lesson 4
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
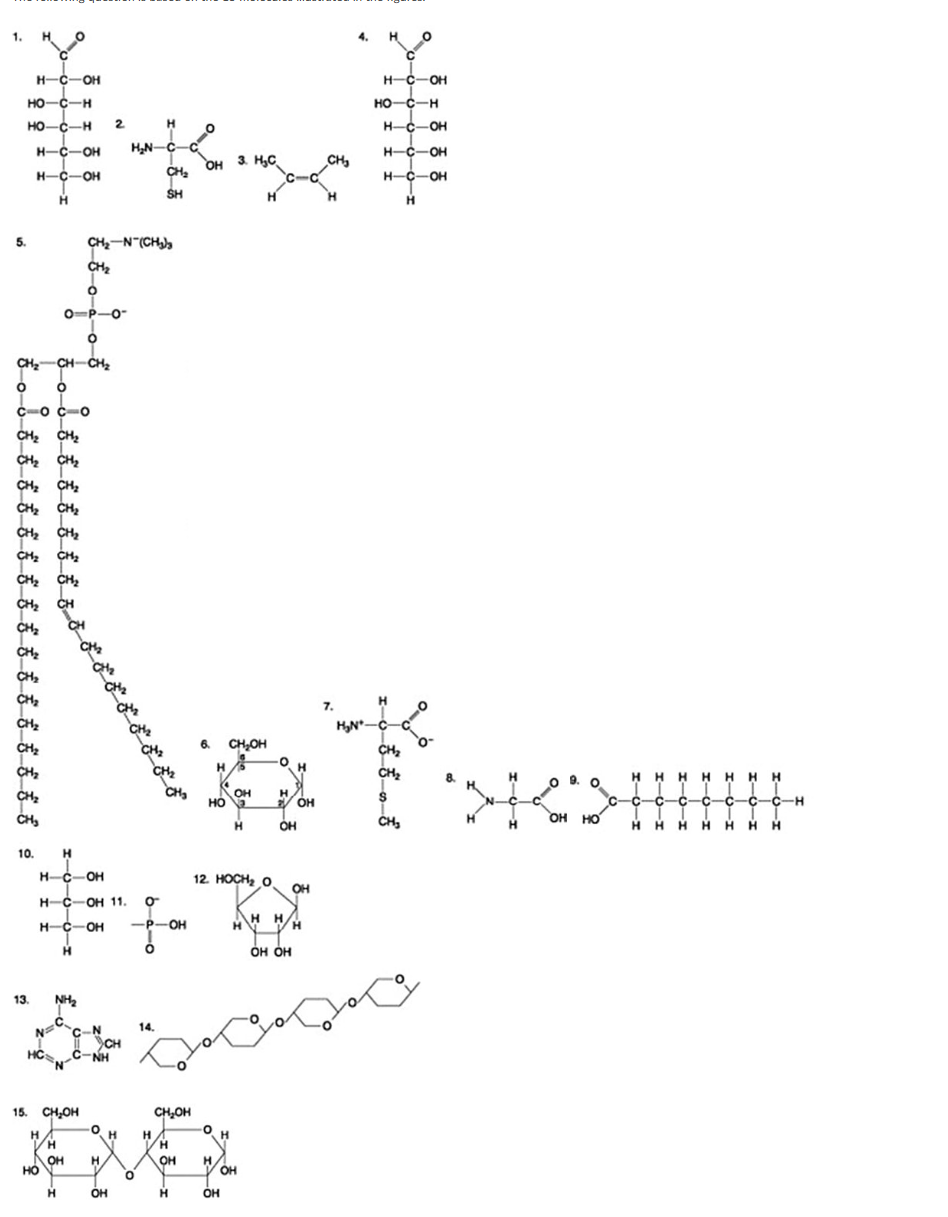
The following question is based on the 15 molecules illustrated in the figures.
Which of the following molecules is a purine nitrogenous base?
13
2
12
5
13
The term insoluble fiber on food packages refers to which of the following molecules?
cellulose
polypeptides
starch
amylopectin
cellulose
How many molecules of water are required for the polymerization of a 22 monomer-long cellulose molecule?
22
21
44
11
21
Which of the following best compares dehydration reactions and hydrolysis?
Dehydration reactions assemble polymers; hydrolysis reactions break polymers apart.
Dehydration reactions eliminate water from membranes; hydrolysis reactions add water to membranes.
Hydrolysis reactions assemble polymers; dehydration reactions break polymers apart.
Dehydration reactions and hydrolysis reactions assemble polymers from monomers.
Dehydration reactions assemble polymers; hydrolysis reactions break polymers apart.
Misfolding of polypeptides is a serious problem in cells. Which of the following diseases are associated with an accumulation of misfolded polypeptides?
Alzheimer's only
Alzheimer's and Parkinson's
Diabetes mellitus
Parkinson's only
Alzheimer's and Parkinson's
Which of the following is similar between RNA and DNA?
number of strands
type of sugar
type of pyrimidine base
type of purine base
type of purine base
Use the following information to answer the following questions.
"The native structure of hemoglobin (HB) comprises of two α and two β subunits, each of which carries a heme group. There appear to be no previous studies that report the in-vitro folding and assembly of Hb from highly unfolded α and β globin in a 'one-pot' reaction. One difficulty that has to be overcome for studies of this kind is the tendency of Hb to aggregate during refolding. This work demonstrates that denaturation of Hb in 40% acetonitrile at pH 10.0 is reversible." (J Am Soc Mass Spectrum 2007, 18, 8-16)
Which of the following statements about hemoglobin is most consistent with the information in the passage?
a quaternary protein with two polypeptides
a tertiary protein with two polypeptides
a tertiary protein with four polypeptides
a quaternary protein with four polypeptides
a quaternary protein with four polypeptides
The term insoluble fiber on food packages refers to which of the following molecules?
amylopectin
cellulose
starch
polypeptides
cellulose
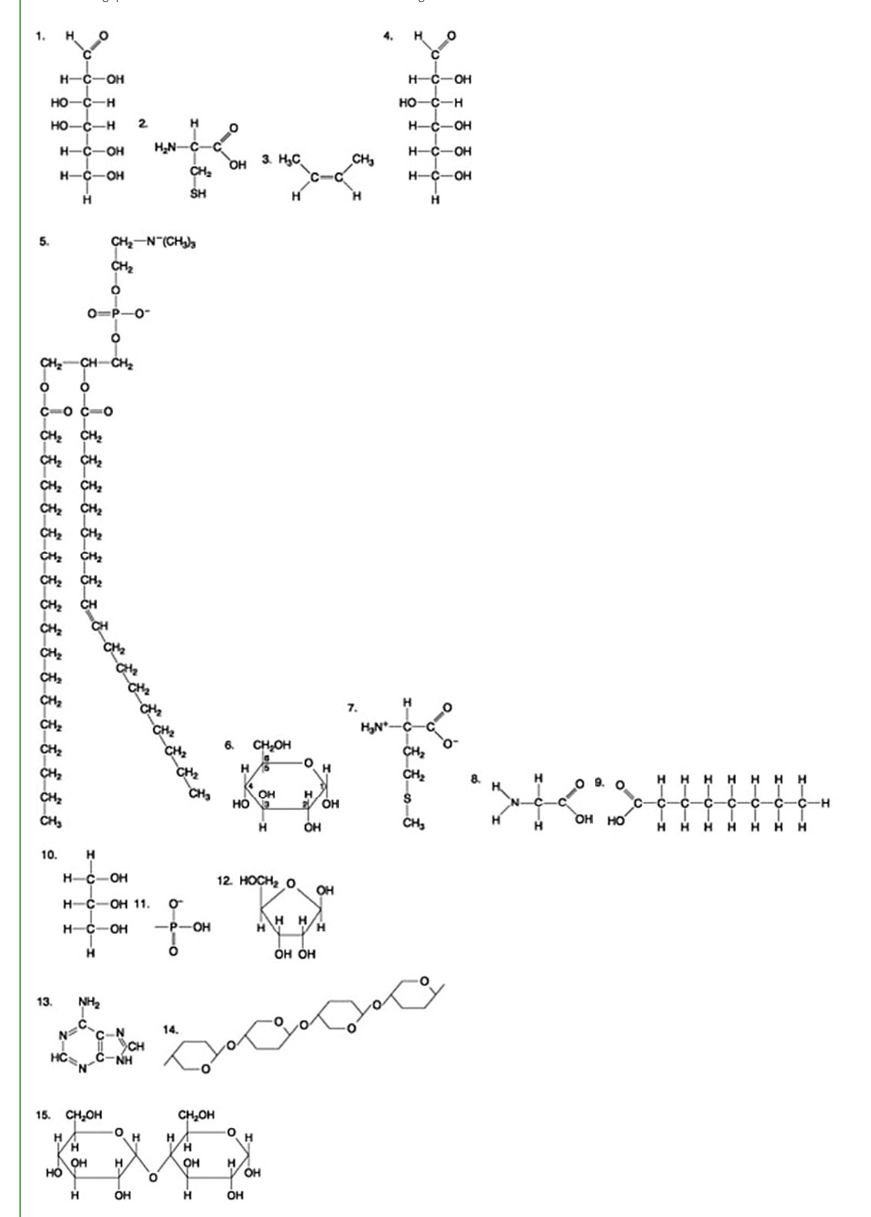
Which of the following molecules is a disaccharide?
6
3
1 and 4
15
15
Which of the following statements describes the interaction of water molecules with phospholipids?
Phospholipids dissolve in water.
The polar heads avoid water; the nonpolar tails attract water (because water is polar and opposites attract).
Phospholipids do not interact with water because water is polar and lipids are nonpolar.
The polar heads interact with water; the nonpolar tails do not.
The polar heads interact with water; the nonpolar tails do not.
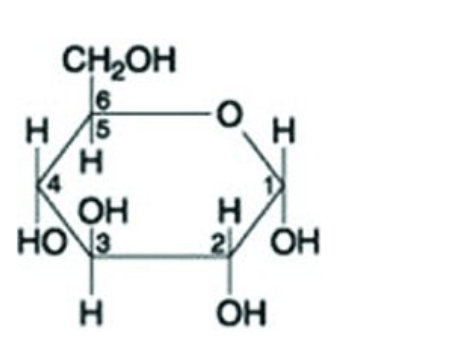
Use the following figure to answer the question.
What is the molecule represented by the figure above?
maltose
a hexose
a pentose
fructose
a hexose
Which parts of the amino acids AA1 and AA2 are involved in the formation of a peptide bond?
AA1–AA2
side chains of both AA1 and AA2
amino group of AA1 and carboxyl group of AA2
carboxyl group of AA1 and side chain of AA2
carboxyl group of AA1 and amino group of AA2
carboxyl group of AA1 and amino group of AA2
If 14C-labeled uracil is added to the growth medium of cells, what macromolecules will be labeled?
RNA
DNA
polypeptides
both DNA and RNA
RNA
Which parts of the amino acids AA1 and AA2 are involved in the formation of a peptide bond?
AA1–AA2
side chains of both AA1 and AA2
amino group of AA1 and carboxyl group of AA2
carboxyl group of AA1 and amino group of AA2
carboxyl group of AA1 and side chain of AA2
carboxyl group of AA1 and amino group of AA2
Which of the following statements correctly describes "saturated fats"?
They are generally liquid at room temperature.
They are more common in plants than in animals.
They have multiple double bonds in the carbon chains of their fatty acids.
They contain more hydrogen than unsaturated fats that consist of the same number of carbon atoms.
They contain more hydrogen than unsaturated fats that consist of the same number of carbon atoms.
Which parts of the amino acids AA1 and AA2 are involved in the formation of a peptide bond?
AA1–AA2
amino group of AA1 and carboxyl group of AA2
side chains of both AA1 and AA2
carboxyl group of AA1 and amino group of AA2
carboxyl group of AA1 and side chain of AA2
AA1–AA2
Which of the following is a biological advantage of storing glucose molecules in branched polymers?
They have more sites for enzymatic attack than do unbranched polymers.
They can be stacked together to save space.
They are insoluble and so provide strength to the molecule.
They are resistant to enzymes.
They have more sites for enzymatic attack than do unbranched polymers.
Why can humans digest starch but not cellulose?
Starch monomers are joined by covalent bonds, and cellulose monomers are joined by ionic bonds.
Humans have enzymes that can hydrolyze the α-glycosidic linkages of starch but not the β-glycosidic linkages of cellulose.
Starch is softer than cellulose.
The monomer of starch is glucose, while the monomer of cellulose is galactose.
Humans have enzymes that can hydrolyze the α-glycosidic linkages of starch but not the β-glycosidic linkages of cellulose.
Use the following information to answer the following questions.
"The native structure of hemoglobin (HB) comprises of two α and two β subunits, each of which carries a heme group. There appear to be no previous studies that report the in-vitro folding and assembly of Hb from highly unfolded α and β globin in a 'one-pot' reaction. One difficulty that has to be overcome for studies of this kind is the tendency of Hb to aggregate during refolding. This work demonstrates that denaturation of Hb in 40% acetonitrile at pH 10.0 is reversible." (J Am Soc Mass Spectrum 2007, 18, 8-16)
Based on the information in the passage, the total number of heme groups present in four hemoglobin protein molecules is ________.
12
4
16
8
16
Which of the following statements about lipids is true?
Lipids are true polymers.
Lipids don't constitute of waxes and pigments.
Lipids mix poorly with water.
Lipids are not organic in nature.
Lipids mix poorly with water.
Ovalbumin, the egg white protein is denatured in scrambled egg. Which of the following is not affected?
tertiary structure of ovalbumin
quaternary structure of ovalbumin
secondary structure of ovalbumin
primary structure of ovalbumin
primary structure of ovalbumin
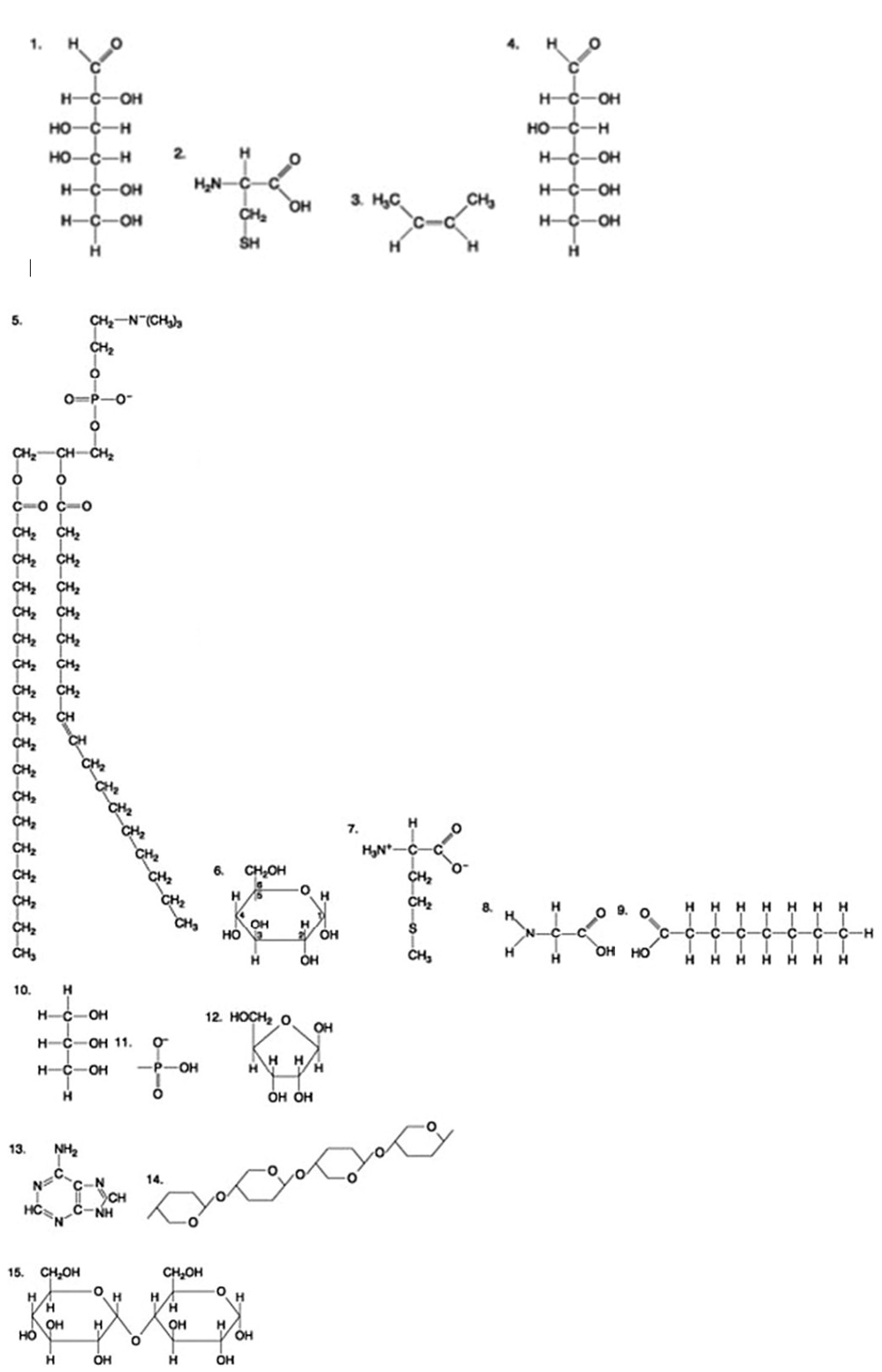
The following question is based on the 15 molecules illustrated in the figures.
Which of the following molecules is the pentose sugar found in RNA?
1
12
13
6
12
Use the following information to answer the following questions.
"The native structure of hemoglobin (HB) comprises of two α and two β subunits, each of which carries a heme group. There appear to be no previous studies that report the in-vitro folding and assembly of Hb from highly unfolded α and β globin in a 'one-pot' reaction. One difficulty that has to be overcome for studies of this kind is the tendency of Hb to aggregate during refolding. This work demonstrates that denaturation of Hb in 40% acetonitrile at pH 10.0 is reversible." (J Am Soc Mass Spectrum 2007, 18, 8-16)
In sickle-cell disease, as a result of a single amino acid change, the mutant hemoglobin tetramers associate with each other and assemble into large fibers. Based on this information alone, what does the sickle-cell hemoglobin exhibit?
only altered quaternary structure
only altered tertiary structure
altered primary structure and altered quaternary structure; the secondary and tertiary structures may or may not be altered
only altered primary structure
altered primary structure and altered quaternary structure; the secondary and tertiary structures may or may not be altered
The label on a container of margarine lists "hydrogenated vegetable oil" as the major ingredient. Which of the following options describes the property of hydrogenated vegetable oil?
It is less likely to block arteries.
It has more double bonds in the fatty acid chains.
It has fewer trans fatty acids.
It is solid at room temperature.
It is solid at room temperature.
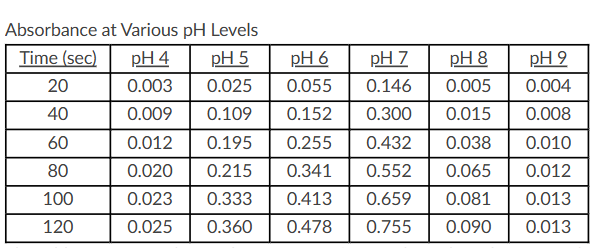
Use the following table to answer the question.
The table represents the results of an experiment where the effects of pH buffers on an enzyme found in saliva (amylase) were studied. A spectrophotometer set at 500 nm was used to measure absorbance at the various pH levels every 20 seconds for 2 minutes. The higher absorbance values would indicate greater enzyme activity. All experiments were conducted at the same temperature.
Which statement correctly identifies the result that the optimum pH for amylase function is 7?
The pH with the lowest absorbance values would indicate the optimum pH for amylase since this pH does not affect the structure or function of the protein.
At pH 9, the enzyme is denatured and will lose its function, but not its structure.
The pH with the highest absorbance values would indicate the optimum pH for amylase since this pH does not affect the structure or function of the protein.
At pH 4, the structure of the enzyme will be altered, and the enzyme would not be able to catalyze the reaction.
The pH with the highest absorbance values would indicate the optimum pH for amylase since this pH does not affect the structure or function of the protein.
If 14C-labeled uracil is added to the growth medium of cells, what macromolecules will be labeled?
DNA
RNA
polypeptides
both DNA and RNA
RNA
An individual experiences abdominal discomfort after consuming a drink containing milk. A loss of function of which enzyme is most likely responsible for the symptoms?
maltose that breaks down the bond between two glucose molecules
lactose that breaks down the bond between two glucose molecules
maltase that breaks down the bond between galactose and glucose
lactase that breaks down the bond between galactose and glucose
lactase that breaks down the bond between galactose and glucose
Which of the following statements about proteins is true?
Some proteins form a complete 3-D structure only when they interact with their targets.
Denaturation leads to bond disruption, and the molecule turns into liquid.
Denaturation is always irreversible.
Final folded structure can reveal the steps of protein folding.
Some proteins form a complete 3-D structure only when they interact with their targets.
Which parts of the amino acids AA1 and AA2 are involved in the formation of a peptide bond?
AA1–AA2
side chains of both AA1 and AA2
carboxyl group of AA1 and side chain of AA2
amino group of AA1 and carboxyl group of AA2
carboxyl group of AA1 and amino group of AA2
carboxyl group of AA1 and amino group of AA2
Which of the following statements is correct regarding starch and cellulose?
They are used for energy storage in plants and animals.
They are polymers of glucose.
They are cis and trans isomers of each other.
They are structural components of the plant cell wall.
They are polymers of glucose.
Which of the following are possible base compositions for double-stranded DNA?
%A %G %C %T %U
5 45 45 5 0
20 20 20 20 20
All of the above
35 15 35 15 0
5 45 45 5 0
Which of the following terms does not describe a molecule of glucose?
a disaccharide
an isomer of fructose
an aldose
a hexose
a disaccharide
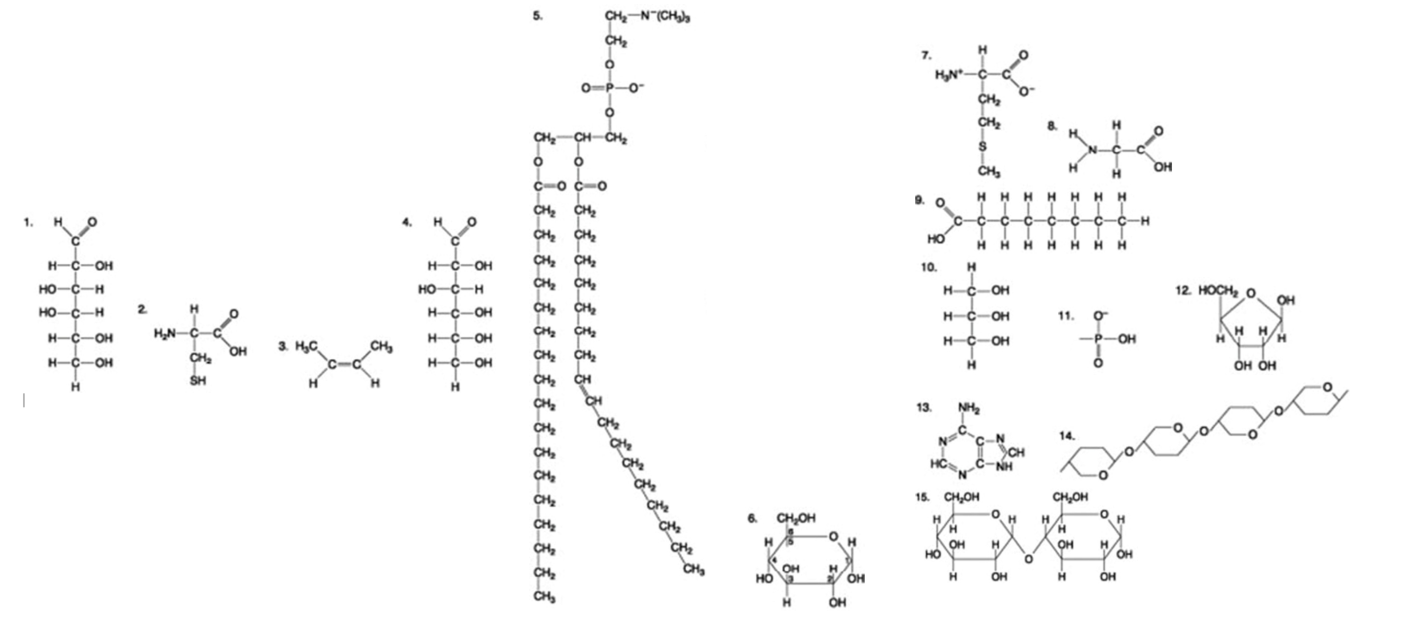
The following question is based on the 15 molecules illustrated in the figures.
A fat (or triacylglycerol) would be formed as a result of a dehydration reaction between ________.
one molecule of 5 and three molecules of 9
one molecule of 5 and three molecules of 10
one molecule of 9 and three molecules of 10
three molecules of 9 and one molecule of 10
three molecules of 9 and one molecule of 10
If one strand of a DNA molecule has the sequence of bases 5′-ATTGCA-3′, the mRNA synthesized following the template will be ________.
5′-TGCAAT-3′
5′-TAACGT-3′
5′-UGCAAU-3′
3′-UAACGU-5′
3′-UAACGU-5′
During a 12-hour fast in a human, the first macromolecule to get depleted is typically ________.
glycogen
starch
glucose
protein
glycogen
Which of the following is a palindromic sequence?
5′-AGGTCC-3′
3′-TCCAGG-5′
5′-GAATCC-3′
3′-CTTAGG-5′
5′-CCTTCC-3′
3′-GCAAGG-5′
5′-GGATCC-3′
3′-CCTAGG-5′
5′-GGATCC-3′
3′-CCTAGG-5′
How many amino acids and peptide bonds does a tetrapeptide have?
three amino acids and four peptide bonds
four amino acids and two peptide bonds
four amino acids and three peptide bonds
four amino acids and four peptide bonds
four amino acids and three peptide bonds
The "flow of genetic information" in molecular biology follows the following path, ________.
DNA is transcribed into RNA, which is translated into protein
DNA is translated into RNA, which is transcribed into protein
RNA is transcribed into protein
DNA is translated into protein
DNA is transcribed into RNA, which is translated into protein
A new organism is discovered in the forests of Costa Rica. Scientists there determine that the polypeptide sequence of hemoglobin from the new organism has 72 amino acid differences from humans, 65 differences from a gibbon, 49 differences from a rat, and 5 differences from a frog. These data suggest that the new organism is more closely related to ________.
humans than to frogs
gibbons than to rats
rats than to frogs
frogs than to humans
frogs than to humans
Which of the following best describes glycogen?
is formed by the hydrolysis of glucose molecules.
a storage polysaccharide found in plants
a fibrous protein that helps in maintaining cellular structure
a storage polysaccharide found in animals.
a storage polysaccharide found in animals.
People who are lactose intolerant cannot extract energy from milk because ________.
they are missing the bacteria that can digest lactose
an enzyme is not functioning correctly
lactose is too big to be digested by the enzymes
milk is fermented to a by-product, which cannot be digested
an enzyme is not functioning correctly
Homo sapiens have 23 pairs of chromosomes. What does the number '23' represent here?
23 double-stranded DNA molecules in each somatic cell
several hundreds of genes on DNA but not on the chromosomes
23 single-stranded DNA molecules in each somatic cell
46 double-stranded DNA molecules in each somatic cell
46 double-stranded DNA molecules in each somatic cell
What is the role of hydrogen in the synthesis of "hardened fats" from vegetable oil?
Hydrogen causes cross-linking between chains.
Hydrogen causes hydrolysis to occur.
Hydrogen increases the carbon chain length.
Hydrogen reduces the number of carbon-carbon double bond.
Hydrogen reduces the number of carbon-carbon double bond.
The "flow of genetic information" in molecular biology follows the following path, ________.
DNA is transcribed into RNA, which is translated into protein
RNA is transcribed into protein
DNA is translated into protein
DNA is translated into RNA, which is transcribed into protein
DNA is transcribed into RNA, which is translated into protein
Which of the following statements about the 5′ end of a polynucleotide strand of RNA is correct?
It has a carboxyl group attached to the number 5 carbon of ribose.
It has phosphate attached to the number 5 carbon of the nitrogenous base.
It has a hydroxyl group attached to the number 5 carbon of ribose.
It has a phosphate group attached to the number 5 carbon of ribose.
It has a phosphate group attached to the number 5 carbon of ribose.
An amino acid is to a protein as a glucose molecule is to ________.
a lipid
a tripeptide
nucleic acid
glycogen
glycogen
The difference between an aldose sugar and a ketose sugar is ________.
the position of the carbonyl group
the number of carbon atoms
the position of the hydroxyl groups
the ring form and the linear chain
the position of the carbonyl group
If 14C-labeled uracil is added to the growth medium of cells, what macromolecules will be labeled?
RNA
polypeptides
both DNA and RNA
DNA
RNA
Which of the following ratios of nucleotides is constant for DNA?
A + U = G + C
A + C = U + G
A + G = T + C
A + T = G + C
A + G = T + C
Homo sapiens have 23 pairs of chromosomes. What does the number '23' represent here?
several hundreds of genes on DNA but not on the chromosomes
23 single-stranded DNA molecules in each somatic cell
23 double-stranded DNA molecules in each somatic cell
46 double-stranded DNA molecules in each somatic cell
46 double-stranded DNA molecules in each somatic cell
Which of the following features of cooking oil and gasoline (hydrocarbons) identifies why the molecules are NOT amphipathic?
They do not have a polar or charged region.
They have hydrophobic and hydrophilic regions.
They are highly reduced molecules.
They do not have a nonpolar region.
They do not have a polar or charged region.
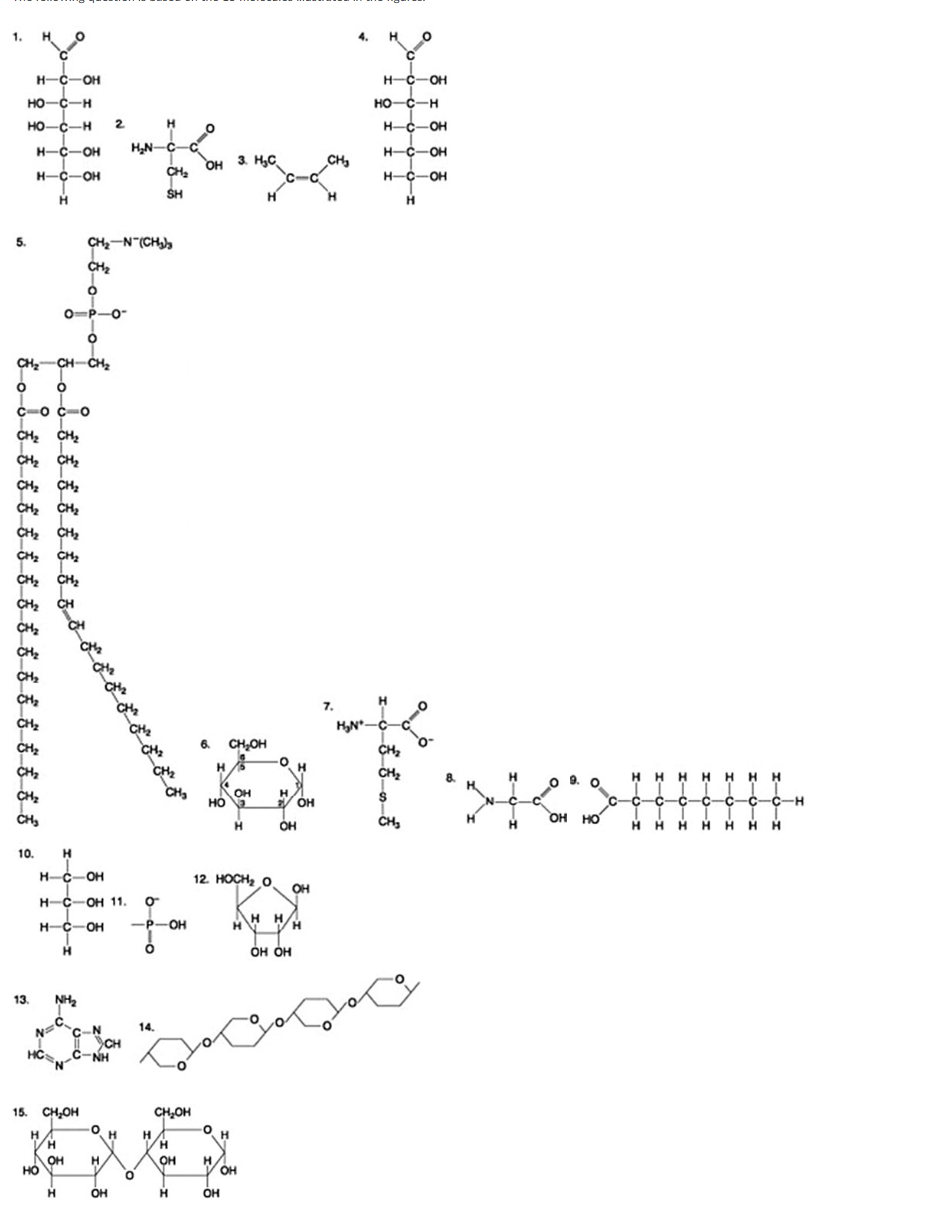
The following question is based on the 15 molecules illustrated in the figures.
Which of the following pairs of molecules could be joined together by a peptide bond in a dehydration reaction?
2 and 3
12 and 13
8 and 9
7 and 8
7 and 8
Which part of an amino acid gives it its unique identity?
the long carbon-hydrogen tails of the molecule
the carboxyl and amino groups
the glycerol molecule that forms the backbone of the amino acid
the components of its side chain
the components of its side chain
Extraction of lipids from a tissue sample requires organic solvent because ________.
organic solvents are easily accessible
organic solvents are cheaper
organic solvents keep the temperature low and manageable
lipids are hydrophobic, and they can only be solubilized in organic solvent
lipids are hydrophobic, and they can only be solubilized in organic solvent
Which polysaccharide is an important component in the structure of fungal cells?
chitin
amylopectin
amylose
cellulose
chitin
When nucleotides polymerize to form a nucleic acid, ________.
covalent bonds form between the bases of two nucleotides
a hydrogen bond forms between the sugar of one nucleotide and the phosphate of a second
a covalent bond forms between the sugar of one nucleotide and the phosphate of a second
hydrogen bonds form between the bases of two nucleotides
a covalent bond forms between the sugar of one nucleotide and the phosphate of a second
Which of the following is not analogous to a biological polymer?
a train
an iron chain
a pearl strand necklace
a truck
a truck
Which of the following has been associated with atherosclerosis and should be eliminated from one's diet for health reasons?
olive oil
butter
liver and kidney organ meat
butter, liver, and kidney organ meat
butter, liver, and kidney organ meat
Which of the following can be used to predict the 3-D structure of the protein?
number of water molecules in the vicinity
side chains of various amino acids
sequence of amino acids in the polypeptide chain
peptide bonds between different amino acid
sequence of amino acids in the polypeptide chain
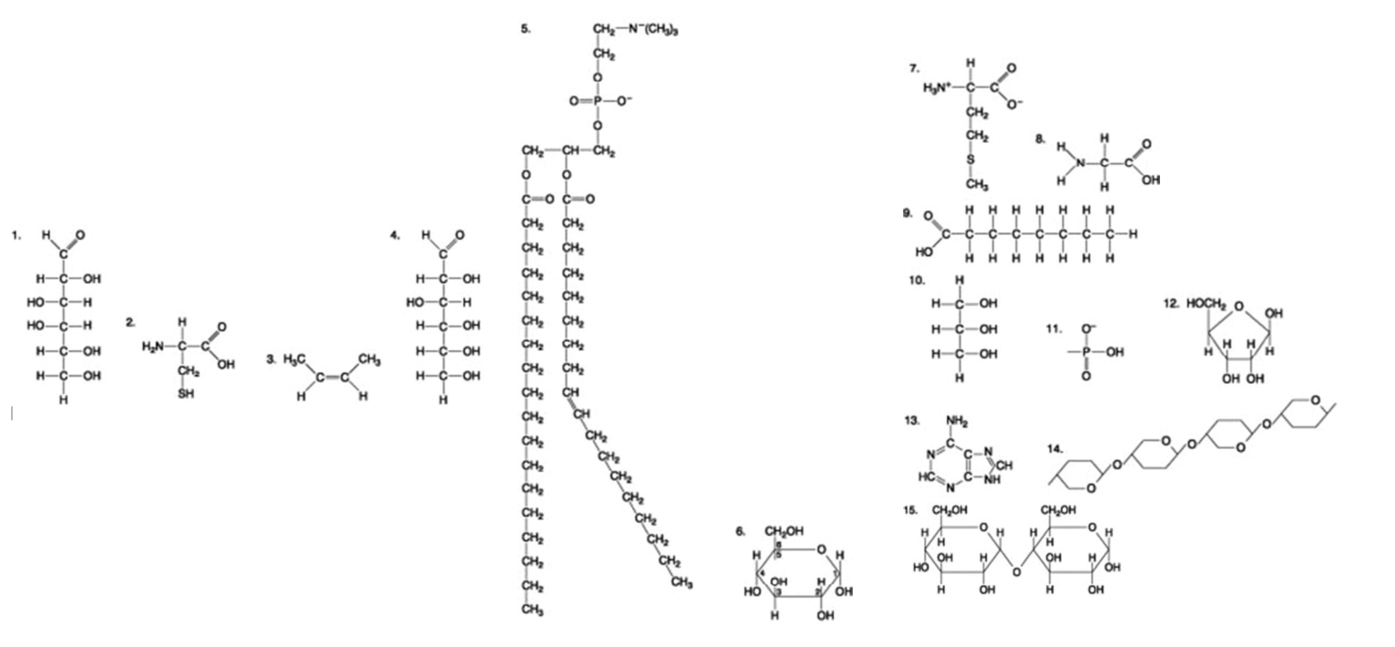
The following question is based on the 15 molecules illustrated in the figures.
Which molecule is a saturated fatty acid?
9
8
5
1
9