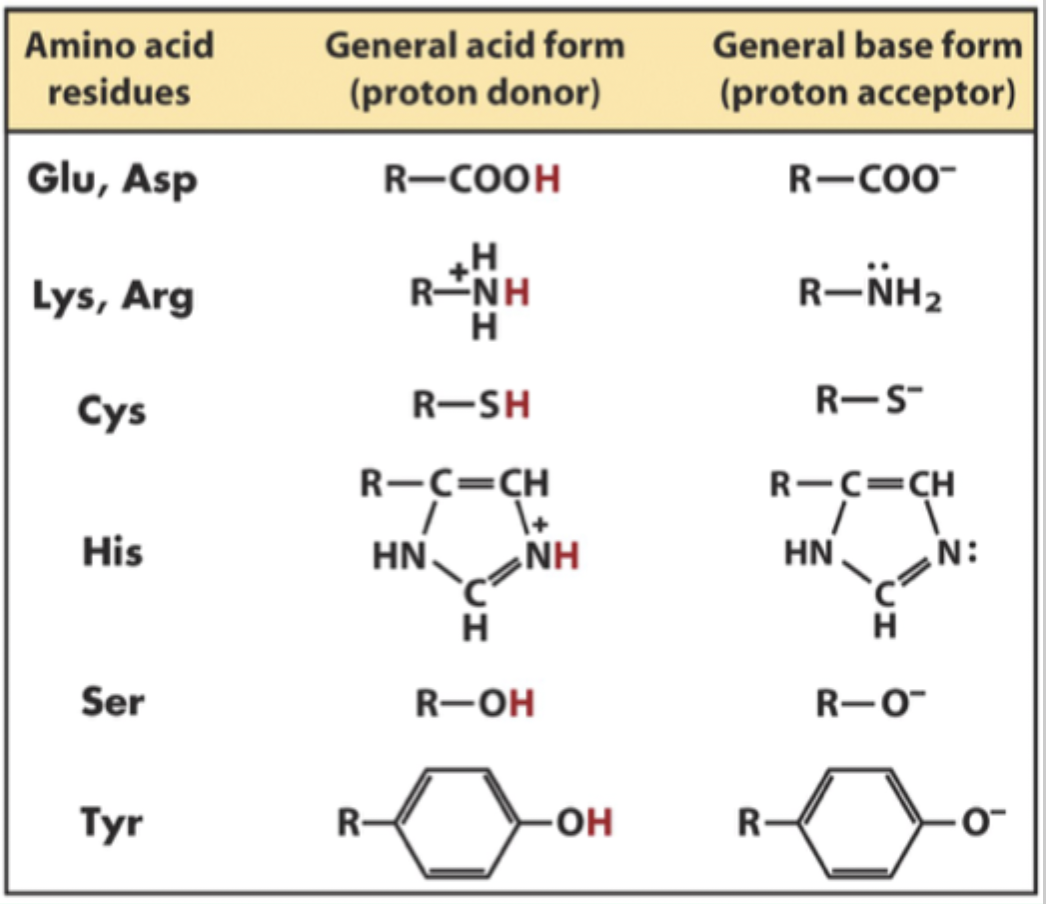
Which amino acids commonly act as general acids/bases?
Glu and Asp
Lys and Arg
Cys
His
Ser
Tyr
What does covalent catalysis do to a reaction pathway?
Alters the pathway of the reaction.
When will a covalent pathway dominate?
If the new pathway has a lower activation energy.
What enzyme components can act as nucleophiles in covalent catalysis?
Amino acid side chains and functional groups of some cofactors.
List three ways metal ions promote catalysis.
Orient substrates
Stabilize charged intermediates
Mediate oxidation–reduction reactions.
What is charge shielding?
Metal ions electrostatically shield negative charges to reduce repulsion during nucleophilic attack.
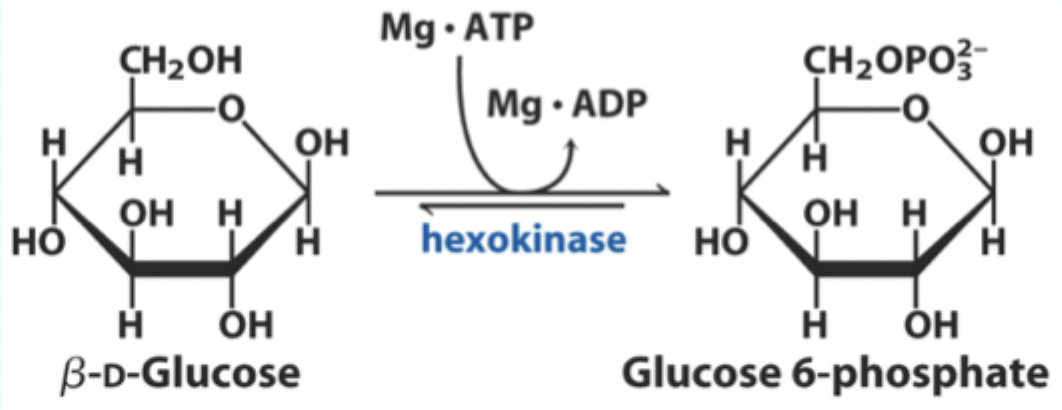
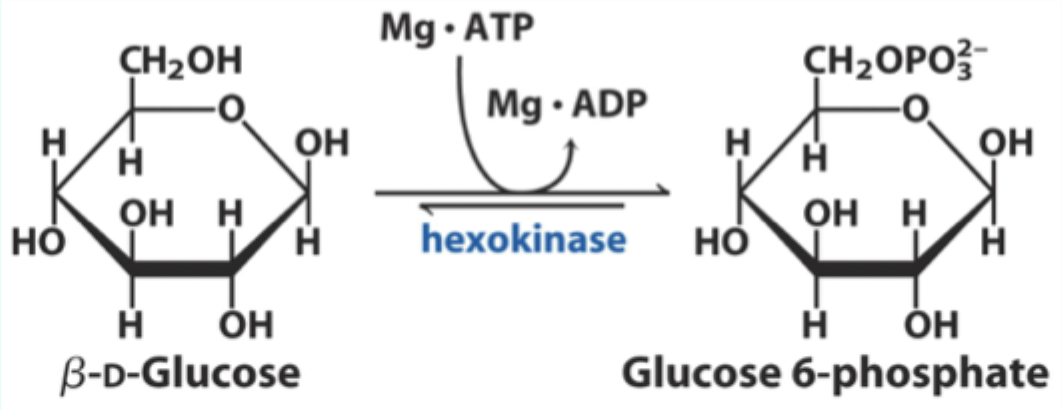
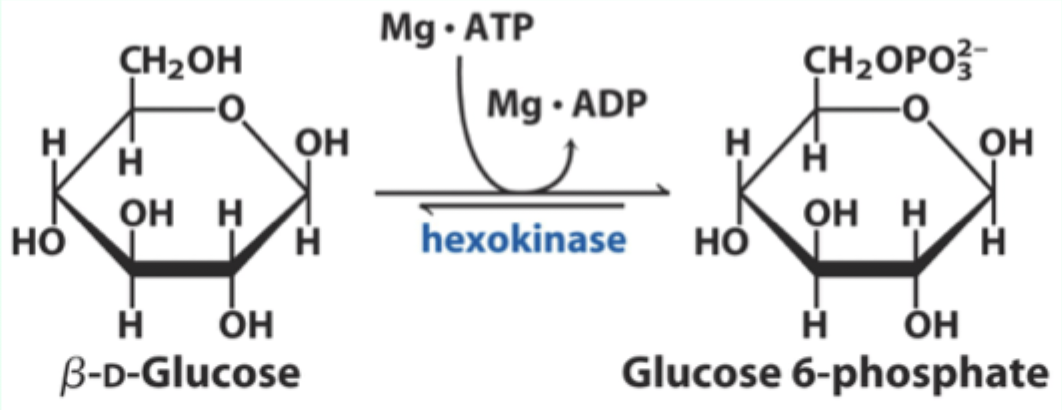
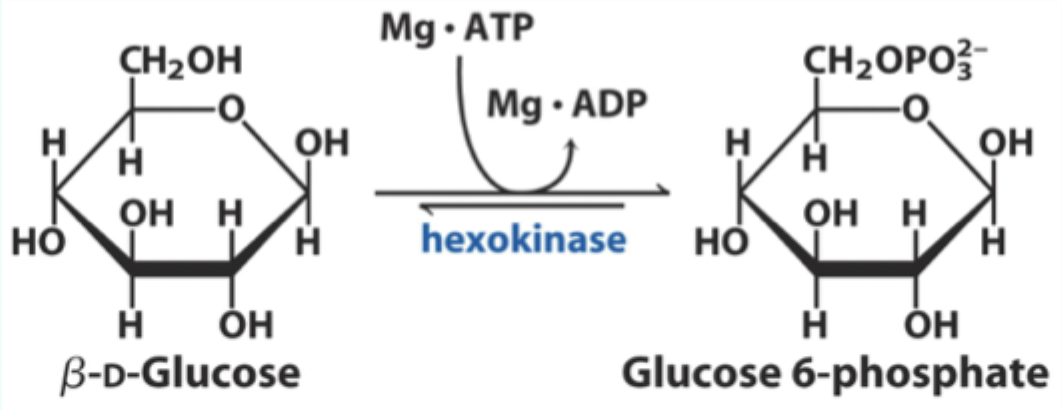
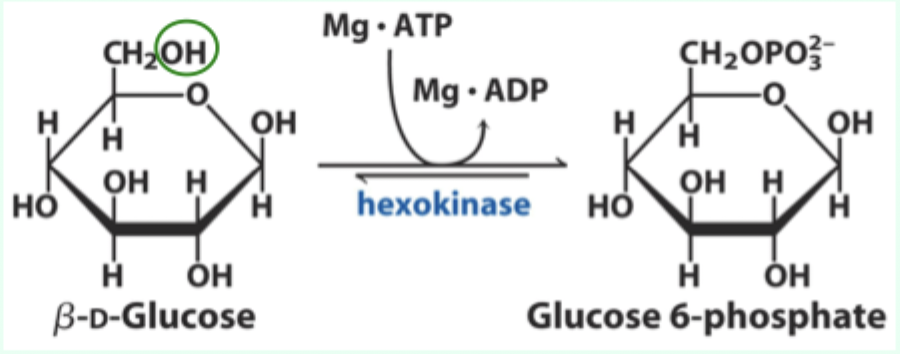
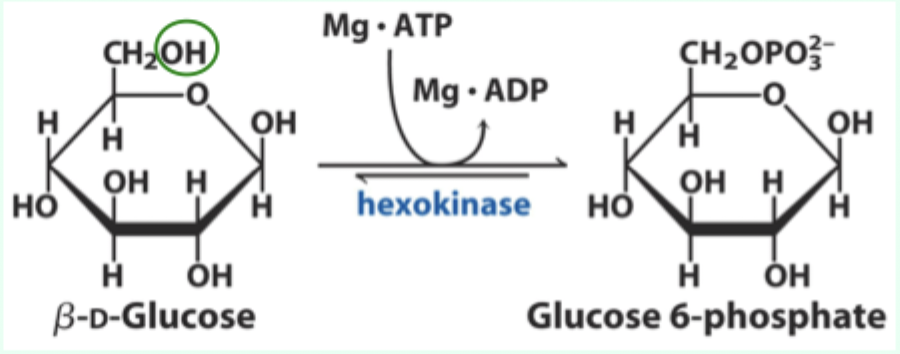
What enzyme catalyzes the conversion of D-glucose to glucose-6-phosphate?
Hexokinase.
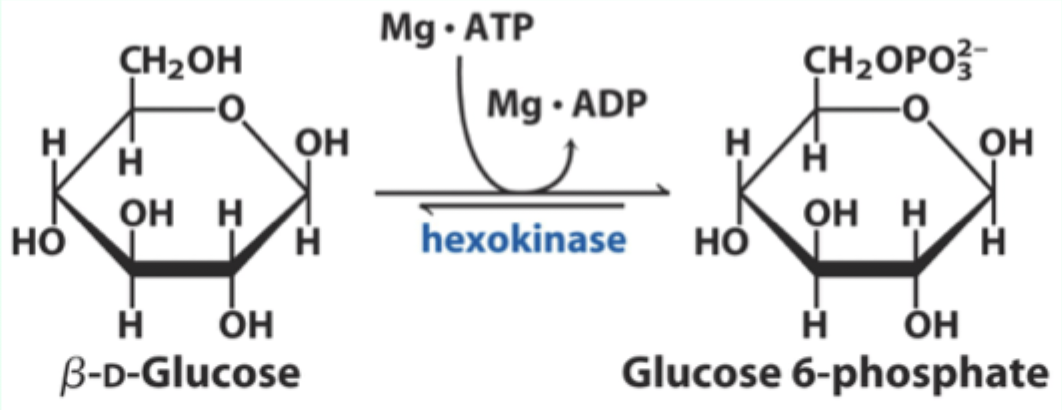
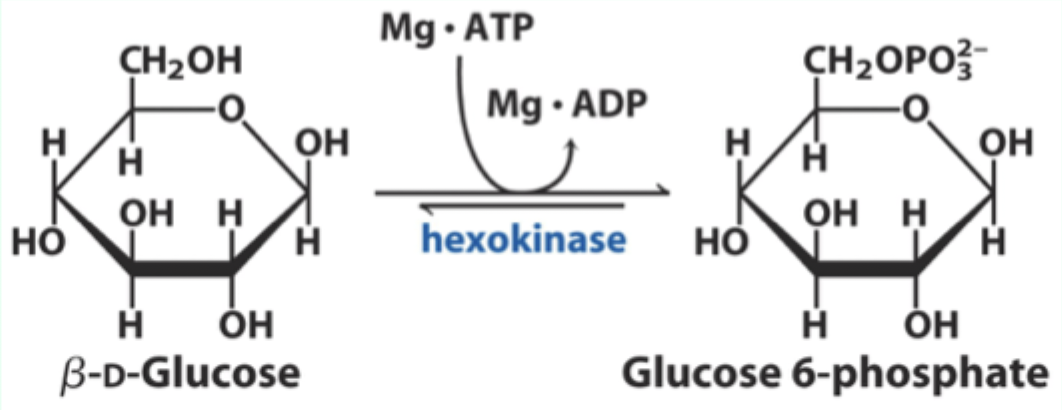
What type of enzyme is hexokinase?
Bisubstrate enzyme.
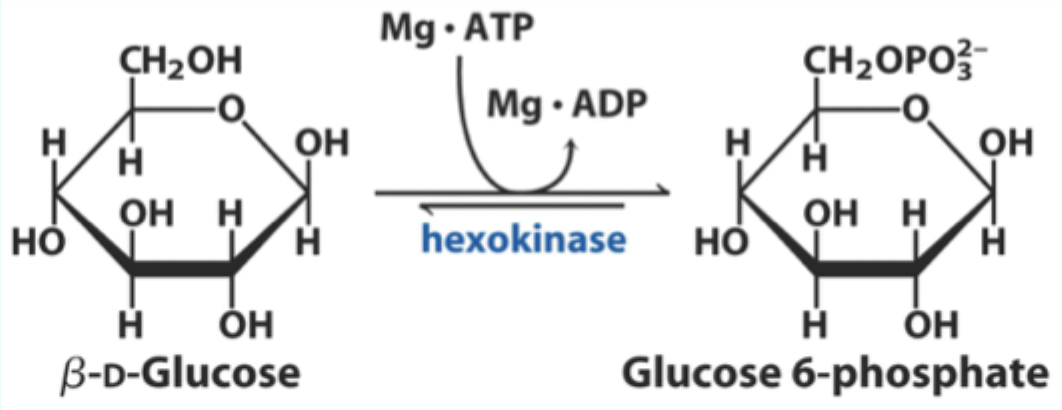
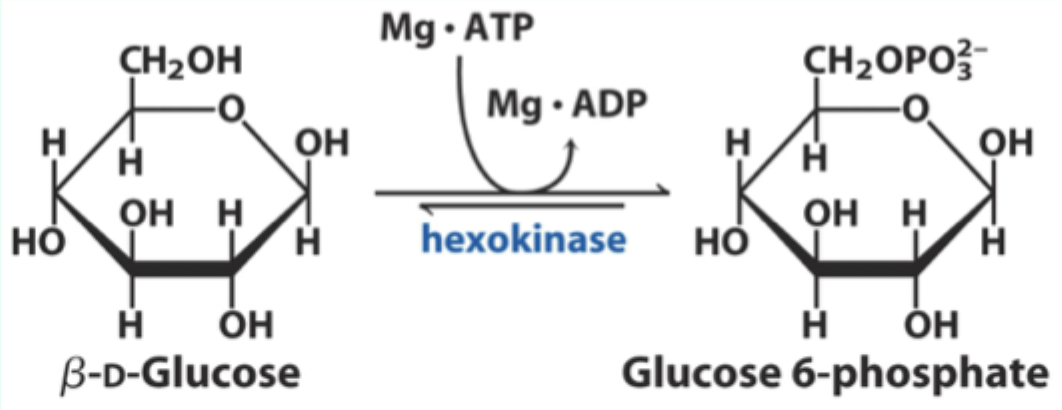
What two substrates does hexokinase use?
D-glucose and Mg·ATP.
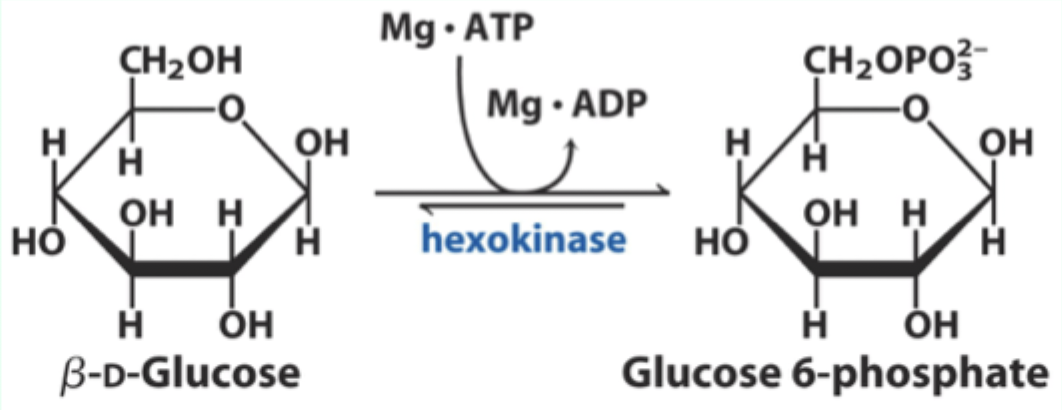
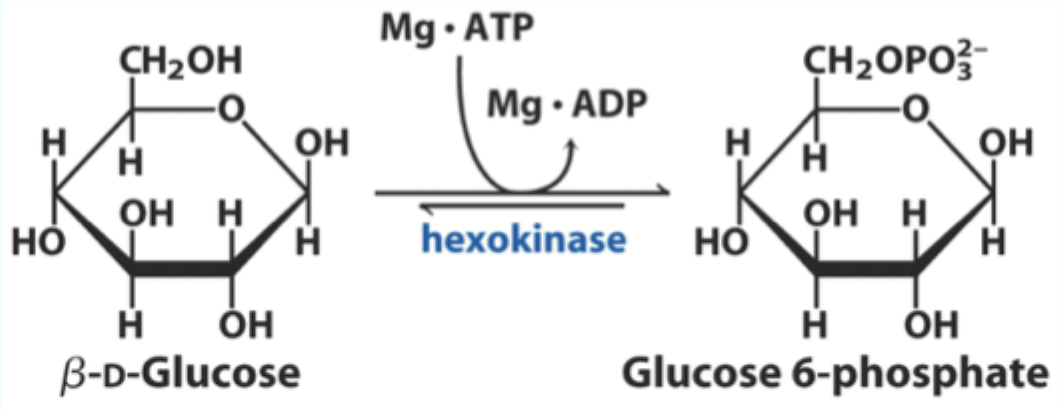
What product does hexokinase form from glucose?
Glucose-6-phosphate.
What happens to hexokinase when glucose is not bound?
It is in an inactive conformation.
What triggers the conformational change in hexokinase?
Binding energy from glucose and MgATP2−.
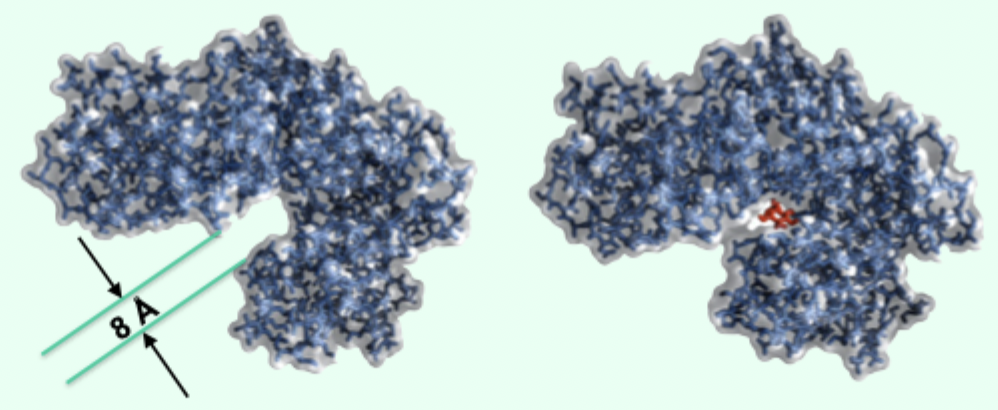
What structural change occurs in hexokinase upon substrate binding?
8 Å movement that closes the active site.
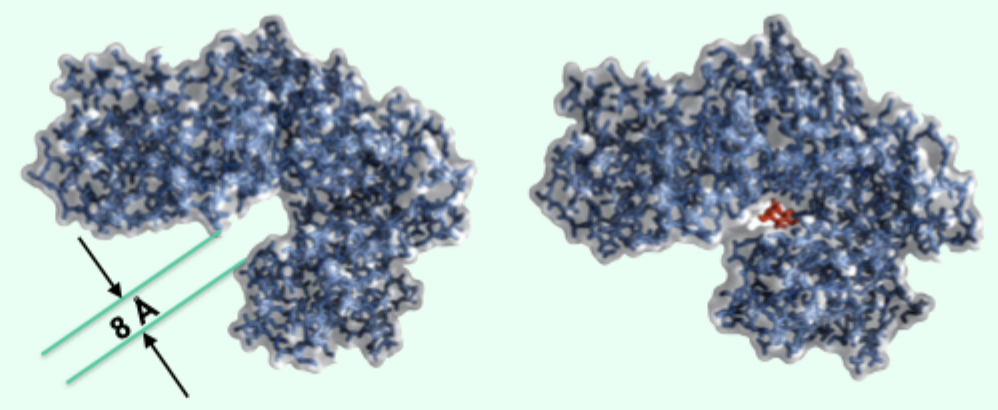
What does induced fit accomplish in hexokinase?
Blocks H2O access and brings MgATP2− and glucose closer.
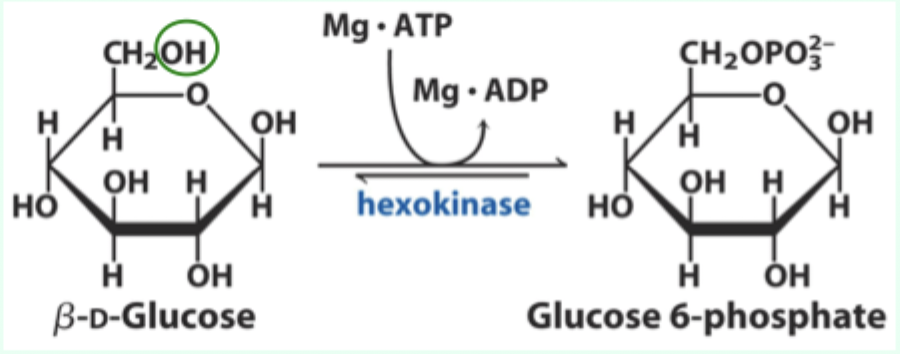
How much does hexokinase favor glucose over water?
~106-fold.
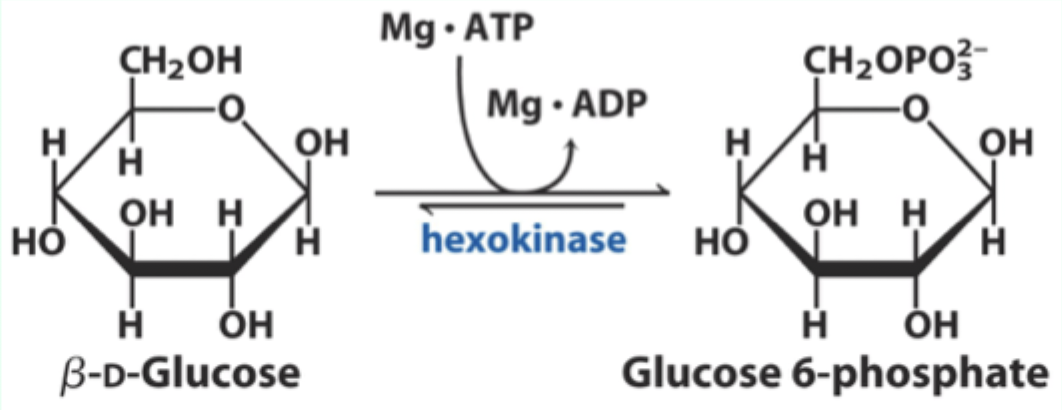
Besides induced fit, what catalytic strategy does hexokinase use?
General acid–base catalysis.
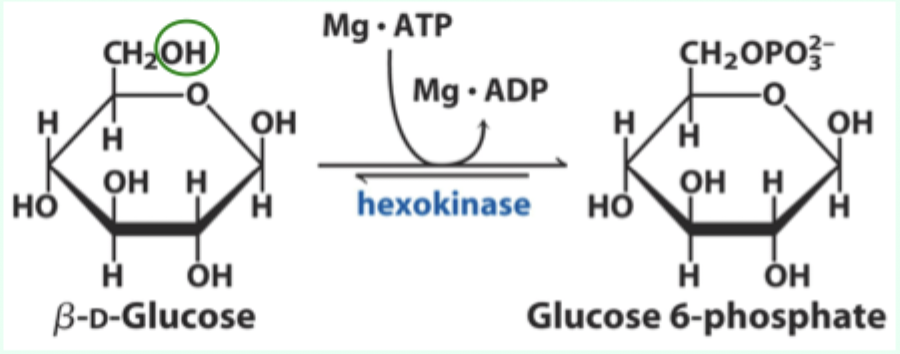
What additional catalytic principle does hexokinase demonstrate?
Transition state stabilization.
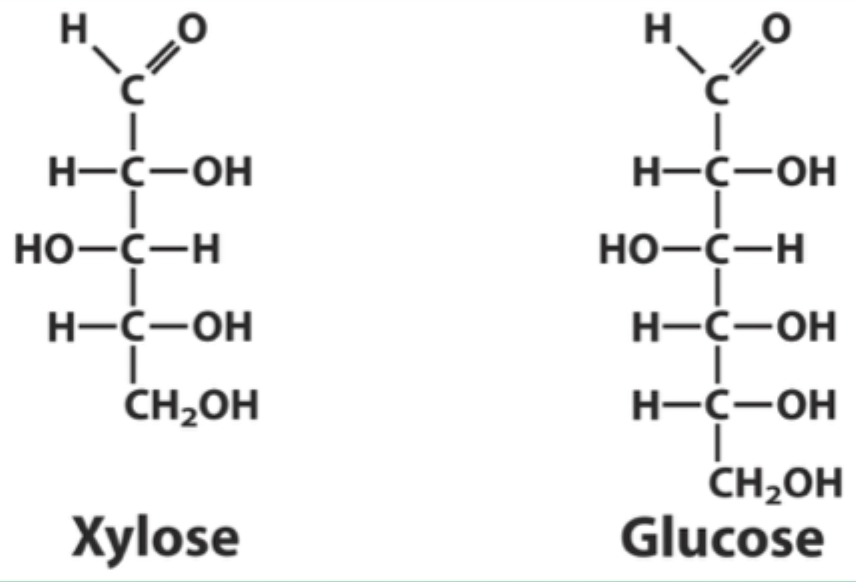
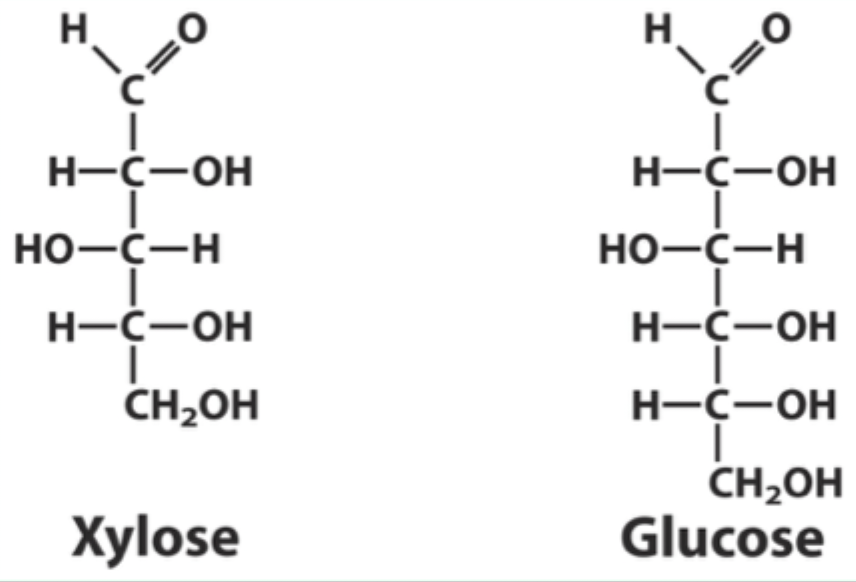
Why can xylose bind hexokinase but not be phosphorylated?
It binds in a position where it cannot be phosphorylated.
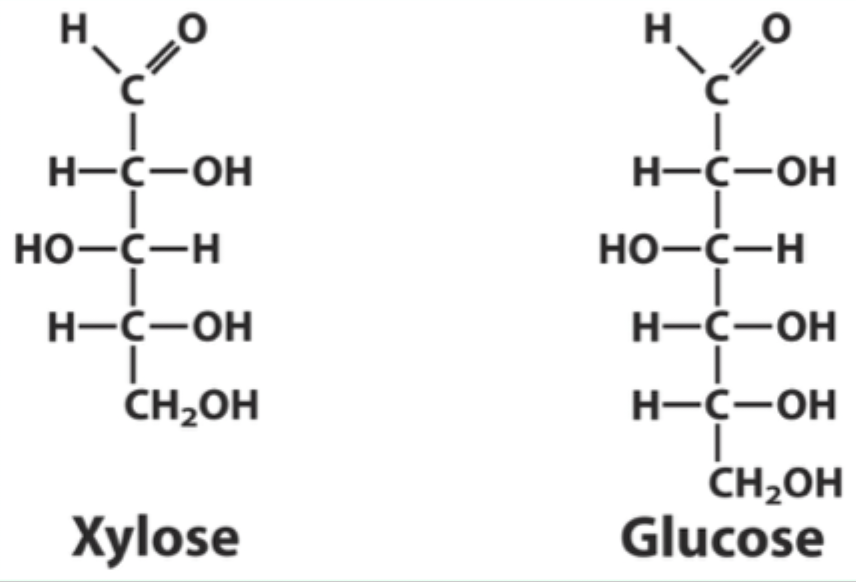
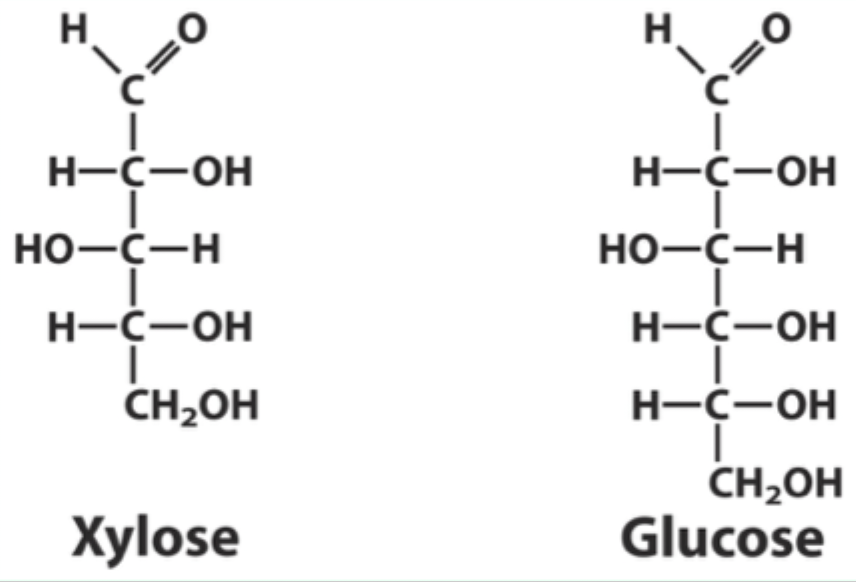
What type of ring does xylose form?
A 5-membered furanose ring.
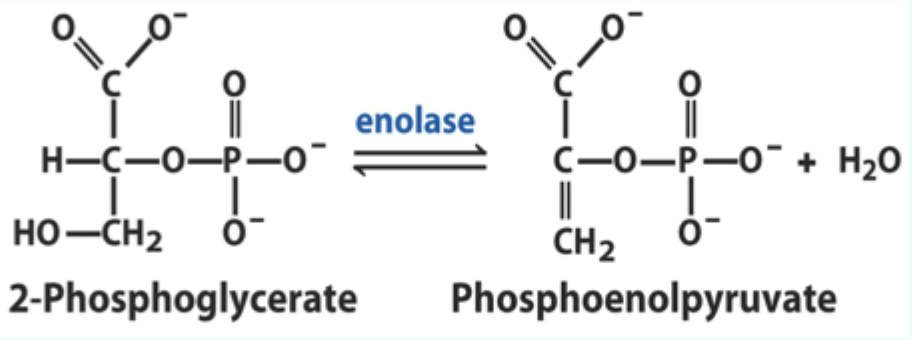
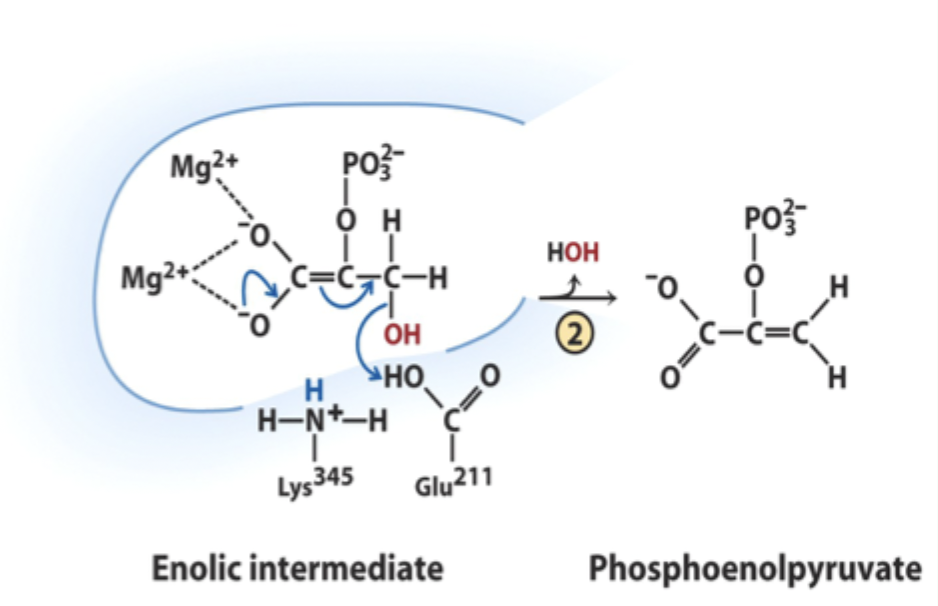
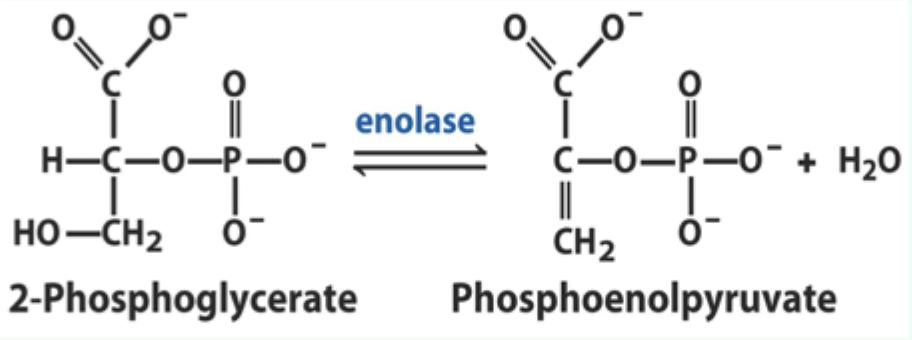
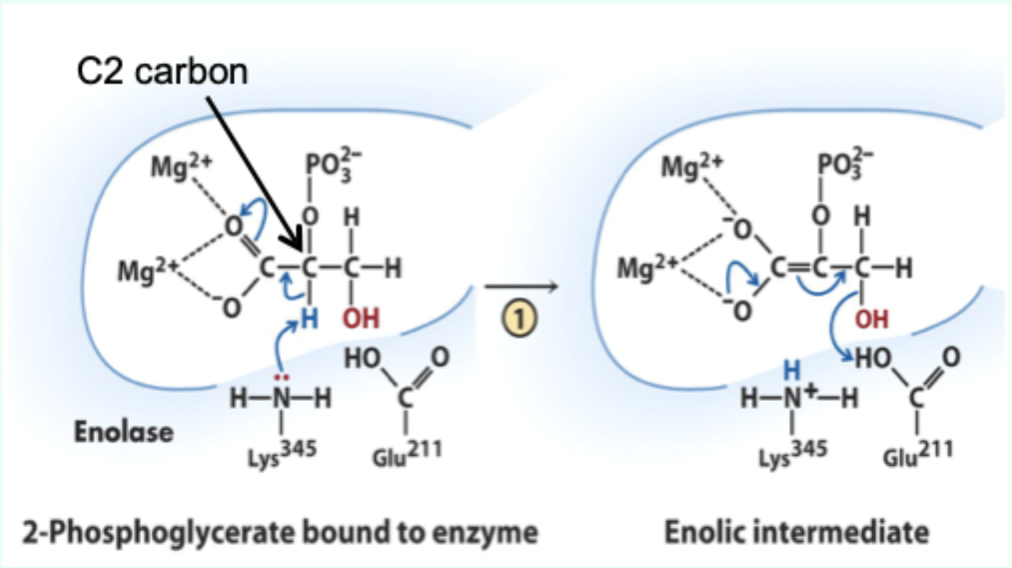
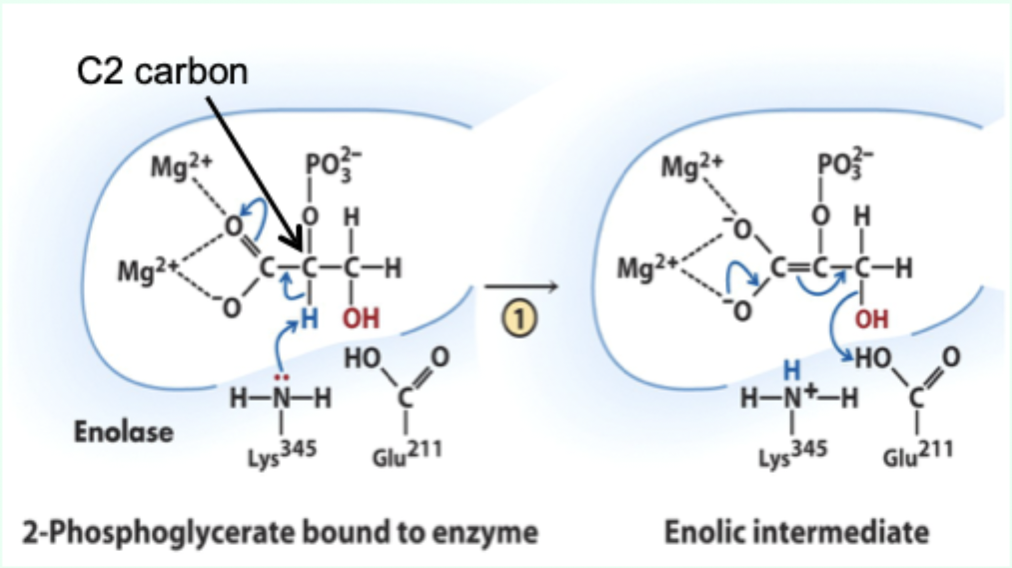
What reaction does enolase catalyze?
Reversible dehydration of 2-phosphoglycerate to phosphoenolpyruvate.
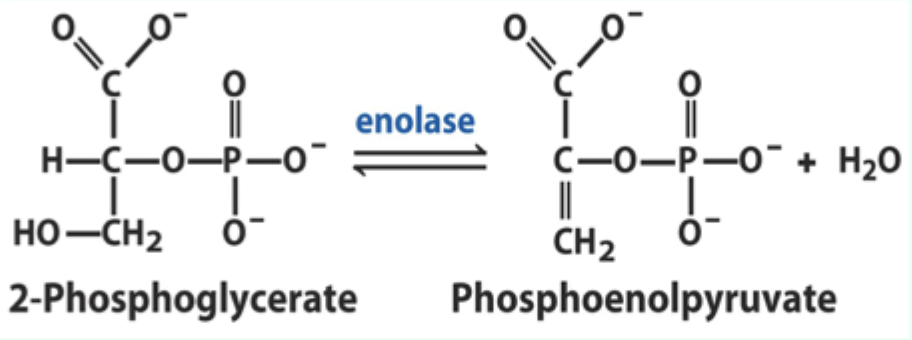
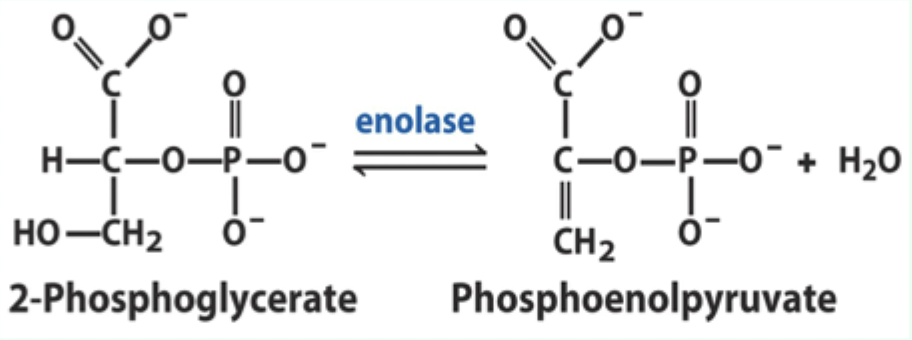
Enolase is an example of what three catalytic strategies?
Metal ion catalysis
General acid–base catalysis
Transition state stabilization
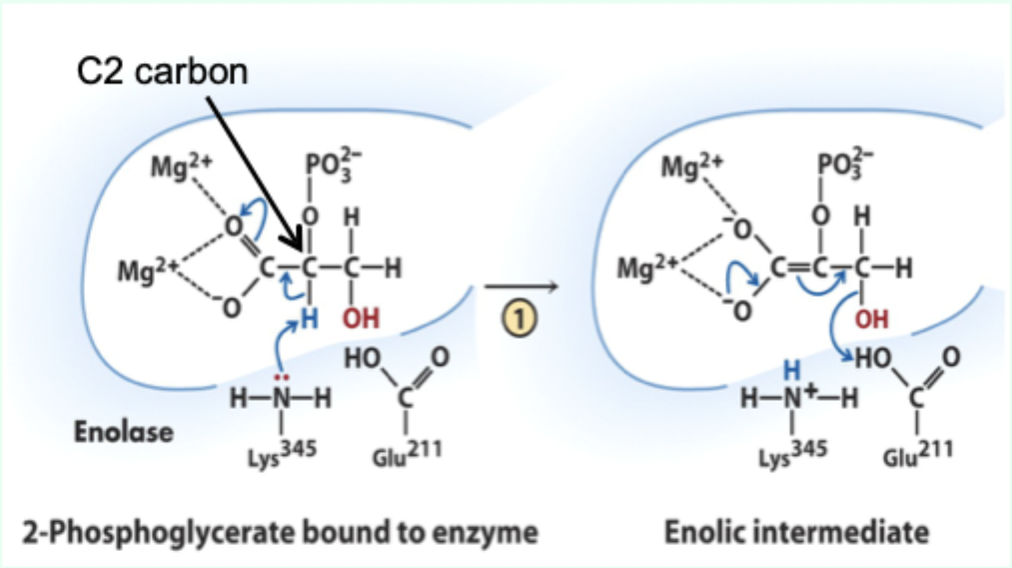
What does Lys345 act as in enolase?
General base catalyst
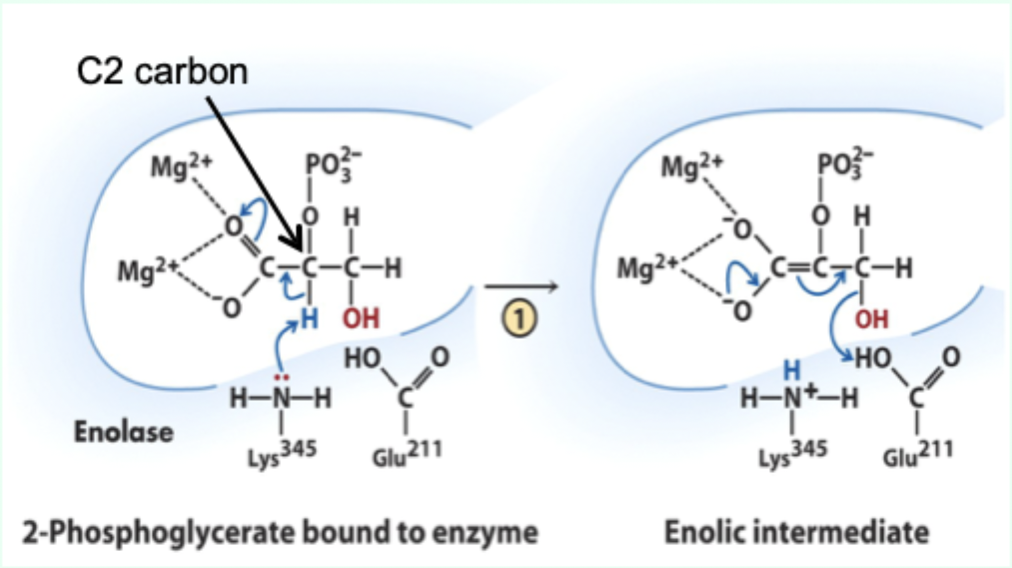
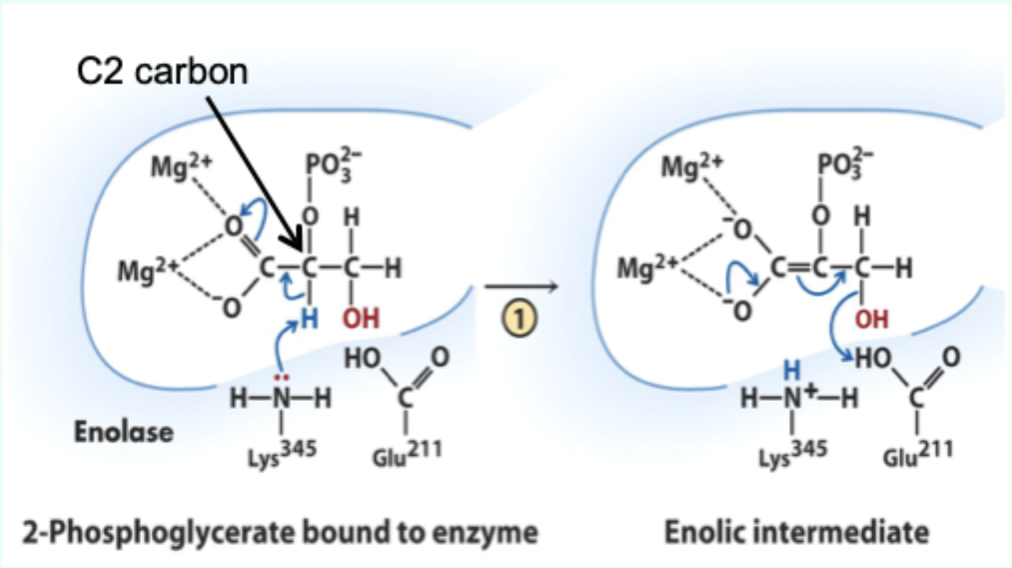
What proton does Lys345 remove?
Proton from C2 of 2-phosphoglycerate.
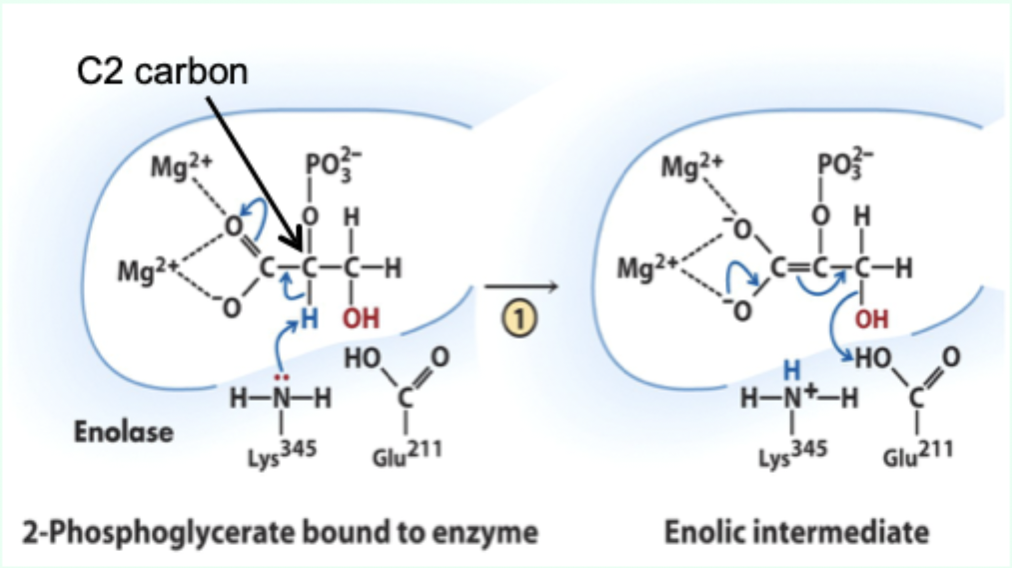
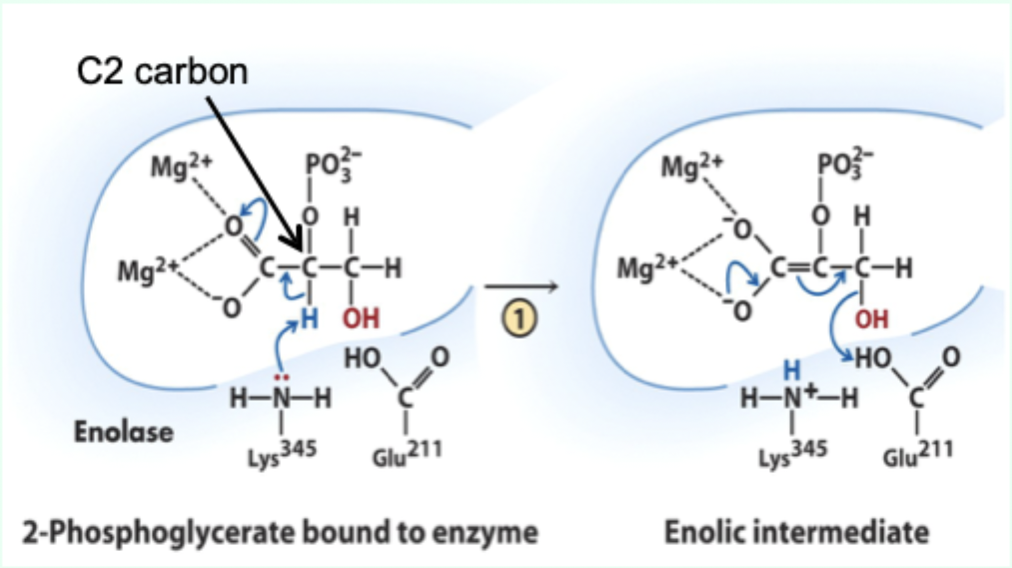
Why is the C2 proton removable in the enzyme but not in solution?
Two Mg2+ ions lower its pKa (make it more acidic).
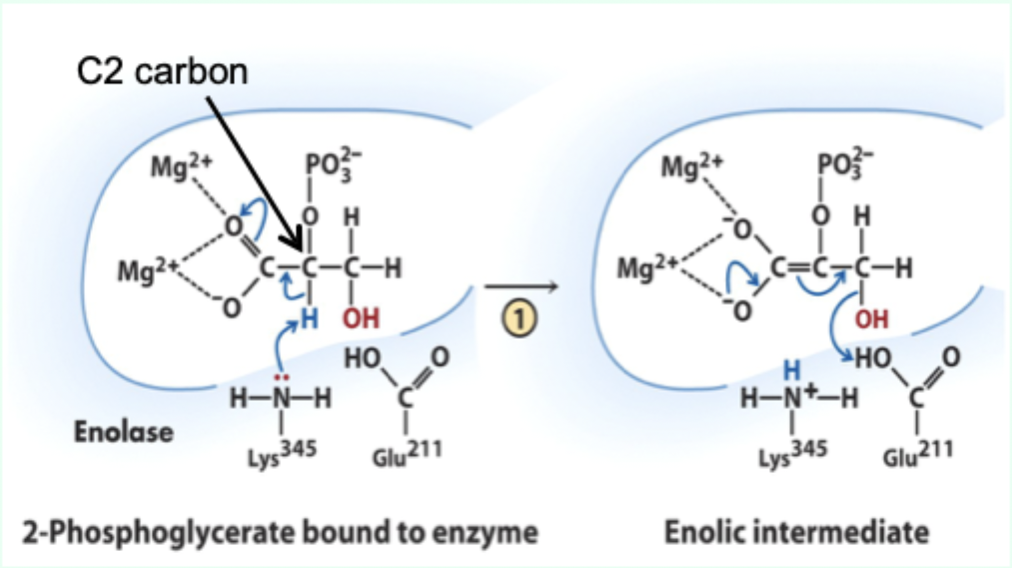
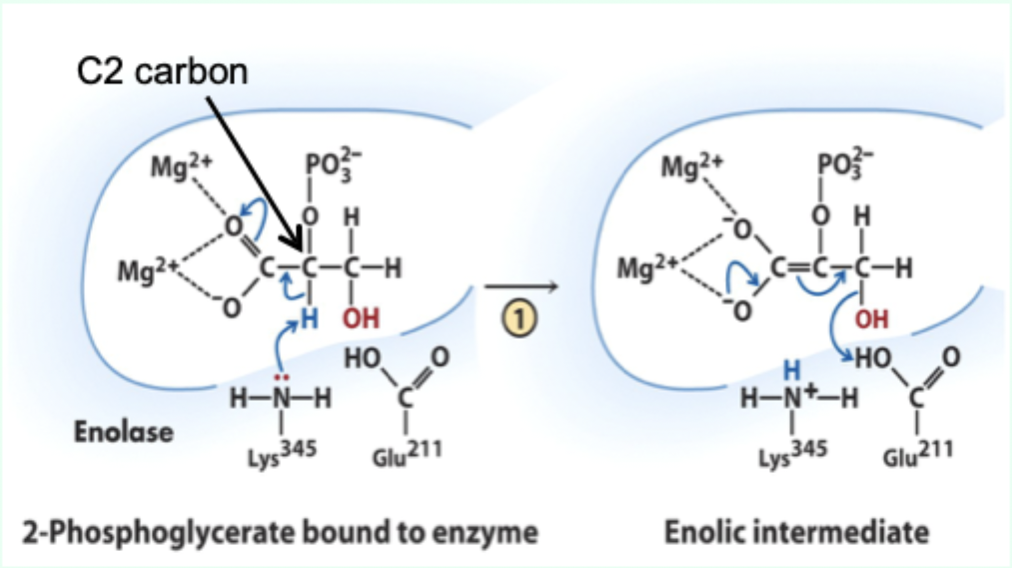
What role do Mg2+ ions play in enolase?
Stabilize negative charges and promote formation of enolic intermediate.
Which residue acts as general acid in step 2 of enolase?
Glu211.
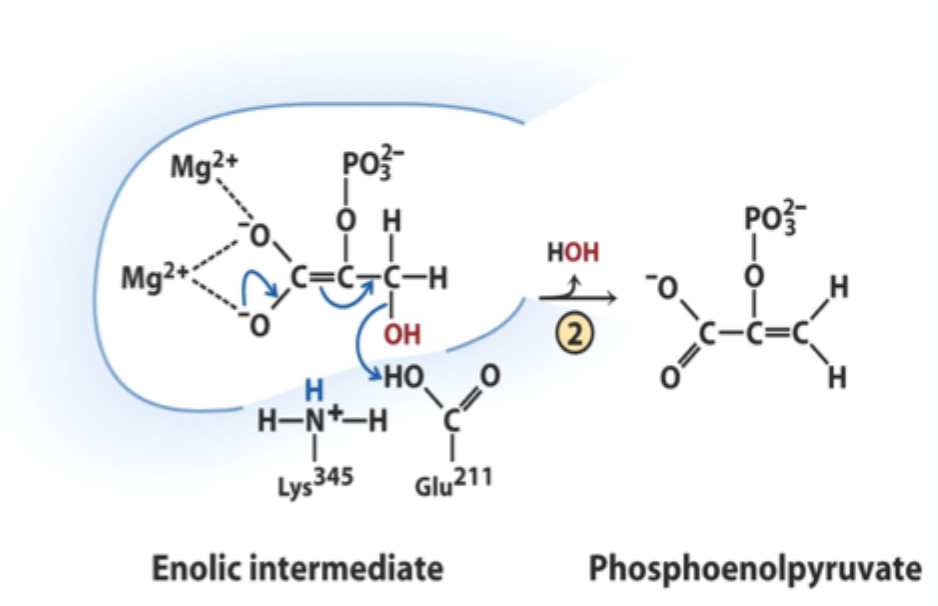
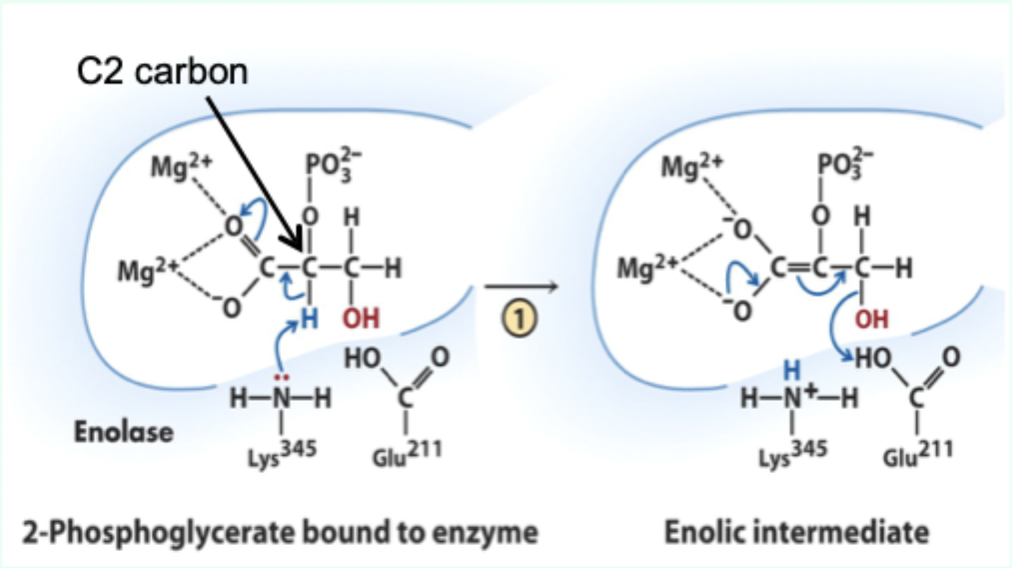
What does Glu211 protonate?
The hydroxyl leaving group to produce water.
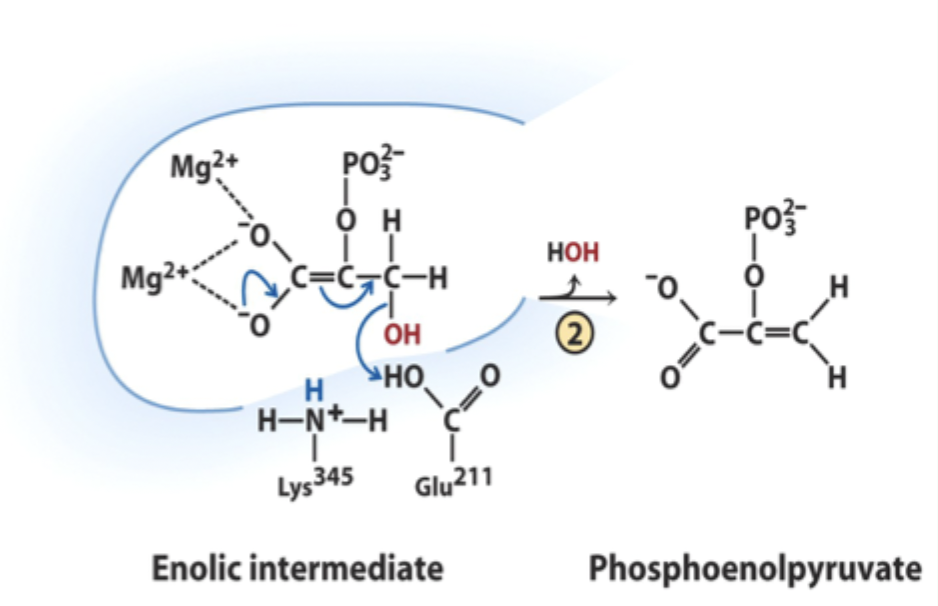
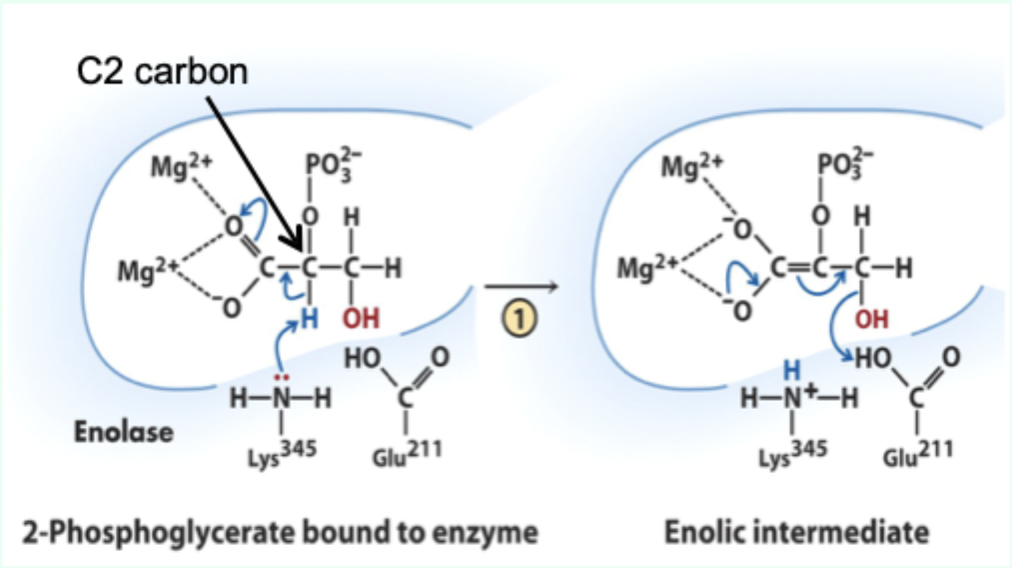
What intermediate forms in enolase catalysis?
Enolic intermediate.