CHE120: Chapter 2 Slides
1/72
Earn XP
Description and Tags
Status: In progress
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
Early Ideas in Atomic Theory
What did early Greek philosophers propose about matter,
Why isn’t their atomic theory considered scientific?
What key takeaway relates to how scientific ideas gain validity?
Greek philosophers in 5th century B.C. suggested that matter was made of tiny, indivisible particles called atoms (“atomos” — Greek for “indivisible”)
Had no experimental evidence, purely based on philosophy
Model persisted for ~2000 years w/o scientific validation until John Dalton proposed Atomic Theory in 1807
Key Takeaway: Scientific ideas require experimental support
Chemistry is a science based on __________ + __________.
Chemistry is a science based on observation + experimentation.
T or F: Theories can become laws.
False → Scientific laws and theories serve different purposes and exist on different tracks.
A law describes what happens (a pattern in nature).
A theory explains why it happens (the underlying mechanism).
Describe how scientific understanding progresses.
Empirical observations → Hypothesis → Law → Theory
Scientific Law v.s. Scientific Theory
Scientific Laws
Def: Concise summary of repeated observation/phenomenon
DESCRIBES WHAT HAPPENS
Does not explain mechanisms
Scientific Theories
EXPLAINS WHY a phenomenon occurs
Def: Explanation of hypothesis that was tested & supported by extensive experimental evidence
Can be revised with new data
T or F: A scientific law can give rise to a theory.
True → However, important nuance to remember is that a law can lead to a scientist developing an explanation for why said pattern (i.e. law) exists, but the law does NOT turn into a theory.
Describe the major components of the scientific method and explain how they relate to one another.
The scientific method consists of 4 major components that work together to generate reliable scientific knowledge/info:
(1) Empirical Facts = Observations + Data
Raw information that needs explanation
(2) Scientific Laws = Tested Generalizations
Repeated observations show consistent patterns
Scientist formulate laws to describe what happens in repeated observations that show consistent patterns (no explanation of why)
(3) Hypotheses = Tentative Explanations
To explain observations or laws, scientists propose hypotheses — testable falsifiable statements to attempt to answer why something happens
(4) Scientific Theories = Tested Explanations
When a hypothesis is repeatedly supported by experiments and evidence, it develops into a theory, which provides a comprehensive explanation of why natural phenomena occur. Theories can be refined as new data emerges.
Dalton’s Atomic Theory
A. List the 5 core postulates of Dalton’s Atomic Theory (the original version).
B. Which 2 postulates from the original theory were revised? When, why, and what is the correct modern view?
A. THE FIVE CORE POSTULATES OF ATOMIC THEORY
All matter is composed of extremely tiny & indivisible particles called atoms
Atoms of same element have the same mass & other properties
Atoms of different elements differ in their masses & other properties
Atoms of different elements combine in simple whole-number ratios to form compounds
In chemical reactions, atoms are neither created nor destroyed. Atoms are rearranged and total mass is conserved.
B. THE TWO MODERN CORRECTIONS OF DALTON’S ATOMIC THEORY
1) “All matter is composed of extremely tiny & indivisible particles called atoms.”
Revised When:
Sub-atomic particles—protons, neutrons, and electrons— were discovered, showing that atoms have an internal structure.
Corrected Modern View:
All matter is composed of extremely tiny particles called atoms.
Atoms have an internal structure (i.e. subatomic particles):
electrons
protons
neutrons
Atoms cannot be divided by chemical reactions, but can be altered in nuclear reactions/fusion (but they lose their elemental properties)
2) “Atoms of the same element are identical in mass and chemical properties.”
Revised When:
Isotopes (same element with different number of neutrons) were discovered
Corrected Modern View:
Atoms of same element have same chemical properties
Atoms of same element can have diff number of protons, and therefore diff masses
In what way is Dalton’s atomic theory still valid, and where does it fail?
Still Works: Dalton’s theory correctly describes chemical reactions — atoms rearrange but are not created or destroyed.
Dalton’s theory correctly describes chemical reactions
atoms are rearranged
atoms are not created or destroyed
mass is conserved
Fails:
Doesn’t acknowledge atomic structure — protons, neutrons, electrons
Doesn’t account for nuclear reactions, which is required to have knowledge of subatomic particles & isotopes
How does Dalton’s atomic theory explain the laws of definite and multiple proportions?
Law of Definite Proportions
Compounds always have the same mass ratios because they contain atoms in fixed whole‑number ratios
(e.g., water = 2 H : 1 O).
Law of Multiple Proportions
When elements form multiple compounds, the masses combine in simple whole‑number ratios because different compounds use different whole‑number combinations of atoms (e.g., CO vs. CO₂).
Core idea: Dalton’s model gives a physical explanation — atoms combine in simple, countable units, producing the mass ratios observed experimentally.
What is the mass relationship between the sub-atomic particles?
Proton mass ≈ neutron mass (both about 1 amu)
Both are much more massive than electrons
Electron mass is ~1/1836 the mass of a proton → effectively negligible in mass calculations
_______ and _______ dominate atomic mass, while _______ contribute almost none.
Protons and neutrons dominate atomic mass, while electrons contribute almost none.
What unit abbreviation does “amu” stand for? How is it defined?
amu = atomic mass unit
1 amu = 1/12 of the mass of a carbon-12 atom (which is the standard for measuring atomic mass
T or F: Isotopes differ in mass, but not in chemical behaviour.
True → Isotopes
How does the following relate to subatomic particles?
atomic number
mass number
Atomic number (Z) = number of protons
Defines the element
Never changes in chemical reactions
Mass number (A) = protons + neutrons
Determines isotope identity
Changes only if neutron count changes
Isotopes
• Same element (same Z)
• Different neutrons → different mass numbers
Using one sentence only, explain why isotopes behave chemically similarly?
Because they are the same element, therefore have the same electron structure (same proton count)
Atomic Symbols (Notation)
General Format:
A (mass number)
Z (atomic number)
X (elemental symbol)
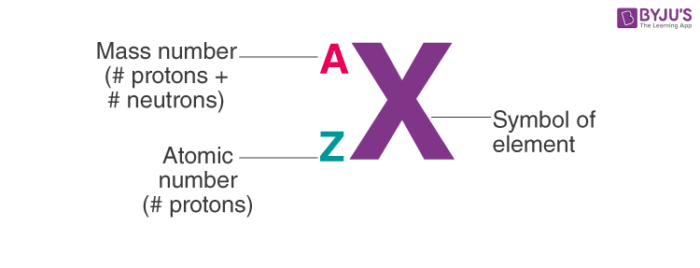
Atomic notation encodes both _________ and _________ in one compact symbol.
Atomic notation encodes both identity (Z) and isotope (A) in one compact symbol.

Based on the image above, determine the following:
(a) Name of element
(b) # of protons
(c) # of neutrons
Element: Carbon‑12
6 protons
6 neutrons (because 12−6=6)
Why are the masses on the periodic table non‑integers?
Each element exists as a mixture of isotopes.
Isotopes = atoms of the same element with different numbers of neutrons → different mass numbers.
The periodic table lists the weighted average of all naturally occurring isotopes.
Because the average includes multiple isotope masses and their abundances, the result is not an integer.
Example:
If an element has:
Isotope A: 10 amu (80%)
Isotope B: 11 amu (20%)
Average atomic mass =
(10)(0.80)+(11)(0.20)=10.2 amu → non‑integer
Key idea:
Non‑integer atomic masses reflect the natural isotope mixture and their relative abundances.
T or F: Elements on the periodic table are arranged by atomic number and mass.
False → Elements on periodic table arranged by atomic number ONLY (not arranged by mass)
Mass Number v.s. Atomic Mass
Mass Number → integer → single atom/isotope
Atomic Mass → weighted average → reflects natural isotope abundance
Describe how the periodic table is organized.
Periods (rows):
Correspond to principal energy levels (n = 1, 2, 3, …)
Elements in the same period have electrons filling the same highest energy level
Groups (columns):
Elements share similar chemical behavior
Because they have similar valence electron configurations
Why do elements in the same group behave similarily?
Same group → same number of valence electrons → similar reactivity & bonding behaviour
Elements arranged horizontally by increasing energy level are called _______ or _______.
Elements arranged horizontally by increasing energy level are called periods or rows.
The ____________ of elements are almost entirely determined by valence electron configuration.
The chemical behaviour of elements are almost entirely determined by valence electron configuration.
Elements in the same vertical column belong to the same _______ or _______.
Elements in the same vertical column belong to the same group or family.
A new period begins on the periodic table whenever a new principal ______________ begins to fill with electrons.
A new period begins on the periodic table whenever a new principal energy level (shell) begins to fill with electrons.
Group 18 elements, known as _________, are generally non-reactive because they have a _________ shell.
Group 18 elements, known as noble gases, are generally non-reactive because they have a full outer shell.
The chemical behaviour of an element is most strongly influenced by its _________________________.
The chemical behaviour of an element is most strongly influenced by its valence electron configuration.
What are the key properties of metals?
Good conductors of heat & electricity
Malleable (can be hammered into sheets)
Tend to lose electrons & form cations (positively-charged ions)
What are the key properties of nonmetals?
Poor conductors (of electricity)
Often brittle (if solid)
Tend to gain electrons & form anions (negatively-charged ions)
What are the key properties of metalloids?
intermediate b/w metals & non-metals
semiconductors
properties depend on conditions (e.g. temperature, purity of sample, etc.)
What does “purity” mean in regards to metalloids?
Purity = fraction of a sample that is the actual element.
High purity → almost all atoms are the metalloid
Low purity → contains small amounts of other elements (impurities)
Metalloids (Si, Ge, B) are extremely sensitive to impurities because tiny amounts of other elements can dramatically change electrical conductivity.
High-Purity Metalloid
• Behaves more like a nonmetal
• Poor conductor at room temperature
• Conductivity increases with temperature
Low-Purity Metalloid (Doped)
• Behaves more like a metal / semiconductor
• Conductivity increases dramatically
• Essential for electronic devices
Ion
Charged atom or molecule
Cation v.s. Anion
(provide examples for each)
Cations (+)
Positively-charged ion
Formed when an atom loses one or more electrons
Example: A neutral sodium atom (Z = 11) that loses one electrons will become a cation with a 1+ charge (11 − 10 = 1+), Na+
Anions (-)
Negatively-charged ion
Formed when an atom gains one or more electrons
Example: A neutral oxygen atom (Z = 8) that gains two electrons will become an anion with a 2− charge (8 − 10 = 2−), O2−
What determines the charge of ions?
Charge is determined by electron transfer (loss or gain of electrons)
Proton count NEVER CHANGES in chemical reactions
Changing protons would change the element, which only happens in nuclear reactions
T or F: The transferring of electrons and protons from or to an ion determines its charge.
FALSE!!! Proton count NEVER CHANGES in chemical reactions. Changing protons changes the element, which only happens in nuclear reactions.
T or F: Transitional metals have variable charges.
True
Group 1 Elements…
(a) Group Name
(b) Location on Periodic Table
(c) What charge do they form?
(a) Group Name: Alkali Metals
(b) Location: Furthest left column (excluding H)
(c) Charge: +1 (lose 1 electron)
Group 2 Elements…
(a) Group Name
(b) Location on Periodic Table
(c) What charge do they form?
(a) Group Name: Alkaline Earth Metals
(b) Location: 2nd column to the left
(c) Charge: +2 (loses 2 electrons)
Group 13 Elements…
(a) Group Name
(b) Location on Periodic Table
(c) What charge do they form?
(a) Group Name: Boron Group
(b) Location: 3rd column of the p-block
(c) Charge: +3 (lose 3 electrons)
Group 15 Elements…
(a) Group Name
(b) Location on Periodic Table
(c) What charge do they form?
(a) Group Name: Pnictogens
(b) Location: 3rd column from the right
(c) Charge: -3 (gain 3 electrons)
Group 16 Elements…
(a) Group Name
(b) Location on Periodic Table
(c) What charge do they form?
(a) Group Name: Chalcogens
(b) Location: 2nd column from the right
(c) Charge: -2 (gain 2 electrons)
Group 17 Elements…
(a) Group Name
(b) Location on Periodic Table
(c) What charge do they form?
(a) Group Name: Halogens
(b) Location: 2nd to last column on the right
(c) Charge: -1 (gain 1 electron)
Group 18 Elements…
(a) Group Name
(b) Location on Periodic Table
(c) What charge do they form?
(a) Group Name: Noble gases
(b) Location: Furthest right column
(c) Charge: 0 (generally don’t form ions)
T or F: The terms "metalloid" and "transition metal" are interchangeable.
False → They are two distinct categories of elements with different locations on periodic table.
Metalloid
Metalloids = Semi-metals
Definition: Elements with properties intermediate between metals and nonmetals.
Location: Found along the diagonal "staircase" line (e.g., Silicon, Germanium, Arsenic).
Properties: Often brittle, semiconductors, and have intermediate conductivity.
Transition Metals
Definition: Elements in groups 3-12 with partially filled d-orbitals.
Location: The central block of the periodic table (e.g., Iron, Copper, Gold).
Properties: Typically malleable, ductile, excellent conductors of heat and electricity, and often form coloured compounds.
Another term for metalloids is __________.
Another term for metalloids is semi-metals.
Which element in the first column of the periodic table is not considered part of group 1?
Hydrogen isn’t considered part of group 1 or alkali metals.
What does it mean when the number of protons & electrons are not equal in an atom?
it means the atom is electrically charged → its an ion
T or F: An ion cannot have a charge of 0.
True
Atoms and molecules acquire charge by….
losing or gaining electrons
The number of ______ in an element are always the same.
The number of protons in an element are always the same.
Formula to determine the charge of an ion
Charge = number of protons — number of electrons
What defines an ionic compound?
Metal + nonmetal
Electron transfer
Form ions held together by electrostatic attraction
What defines a molecular compound?
Nonmetal + nonmetal
Electron sharing
Form molecules
What happens when many nonmetals form ions?
They gain electrons until they have the same number of electrons as the next noble gas.
What happens when many main‑group metals form ions?
They lose electrons until they have the same number of electrons as the preceding noble gas.
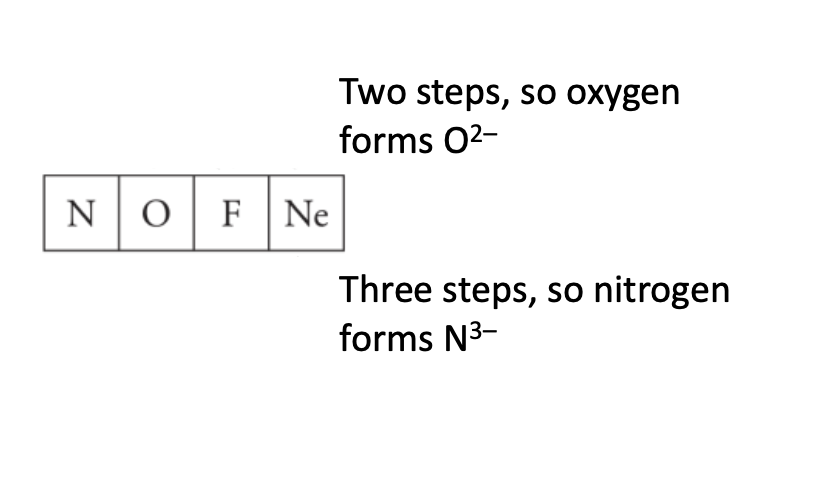
Predict the ionic charge of O and N.
Polyatomic Ion
Molecules with 2 or more atoms w/ an overall positive (+) or negative (-) charge

Oxyanion
Polyatomic ions w/ one or more oxygen atoms are called oxyanions w/ an “-ate” suffix

Monatomic Ion
Definition
Example
Def: Ions formed by 1 atom
E.g: Na+ (Sodium ion)
Polyatomic Cations
Definition
Provide name and formula of the 2 most common examples.
Molecular ion with…
2 or more atoms
Positive charge
Examples:

Polyatomic Anions
Definition
What is the simplest type?
Provide name and formula of the 4 most common examples.
Definition
Molecular ions w/
2 or more atoms
negative charge
Simplest Type of Polyatomic Ion
Oxyanion = [oxygen] + [nonmetal] or [transition metal]
ends with “-ate” in its name
Most Common Examples
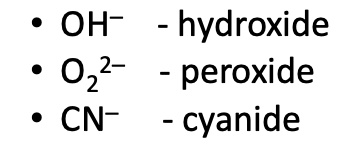
For each of the major “-ate” oxyanions, list:
Name
# of Oxygens (hint: consonants)
# of Charge (hint: vowels)
Polyatomic Ion Formula
Hint: “Nick Clause Caused Super Crazy Plots”
Atomic Number
The atomic number (Z) defines element by the total number of protons in its nucleus.
Mass Number
Mass Number (A): The total count of protons and neutrons (nucleons) in an atom's nucleus.