Types of Chemical Reactions: Acid-Base, Redox, and Theories
1/74
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
What are the two main types of chemical reactions discussed?
Acid-Base Reactions and Redox Reactions
What is the Arrhenius Acid-Base Model?
An acid dissolves in water to give H+ (a proton) and a base dissolves in water to give -OH (hydroxide).
What is a limitation of the Arrhenius model?
It ignores the role of water and is too simple.
What is a better representation of H+ in aqueous solution?
H3O+ (the hydronium ion) is a better way to represent H+.
Name two common Arrhenius acids.
HCl and H2SO4
Name two common Arrhenius bases.
NaOH and KOH
What is the general form of an Arrhenius Acid-Base Reaction?
Acid + Base → Salt + Water
What are spectator ions in an Arrhenius reaction?
Ions that do not participate in the actual chemical change, such as Na+ and Cl-.
What is the Bronsted-Lowry definition of an acid?
An acid is a proton donor.
What is the Bronsted-Lowry definition of a base?
A base is a proton acceptor.
What is the significance of conjugate acids and bases?
Every acid has a conjugate base and every base has a conjugate acid.
What does it mean for water to be amphiprotic?
Water can act as both an acid and a base.
What is the Lewis definition of an acid?
A Lewis acid is an electron pair acceptor.
What is the Lewis definition of a base?
A Lewis base is an electron pair donor.
How does the Lewis theory relate to Bronsted-Lowry and Arrhenius theories?
Lewis theory encompasses and explains both Bronsted-Lowry and Arrhenius theories.
What is a nucleophile?
A nucleophile is a Lewis base that donates an electron pair.
What is an electrophile?
An electrophile is a Lewis acid that accepts an electron pair.
What factors contribute to the acidity of a compound?
Strong interactions between acidic H and water, and the stability of the conjugate base.
What common feature do bases share?
They all have a lone pair of electrons available to form a new bond.
Why are electronegative atoms important in basicity?
Electronegative atoms typically have lone pairs but are also inclined to hold onto their electrons.
What role do nitrogen-containing compounds play in biological systems?
Many biological bases have nitrogen at their center, which is crucial for hydrogen bonding in DNA and RNA.
What is the reaction between NH3 and H2O?
NH3(aq) + H2O(l) ⇌ NH4+(aq) + -OH(aq)
What is the importance of Lewis structures in identifying acids and bases?
Lewis structures help visualize the electron pairs and bonding in acids and bases.
How can you identify an acid based on its structure?
Acids typically have acidic hydrogens bonded to electronegative elements.
What is the relationship between Bronsted acids and Lewis acids?
All Bronsted acids are also Lewis acids.
What is the significance of the equilibrium arrows in Bronsted-Lowry reactions?
They indicate that the reaction can proceed in either direction.
What is the net ionic equation for the reaction between HCl and NaOH?
H+(aq) + -OH(aq) ⇌ H2O(l)
What is N-methylation in the context of epigenetics?
N-methylation refers to the addition of a methyl group to adenine, specifically forming N-6 methyl adenine, affecting gene expression.
What is the general reaction for simple acid-base reactions?
Simple acid-base reactions involve a proton transfer from an acid to a base.
What occurs in the reaction H2O + H2O?
It produces H3O+ and OH-.
What distinguishes strong acids from weak acids?
Strong acids are fully ionized in solution, while weak acids are not fully ionized.
What is the effect of a strong acid on a light bulb in an experiment?
A strong acid causes the bulb to glow brightly due to complete ionization.
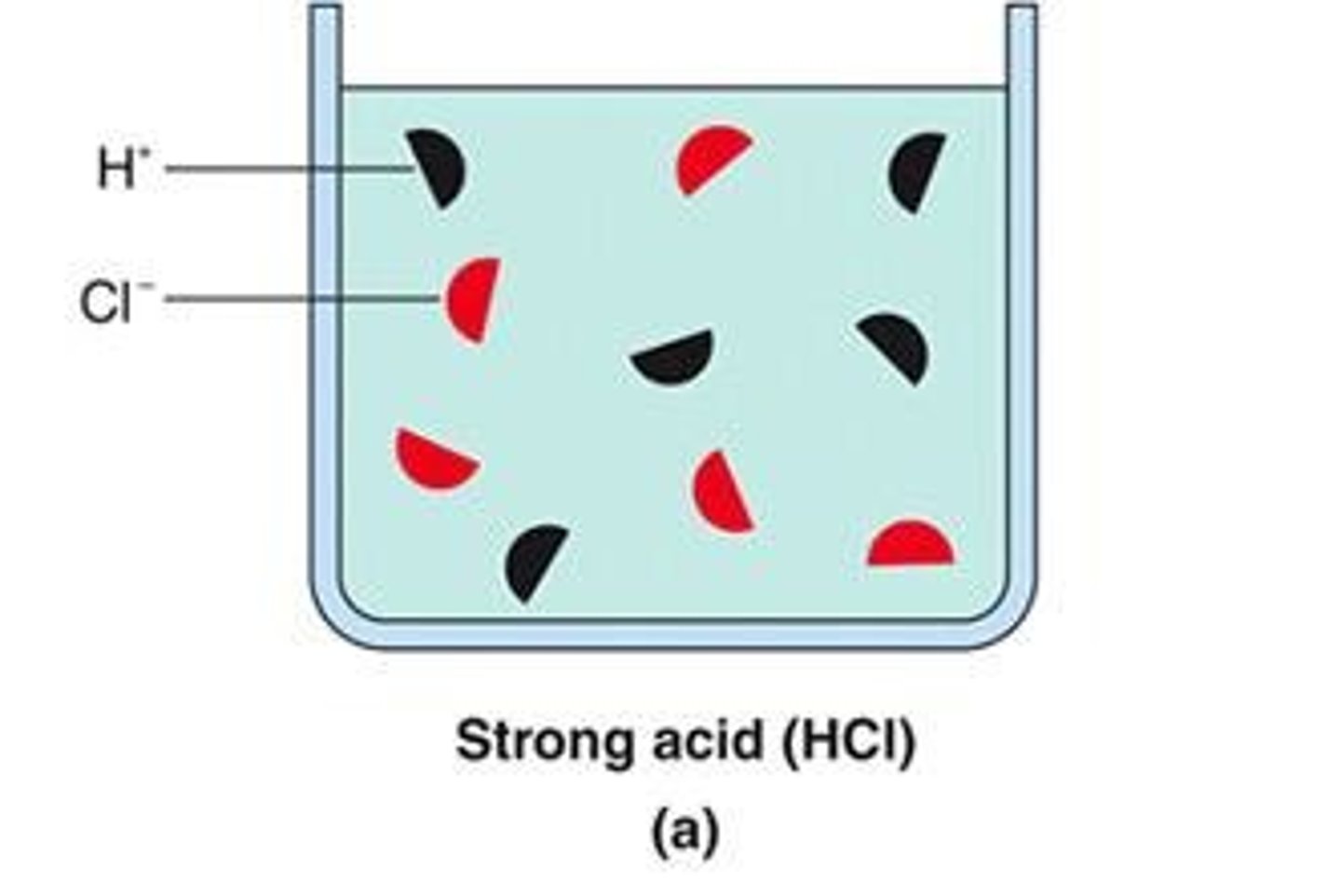
What happens to HCl in a 0.1 M solution?
HCl completely ionizes to produce H+ and Cl-.
What is the ionization equation for a weak acid like acetic acid (CH3COOH)?
CH3COOH ⇌ CH3COO- + H+.
How does acid strength vary across a row in the periodic table?
Acid strength increases across a row due to increasing electronegativity and bond polarization.
Why is HF a weak acid despite fluorine's high electronegativity?
HF has a strong H-F bond that is difficult to break, resulting in weak ionization.
What factors affect acid strength down a group?
Acid strength down a group is influenced by bond strength (H-X bond weakening) and entropy changes.
What is the relationship between acid strength and ΔG?
Stronger acids have more negative ΔG values, indicating a greater tendency to ionize.
What happens to solute-solute and solvent-solvent interactions when an acid dissolves in water?
Solute-solute and solvent-solvent interactions are overcome, requiring energy.
What is bond enthalpy?
Bond enthalpy is the energy required to break a bond, such as the H-X bond in the gas phase.
What is the significance of resonance in strong acids?
Resonance allows for the stabilization of conjugate bases by spreading out negative charge.
What is the definition of a strong acid?
A strong acid is one that completely ionizes in solution, such as HCl, HBr, HI, HNO3, and H2SO4.
What are examples of strong bases?
Strong bases include any group I or II hydroxide, such as NaOH and KOH.
How does the stability of a conjugate base relate to acid strength?
The weaker the conjugate base, the stronger the acid; stability can be enhanced by charge delocalization.
What is the role of entropy (ΔS) in acid-base reactions?
Entropy changes can influence the favorability of the reaction and the strength of the acid.
What does a concentrated solution of an acid imply?
Concentration refers to the amount of acid in a given volume, not its strength.
What is the difference between dilute and concentrated solutions?
Dilute solutions have a lower concentration of solute compared to concentrated solutions.
What happens to the H-X bond during acid ionization?
The H-X bond breaks, requiring energy, while an O-H bond forms, releasing energy.
What is the effect of electronegativity on acid strength?
Higher electronegativity leads to stronger acids due to more polarized bonds.
How does the size of atoms affect acid strength down a group?
As atomic size increases, bond strength decreases, making it easier to ionize.
What is a resonance hybrid?
A resonance hybrid is a representation of a molecule that shows delocalization of electrons across multiple structures.
What is the significance of the conjugate base's stability in determining acid strength?
A stable conjugate base indicates a stronger acid, as it can better accommodate the negative charge.
What is the relationship between acid strength and bond strength?
Weaker H-X bonds lead to stronger acids, as they are easier to break during ionization.
What happens to atoms during a chemical reaction?
Atoms are rearranged, and their connections change.
What is the definition of a chemical reaction?
A process involving the rearrangement of atoms, where atoms are conserved.
What is the difference between limiting and excess reagents?
The limiting reagent runs out first, while excess reagents are leftover after the reaction.
What does the balanced reaction provide in terms of stoichiometry?
It gives molar relationships between reactants and products in a chemical process.
What is the theoretical yield?
The maximum amount of product that can be produced from the reactants on hand.
What is the actual yield?
The amount of product that is actually obtained from a reaction.
How do you calculate percent yield?
Percent yield = (actual yield / theoretical yield) × 100.
What is molarity?
Molarity = moles of solute / liters of solution.
What is a solute?
The substance that is dissolved in a solution, usually in minority.
What is a solvent?
The liquid in which a solute is dissolved, usually in majority.
What does it mean to dilute a solution?
To reduce the concentration of a solution by adding more solvent.
What is the purpose of solution stoichiometry?
To determine how much solution is needed to complete a reaction using known concentrations.
What happens when HCl gas is dissolved in water?
It forms HCl(aq), which conducts electricity.
What is the significance of a solution conducting electricity?
It indicates that ions are present in the solution, suggesting a chemical reaction has occurred.
What is the formula for calculating molarity?
M = moles of solute / liters of solution.
What is an example of a chemical reaction?
Combustion of hydrocarbons, such as burning gasoline.
Can a solution formation involve a chemical reaction?
Sometimes; it depends on the nature of the solute and solvent.
What is the role of valence electrons in chemical reactions?
Changes in the location of valence electrons drive reactions and reactivity.
What is the relationship between concentration and volume in solution stoichiometry?
Knowing the concentration allows conversion between moles and liters of solution.
What is the effect of temperature on solubility?
Generally, solubility increases with temperature for solids in liquids.
What is the Lewis structure used for?
To predict the properties of molecules, including their behavior in solutions.
What are some criteria for classifying chemical reactions?
Type of reaction (synthesis, decomposition, etc.), energy changes, and reactants/products involved.