study guide
1/128
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
129 Terms
what does antiboidy look like
what are antibodies
seceretd form of B-cell receptr
clears extracellular pathogens + toxins
what are immunoglobulins
cell surface b-cell antigen receptor and secreted antibody
what do antibodies do
bind pathogens
, which leads to inactivate or destory them
humoral immune response
immune response using antibodies
antibodies secreted by B cells
what are effector cells
short-lived activated immune cells that carry out the response
fights infection
effector cell examples
monocyte
lymphocytes
neutrophils
eosinopihls
basophils
macrophaes
erhtocytes
plaeltles
what are antigens
any substance recogniced by b or t lymphocytes
what is immunity
protection agsint foreign pathogens or substances
what are common types of antigens
proteins
glyocporteins
polysachs
nickel
drugs
chemicals
what cells recognize antigens
b and t cells
b cell function
make antibodies
t cell function
interact with other cells, maybe add more
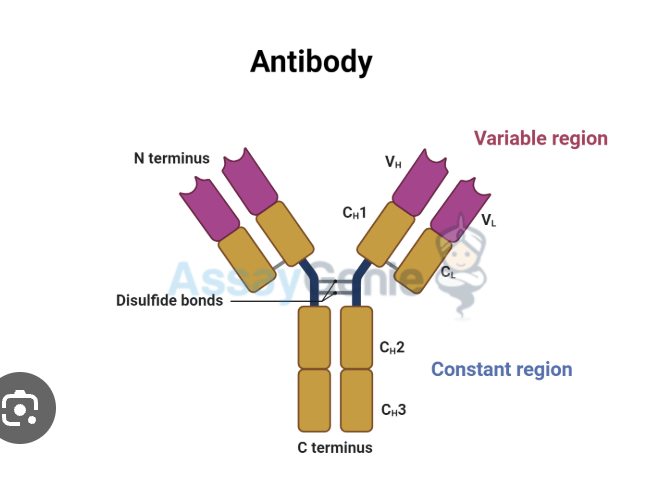
antibody structure basic
2 heavy chains+ 2 light chains
quatnery protein
heavy vs light chain
heavy: n-term + c-term
light: kappa or lambda
both have variable and constant regions
variable domains
vh heavy
Vl (varibalelight
what do variable regions do
form antigen-bindign site
determine speciificty
Hypervariable regions (CDRs)
3 regions: HV1, HV2, HV3
Also called CDR1, CDR2, CDR3
Directly bind antigen
antigen-binding site
Formed by VH + VL
Each antibody has 2 identical binding sites
Can bind to pockets, grooves, surfaces
vary in shpe and physcial poreoties
constant region function
determiens effector function
control interaction with immune cells
Fab fragment function
“Fragment antigen binding”
Binds directly to antigen
has variable regions
Fc fragment function
“Fragment crystallizable”
Interacts with immune cells
Triggers immune response
determins effector function
disulfide bonds
Hold chains together
Stabilize antibody structure
2 heavy + 2 light → variable binds antigen (CDRs)
constant= immune action then fab binds and FC signals
primary portien
amino acid seqeunce (peptide bonds)
secondary
alpha /beta sheet folding local
tertiary
3d shape of one chain (functional shape)
quat
multiple chain (final protein)
IgG
monomer
most abunodna in antibodies
blood lymph intestine
crosses placenta,
neutralied toxins
triggers compleet+ phag
protects the fetus
IgM
penatmer (5 mono units+ a J chain)
6% of antiboides
stays in blood
first eposnse in infection
short lived
causes clump (aggulutination)
IgD
monomer
0.02
b cells and blood
activates b cells, assists in immune reposne
IgA
monomer for blood, and dimer for secretions
13
mucus, saliva, tears, breast milk
protects mucuols surfaces which prevents pathogen attachment
IgE
monomer
0.002
mast cells and basophils
causes histomaine release
fights worms
how do immungolgobls differ
different heavy chains
which antibidy neturlizes pathogens
igG
why is igG special
its the most abudnanct
inly one thst crosses placenta
provides long-term immunity
protects fetus
what are monoclonal antiboides
from one clone of b cels
all the same same speicifcity identicl
how are monoclonala tnbodies made
fusion b cells + myeloma cells
peg gell
forms hyrbdioma
hyrbidoma
b cell and myleoma cell
makes antiboidei and lives forever
HAT medium
selection media made to select hybdrioma cells
use of monoclonal antibodies
treat cancer
arhtitis
and target specific atngiens
what are immunogloilbins
b cell receptors + seceretd antibodies
10^11 types
what creates antiboidy diveristy
vdj recomib
juncitnal diversty
somatic hypermutation
junctional diversity
Addition/removal of nucleotides
Happens during gene rearrangement
Increases variability
Somatic hypermutation
Mutations in mature B cells
Affects variable (V) regions only
Improves antigen binding
Germline configuration
Ig genes exist as separate segments before rearrangement
Not yet functional
Gene basics
Gene = DNA → protein/RNA
Exons = coding
Introns = noncoding
Central dogma
DNA → (transcription) → pre-mRNA
pre-mRNA → (splicing) → mRNA
Introns removed, exons joined
locus
Physical location of a gene on a chromosome
TP53 gene
Controls damaged DNA (repair, stop, death)
11 exons + 10 introns
Order of Ig gene segments
Heavy chain:
V → D → J → C
Light chain:
V → J → C (no D segment)
Front: Where are Ig genes located?
Heavy chain → chromosome 14
Light chains → chromosome 2 (κ) & chromosome 22 (λ)
germline organization
Ig genes exist as separate segments in order on chromosome
Called germline configuration
what is vdj recom
Rearrangement of gene segments in B cells
Creates antibody diversity
What controls V(D)J recombination?
Recombination signal sequences (RSS)
Recognized by RAG1 & RAG
role of rag1 and rag2
Cut DNA at RSS
Bring segments together
Enable joining of V, D, J
Heavy chain recombination steps
D + J gene segments join
V gene segments joins DJ sequence → forms VH exon
heavy chain C region enocded by exons, the c region exons are joined to VH exon by spolicing of heavy chain RNA
c lleader peptide directs protien inot the cell’s secretor pathways and is then cleaved
maybe make better
heavy chain srtucture
V region = V + D + J
C region = multiple exons
Leader (L) → directs secretion
light chain recomb
v and j gene segment in genomic dna formed to compelte variable light chain exon
light chain C is enocded into seperate exons and is joined to Vl by splicing of light chain RRNA
leader peptide directs portien onto the cells seceteaory pathway and cleaved
What creates diversity in Ig genes?
Random recombination of V, D, J
Different numbers of segments (polymorphism)
isotype switching
Changes C region only
Keeps same antigen specificity
t cell receptor
membrane bound has v and c domains
recognzied peptides and mhc complex not the antigen alone
can t cells recgonzie free antigens
no, only antigens bound to mhc
what is MHC
cell surface glycoprotein
displays antigen peptides
where does the peptide on MHC come from
from pathogens
mhc classes
1: most cells
2: only APC, antigen rpesentiv cells
whats are apc
Cells that process + present antigen to T cells
Include: dendritic cells, macrophages, B cells, Langerhans cells
denditic cells
macorphage
b cells
mhc.1 func
Presents intracellular (inside cell) antigens
Recognized by CD8 T cells
MHC II function
Presents extracellular antigens
Recognized by CD4 T cells
peptide binding site on MHC
Peptide-binding cleft (groove)
Steps of MHC I antigen presentation
Protein in cytosol → broken into peptides (proteasome)
Peptides transported into ER (TAP)
MHC I assembled + loaded with peptide
Goes to surface → recognized by CD8
proteasome function
Breaks intracellular proteins → peptides
What helps MHC I assembly?
Calnexin, calreticulin, ERp57, tapasin
Form peptide-loading complex
MHC II antigen processing
Extracellular proteins → taken into vesicles
Broken down in acidic vesicles
Loaded onto MHC II
Presented to CD4
eptiope
part of antigen that antiboidy binds
multivalent antigen
Has multiple epitopes
Or repeated copies of same epitope
types of epitopes
Linear: continuous amino acids
Discontinuous: brought together by protein folding
plasma b cell function
secrets antibodies
highyl specifilaied effector B cells
b cell devlops where
bone marrow
order of iG geen rearrngment
heavy chain first then light chain
Early B-cell development steps
occurs in bone marrow
stem cells differinaite into pro-b cell
d and g gene segment come together , then v segents joins DJ
a u heavy chain is then made
pre-b cell receptor forms
light chain then rearranges
IgM is expressed and diffeirnates into an immature B cell
can you check
pre b cell receptor
wuality contorl checkpoint
checks is u heavy chain works
imature b cell
expression of IgM on surface
negatuve selection
removes B cells that binds self-antigens
prevents automminuty
positive selection
b cells compete for suvival
in seocndary kympohid irgans
where do t cells develop
thymus
immature= thymocytes
Structure of thymus
Cortex (outer): immature T cells desne
Medulla (inner): mature T cells, less dense
Supported by thymic stroma (epithelial cells)
thymus involution
thymus shrisk with age
What do macrophages do in thymus?
Remove dead/self-reactive T cells (cleanup)
Pre-T cell receptor function
Tests TCR formation
Ensures proper development (quality control)
T cell stages (CD markers)
Double negative: no CD4/CD8
Double positive: CD4 + CD8
Single positive: either CD4 OR CD8
postiive selection
in cortext
keeps t cells that recognize self -mhc
negative selection
Removes T cells that bind self-antigens
Prevents autoimmunity
t cells become
cd4 helper
cd8 cytotoxic
Central vs Peripheral tolerance
Central: in thymus/bone marrow (development stage)
Peripheral: outside (after development)
Which MHC do APCs express
MHC II (main) → activates CD4 T cells
Also have MHC I
Why are dendritic cells important?
Main activators of naïve T cells
Link innate → adaptive immunity
Immature vs mature dendritic cells
Immature: in tissues, capture antigen
Mature: in lymph nodes, activate T cells
Where do naïve T cells encounter antigen?
Lymph nodes (secondary lymphoid organs)
Enter via HEV (high endothelial venules)
Signals required to activate naïve T cells
Signal 1: TCR + CD4/CD8 binds peptide-MHC
Signal 2: CD28 binds B7 (co-stimulation)
BOTH required