colours of transition metals
1/3
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
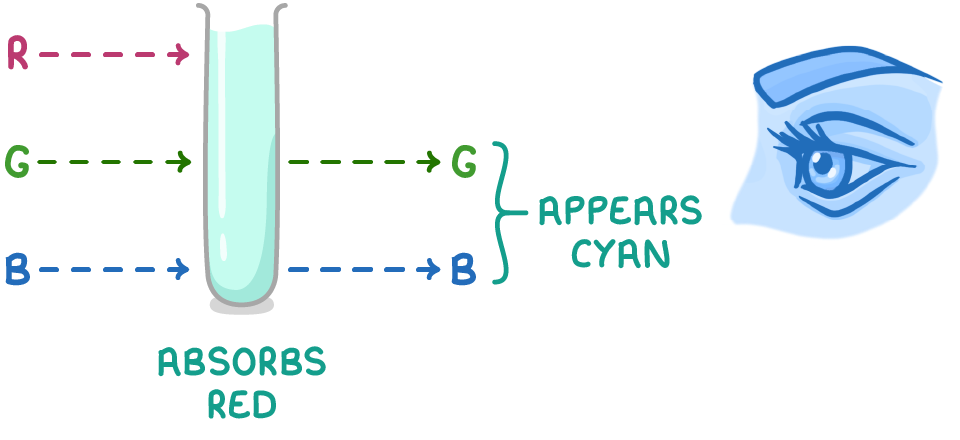
why are the colours of different transition metal complexes different?
d orbitals have different e/d orbital Es are split
e- move to excited state/higher E d orbitals
e- absorb visible light
colour seen is that from complementary colours that are reflected not absorbed
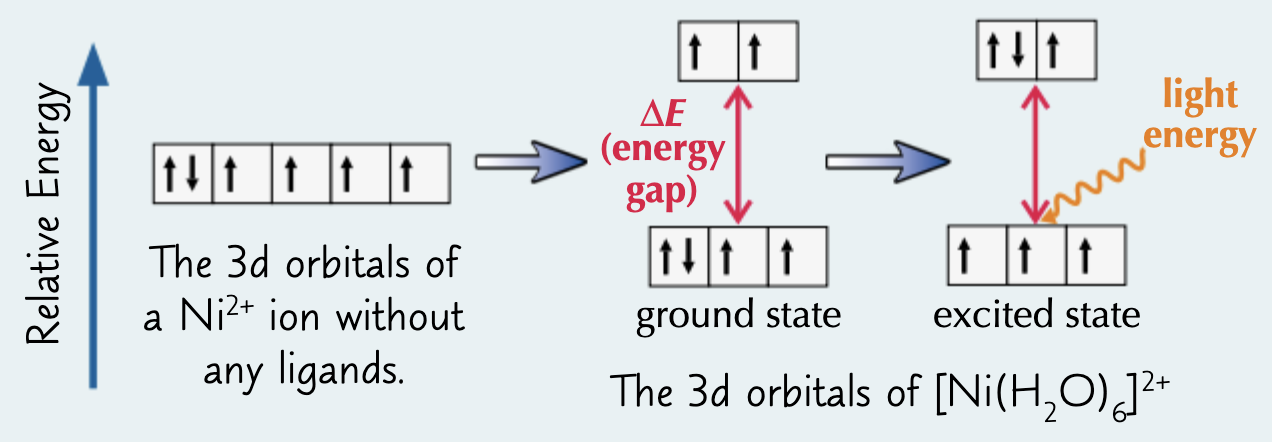
give the eqn for calculating the difference in E and state what each symbol corresponds to:
ΔE = h ν = hc / λ, where:
ΔE: energy absorbed / J
h: Planck’s constant = 6.63 × 10-34 Js
v (Greek letter nu): freq of light absorbed / Hz
c: speed of light = 2.998 × 108 ms-1
λ: wavelength of light absorbed / m
give 3 factors which might affect the colour of a transition metal complex:
change in oxidation state
change in coordination number
change in ligand
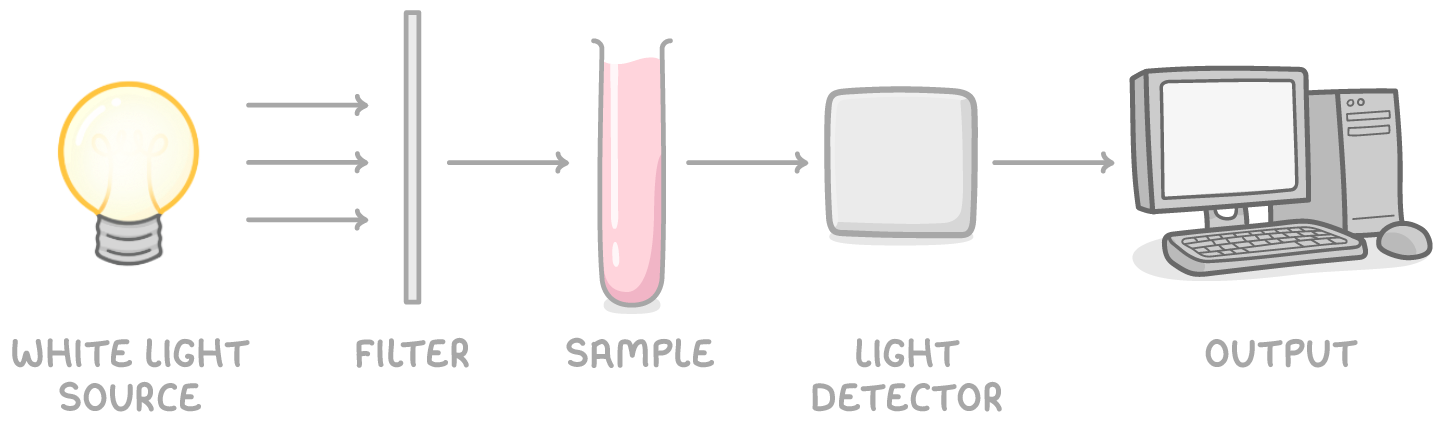
how can we use colorimetry to determine the concentration of transition metal ions in a solution of unknown concentration?
add an appropriate ligand to intensity colour
make up solutions of some known concentrations and measure absorbance
plot calibration curve of of absorbance v conc
measure absorbance of coloured complex and find conc from graph