Organic Chemistry Unit 3
1/76
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
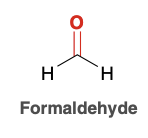
Formaldehyde
Common aldehyde
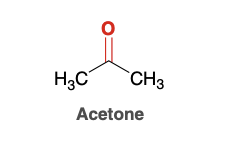
Acetone
Acetone
Common Ketone
Nomenclature of Aldehydes
When naming the parent, the suffix “-al” indicates the presence of an aldehyde group (In an alkane the ending “e” changes)
When numbering the parent chain of an aldehyde, the aldehydic carbon is assigned number 1, despite the presence of alkyl substituents, π bonds, or hydroxyl groups (indicate chirality with R/Z)
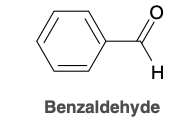
A cyclic compound containing an aldehyde group immediately adjacent to the ring is named as a carbaldehyde
Benzaldehyde
Nomenclature of Ketones
When naming the parent, the suffix “-one” indicates the presence of a ketone group (in an alkane, the “e” changes to “one”
The carbon number connected to the ketone can be placed before suffix or before the parent
Acetophenone
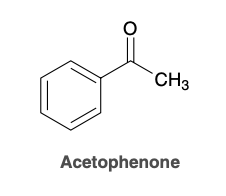
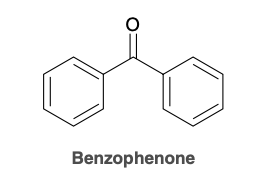
Benzophenone
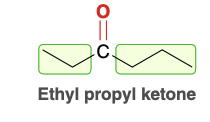
Ethyl Propyl ketone
what is of higher significance in naming, ketone or aldehyde
Aldehyde
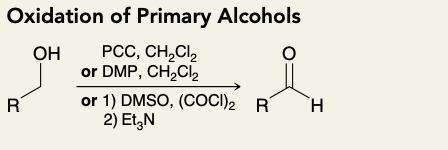
Oxidation of Primary Alcohols (into aldehhyde)
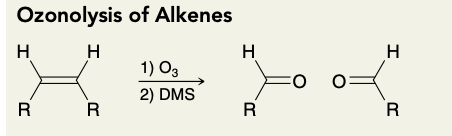
Ozonolysis of Alkenes(into aldehyde)
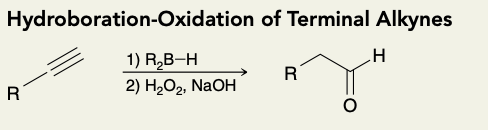
Hydroboration- Oxidation of Terminal Alkynes
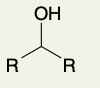
Oxidation of Secondary Alcohols (into ketone)
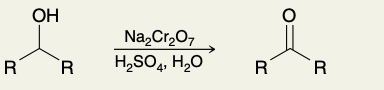
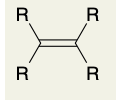
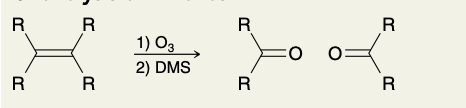
Ozonolysis of Alkenes (into ketone)
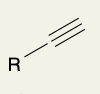
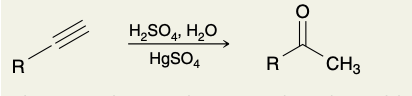
Acid- Catalyzed Hydration of Terminal Alkynes
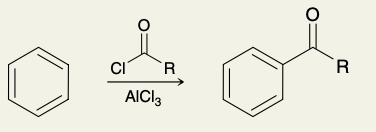
Friedel-Ceafts Acylation (into ketone)
Reactivity of Ketones vs aldehydes
Aldehydes are generally more reactive than ketones toward nucleophilic attack
A ketones two alkyl groups contributes to more steric interacts during the attack compared to an aldehydes one alkyl group (steric effects)
Since alkyl groups are electron donating, a ketone having two electron donating alkyl groups (one more than an aldehyde) stabilize the partial positive charge of electrophilic carbon, making it less reactive.
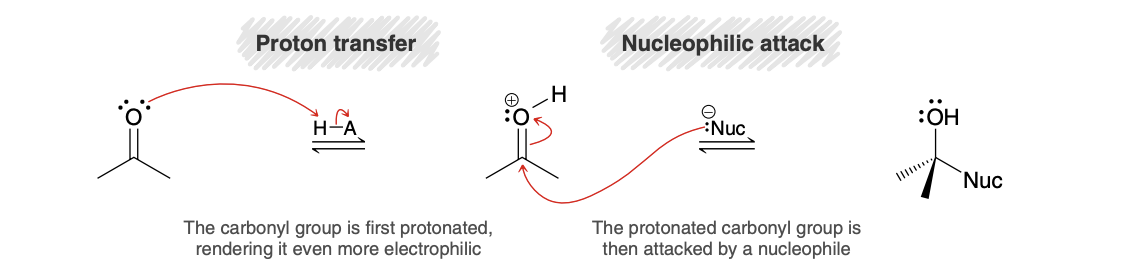
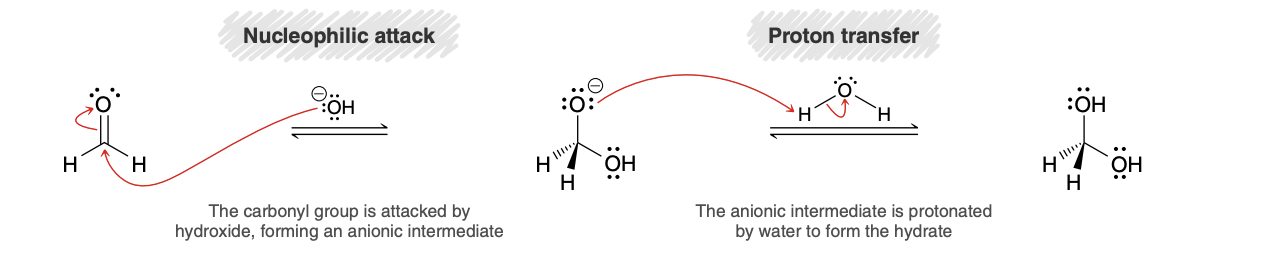
Nucleophilic addition in basic conditions (product/mechanism)
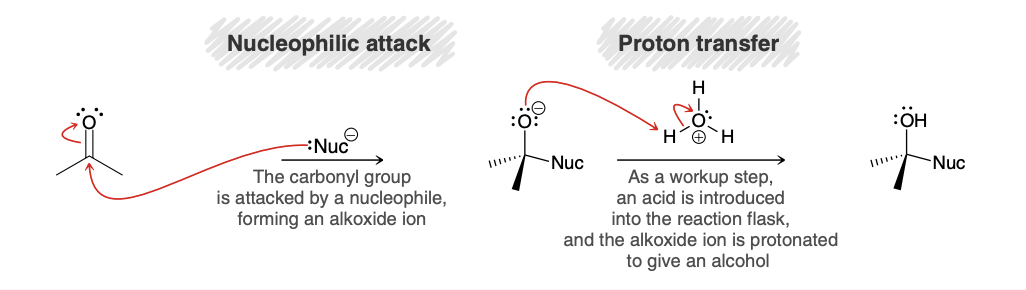
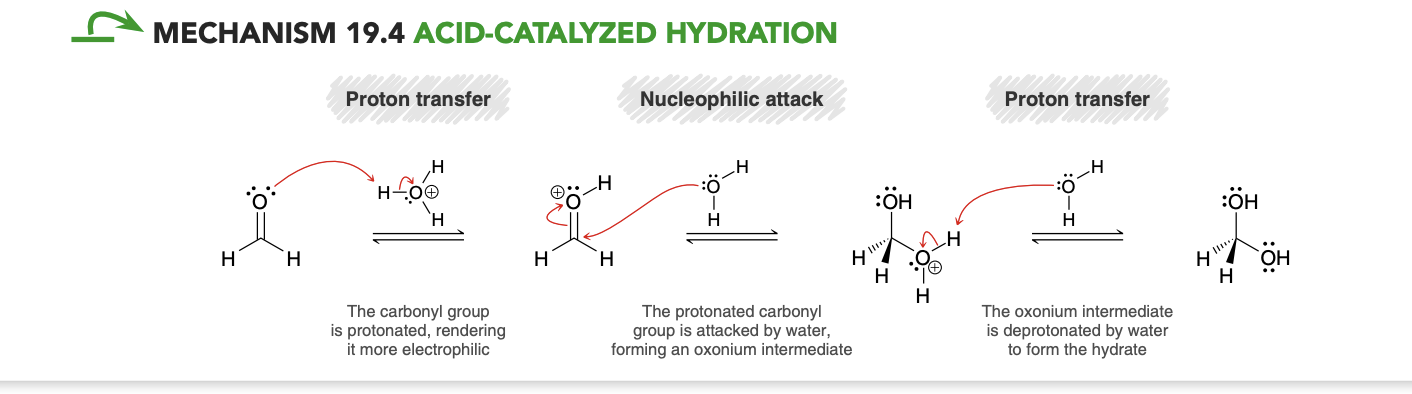
Nucleophilic addition in acidic conditions (product/mechanism)
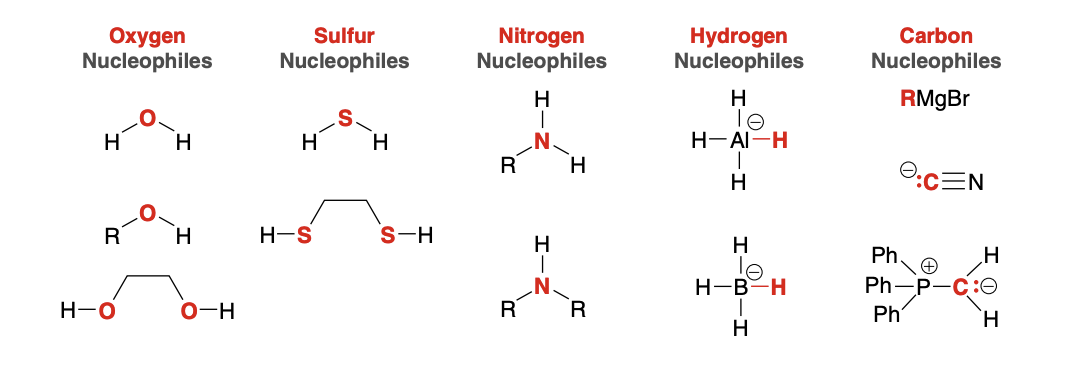
Possible Nucleophiles
Hydrate
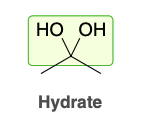
Hydrate formation (ketone)
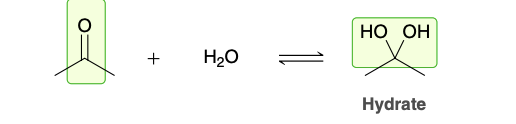
Position of Equlibrum during hydrate formation
The position of equilibrium generally favors the carbonyl group rather than the hydrate, except in the case of very simple aldehydes, such as formaldehyde
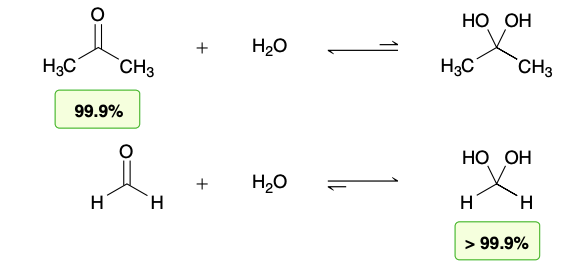
Base catalyzed Hydration (formaldehyde)
Acid Catalyzed hydration(formaldehyde)
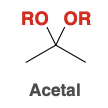
Acetal
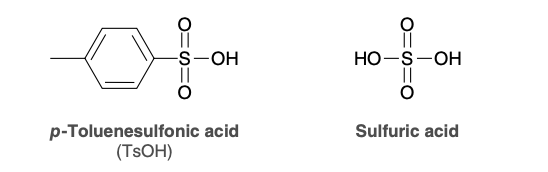
Common acid catalysts
TsOH and H2SO4
Acetal Formation Reagents
Aldehyde/Ketone
ROH
Acid Catalyst
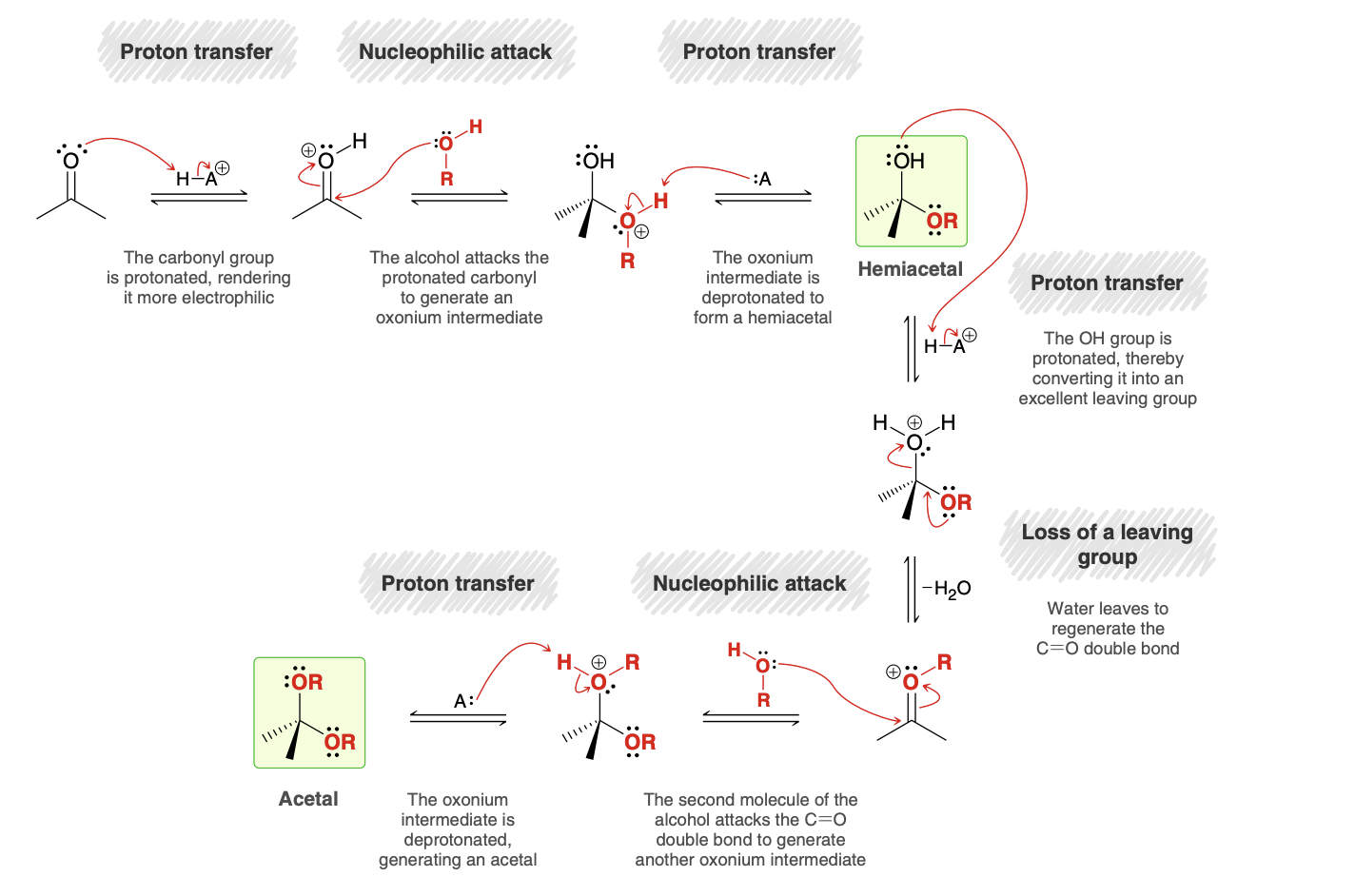
Acetal Formation mechanism
Notes: The identity of the acid, HA+, is most likely a protonated alcohol, which received its extra proton from the acid catalyst.
The oxonium ion is deprotonated by a weak base (A), which is likely to be a molecule of alcohol present in solution.
Acid is not consumed in the process
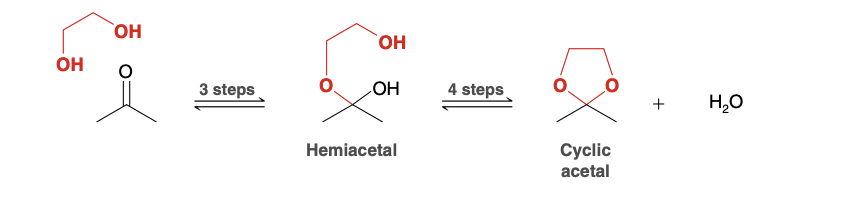
Cyclic Acetal formation
Acetals as a protecting group
Can protect ketones from reacting (i.e. in reactions involving carboxylic acid)
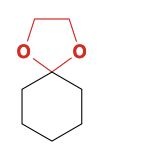
Reagents for forming protecting group
Reagents for removing a protecting group
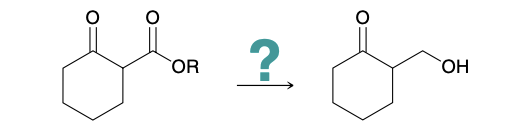
Protecting group example problem
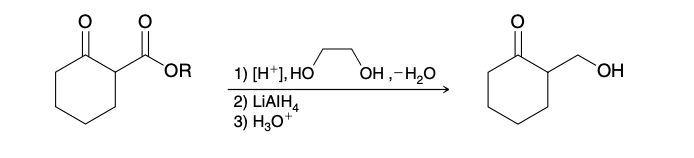
Hemiacetal
Very difficult to isolate and is usually an intermediate
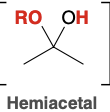
Stable hemiacetal formation
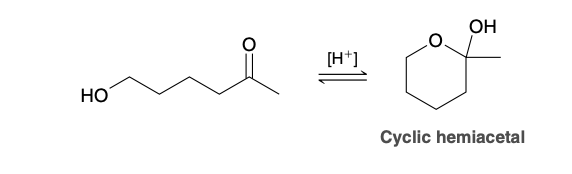
Imine
Forms when a primary amine reacts with an aldehyde/ketone
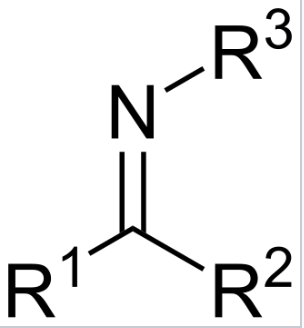
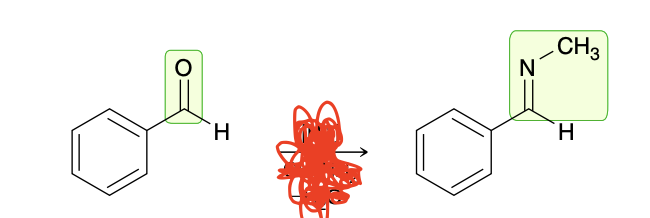
aldehyde to imine
Ketone/Aldehyde
CH3NH2
-H20
Acid catalyzed
Imine Formation Mechanism
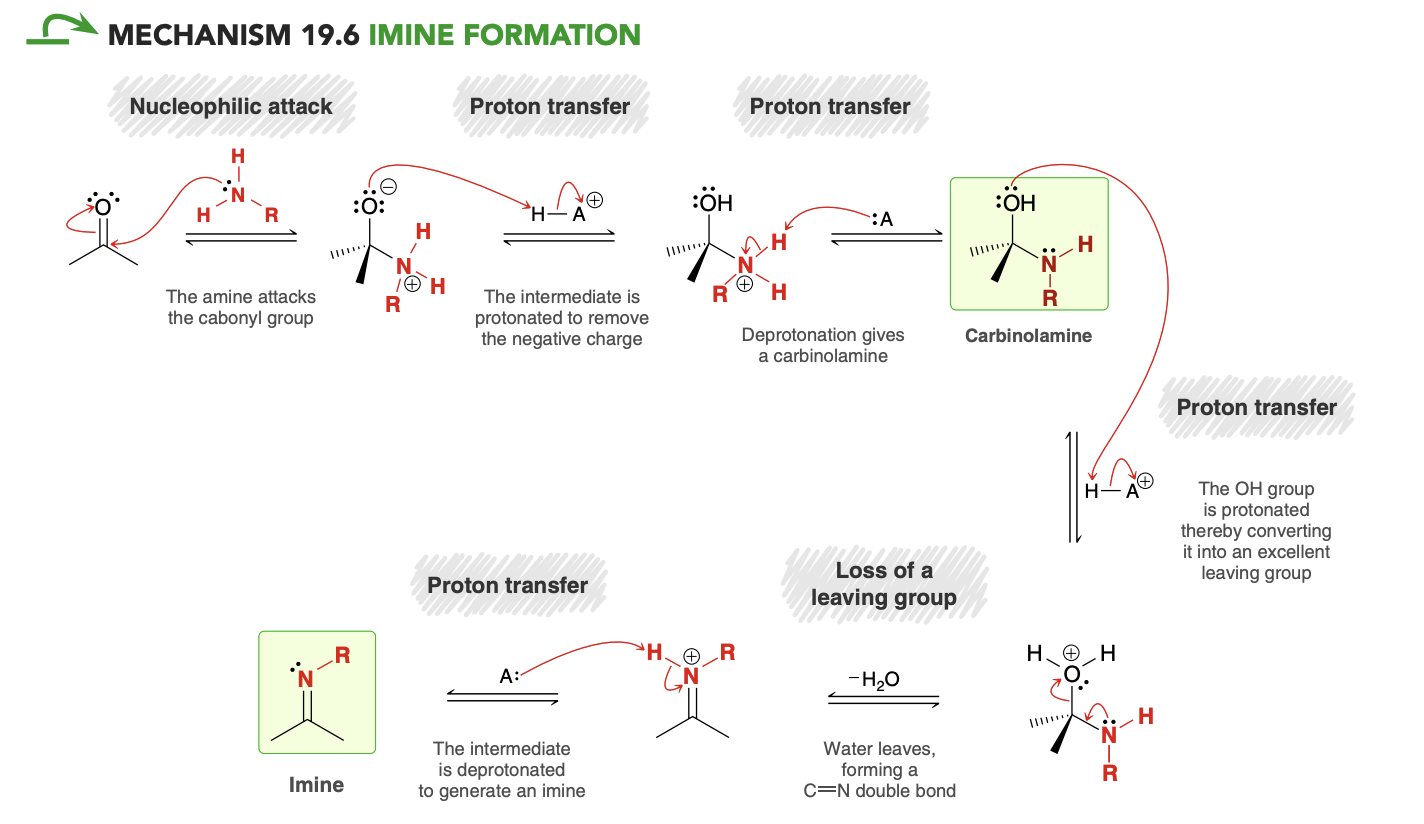
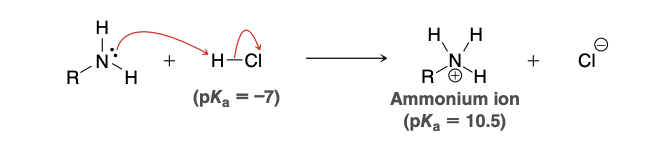
In imine formation, why doesn’t the ketone get protinated
in the presence of an amine, any strong acid catalyst will transfer its proton to the amine, giving an ammonium ion
This process is effectively irreversible due to the vast pKa difference. The acidic species in solution will be ammonium ions rather than HCl, and under these conditions protonation of the ketone is highly unlikely, since protonated ketones are extremely acidic (pKa ≈ −7).
Relates to why ideal pH for imine formation is 4-5
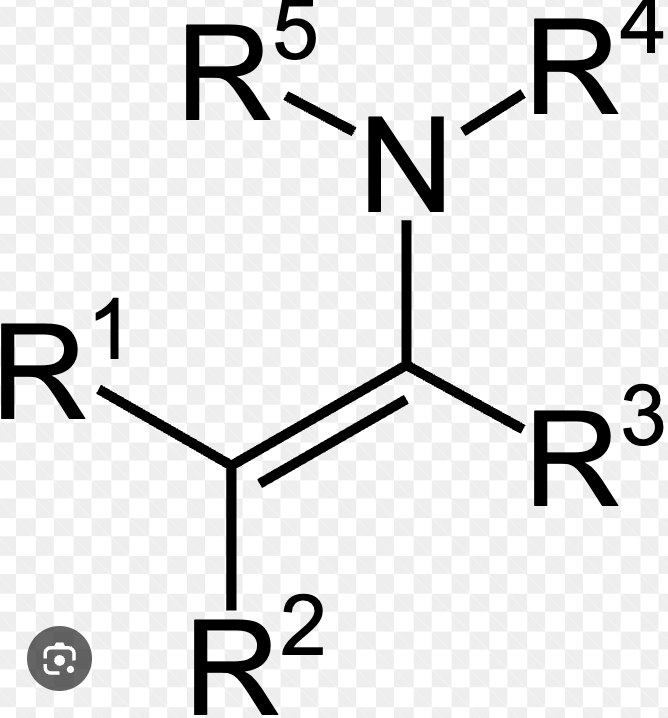
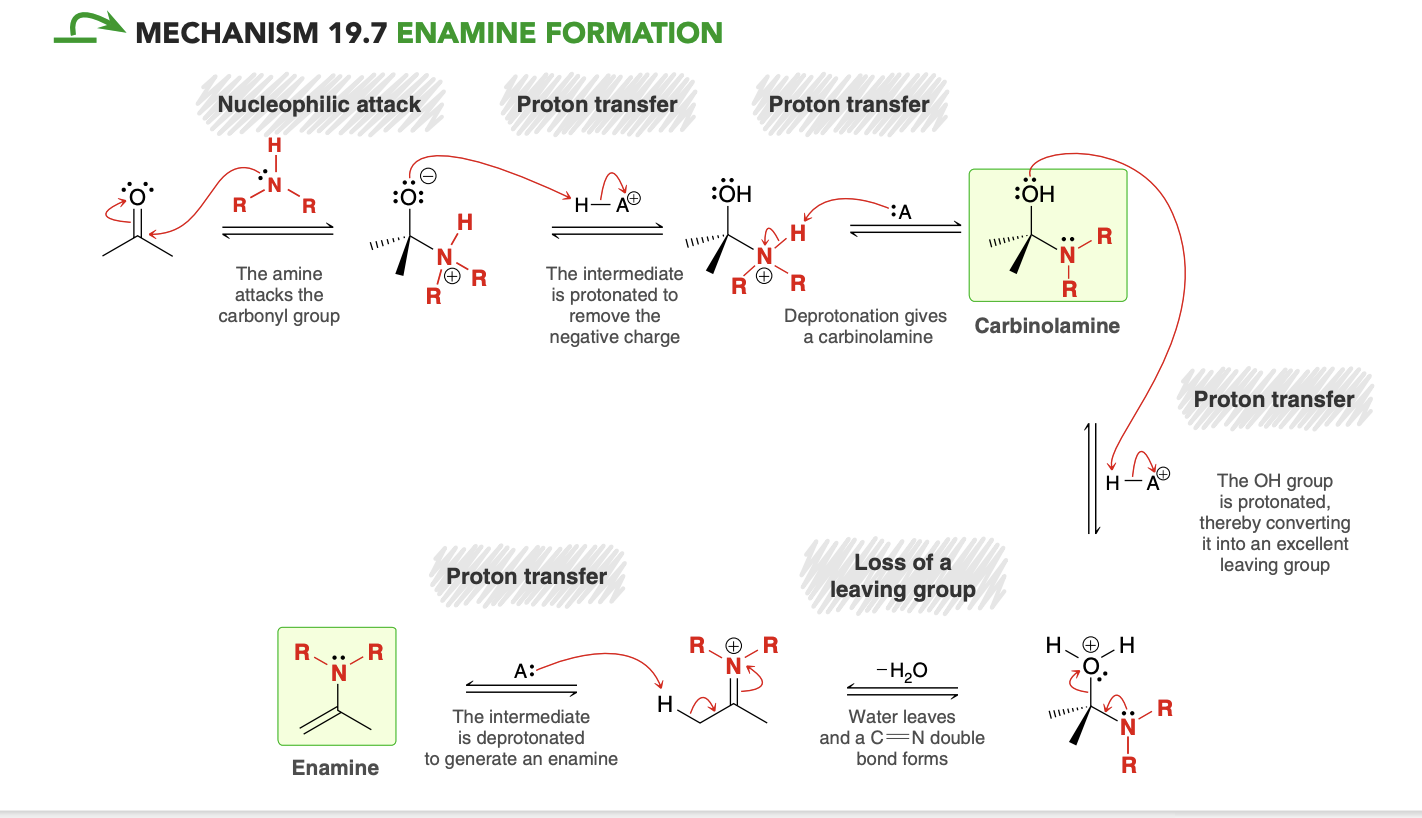
Enamine
Forms when an aldehyde/ketone reacts with a secondary amine in acidic conditions
Enamine Formation Reagnets
Aldehydes/Ketones
R2NH
-H2O
Enamine Formation
Identical to mechanism for imine formation except for the last step
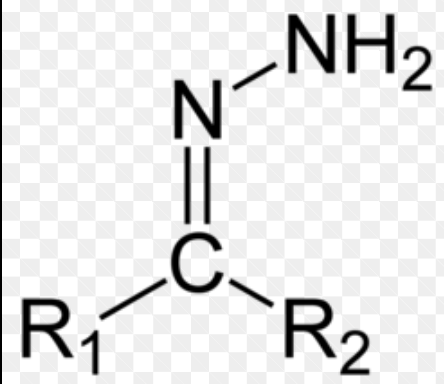
Hydrazone
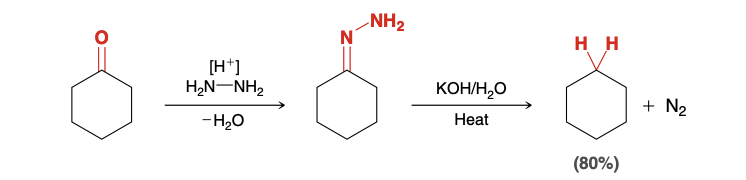
Wolff-Kishner Reduction (starting with ketone)
Reducing a ketone to an alkane
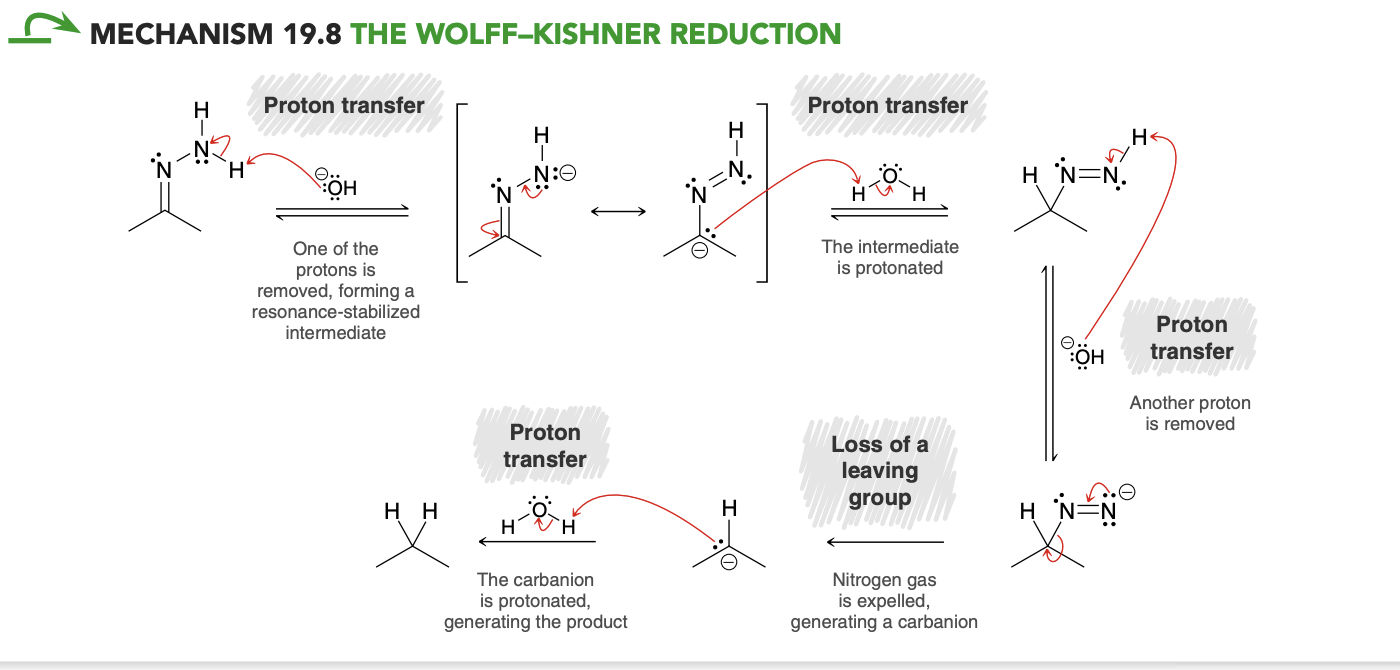
Wolff-Kishner Reduction Mechanism(don’t need to know?)
N2 gas step is irreversable and allows favorable yields.
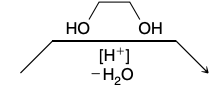
Hydrolysis of Acetals
Do not undergo hydrolysis under aqueous basic conditions
Reagent is H3O+
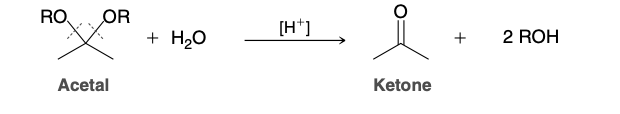
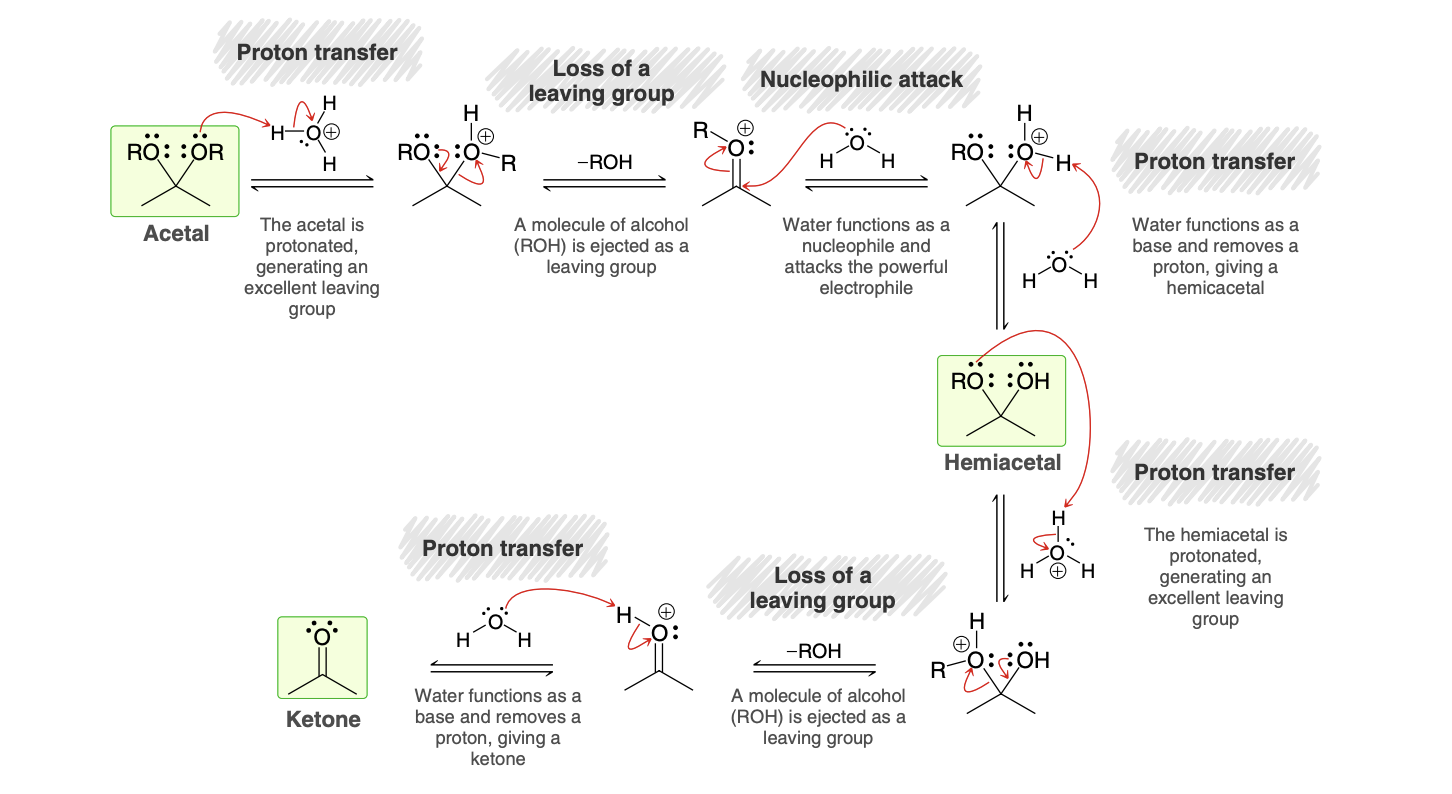
Hydrolysis of Acetals Mechanism
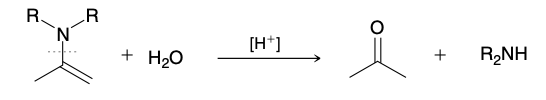
Imine Hydrolysis
Enamine Hydrolysis
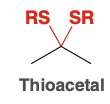
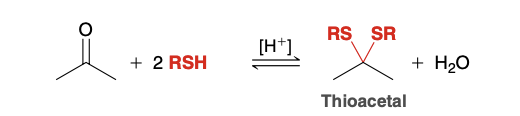
Thioacetal
thioacetal reagents
2 RSH
Acid Catalyst
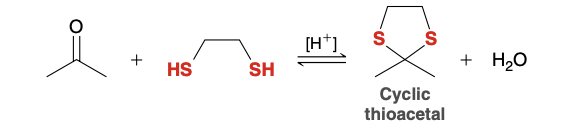
Cyclic thioacetal Reagent/formation
Ketone/Aldehyde
HS-CH2-CH2-SH
Acid Catalyst
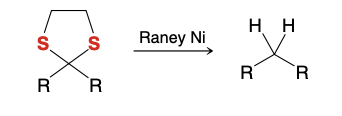
Desulfurization
Removing Sulfur molecules
Can be used in non-cyclic thioacetals too
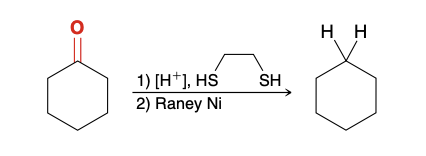
Thioacetal
Raney Ni
Full reduction of ketone using a thioacetal
Hydride
H-
Common Hydride reducing agents
LiAlH4 (+ H30+ in second step)
NaBH4(, MeOH)
LiALH4 vs NaBH4
LiAlH4 is much stronger and will fully reduce a ketone
Mechanism of ketones/aldehydes reduction with hydride agents
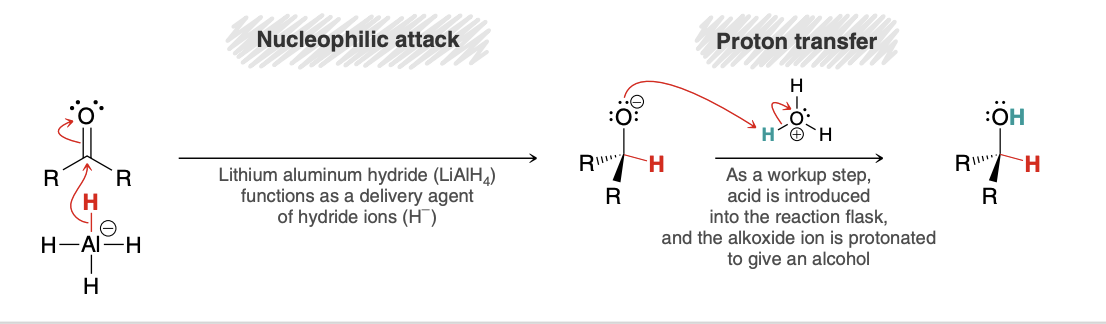
What happens if chiral center formed(hydride reduction)
Racemic mixture forms
Grignard Reagent
R-CH2-MgBr
(with H3O+)
Produces a nucleophilic(negative charge) carbon
Grignard reaction with Ketone/aldehyde
When treated with a Grignard reagent, aldehydes and ketones are converted into alcohols, accompanied by the formation of a new C-C bond
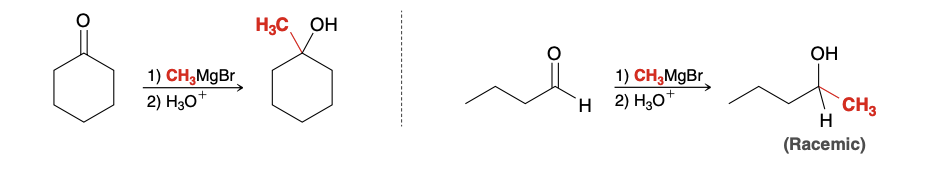
What happens if racemic micture is formed in Grignard Reaction
Racemic mixture formed
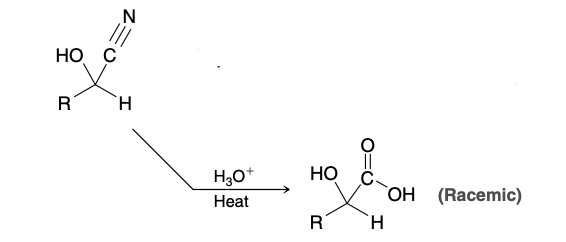
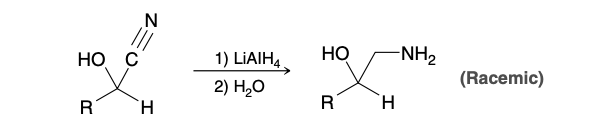
Cyanohydrin
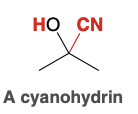
Cyanohydrin Reagents
HCN, KCN(produces more CN- anions)
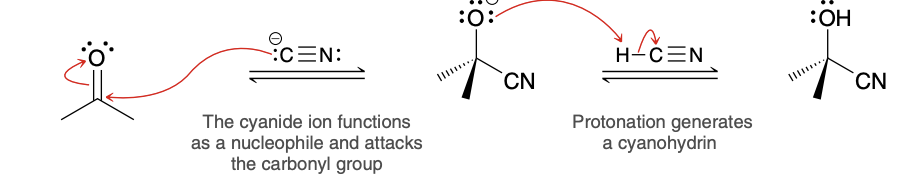
Cyanohydrin formation mechanism
Wittig Reagent
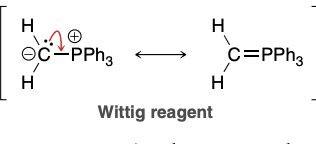
Wittig Reaction
Very useful for forming mono/di/tri substituted alkenes
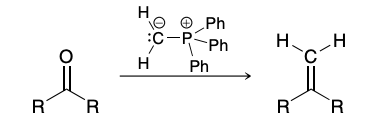
Wittig Reaction mechanism
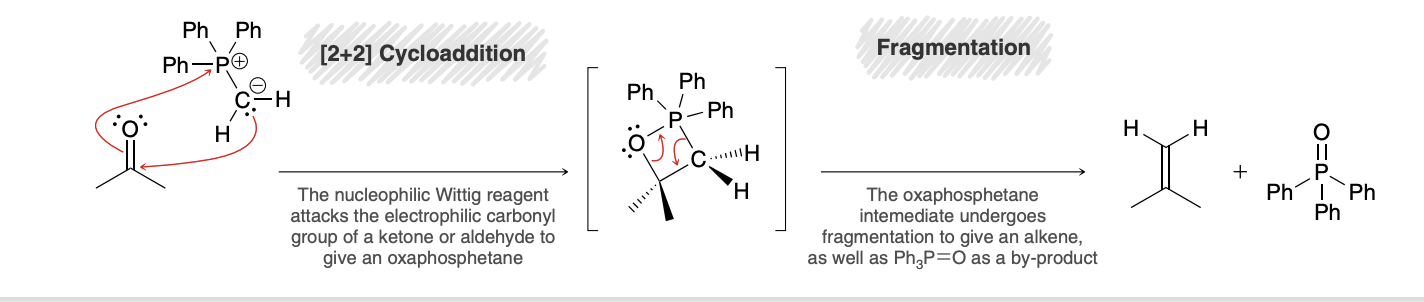
Wittig reagent formation
Start: Alkyl halide
1)PPh3 2) n-BuLi(any very strong base)
Sn2
Stereoselectivity of Wittig reaction
For a Wittig reagent only containing a simple alkyl halide: (Z)-alkene is favored
For a Wittig reagent containing an electron-withdrawing group: (E)-alkene is favored
Why in electron withdrawing group containing Wittig reagents does the (E)-alkene predominate?
Better stabalize the carbanion by resonance and is the stabilized, lower energy Wittig reagent
(E)-alkene is generally more stable, lower in energy
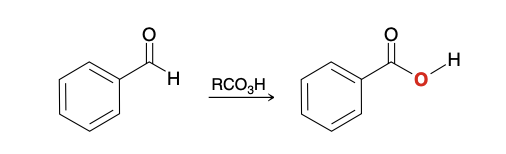
Baeyer– Villiger Oxidation of Aldehydes and Ketones
Converts ketones/aldehydes into esters
mCPBA
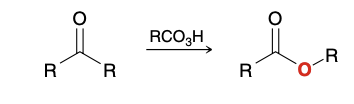
Regioselectivity of ester formation in unsymmetrical ketone
Certain groups will migrate better than others, and the ester oxygen will be inserted accordingly
Will be inserted on the side on the following priority; H > 3° > 2°, Ph > 1° > methyl
Cyanohydrin to amine
Cyanohydrin to carboxylic acid