BIOC 4331 Lecture 9
1/63
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
What is oxidation?
Loss of electrons

What is reduction?
Gain of electrons

Do oxidation and reduction occur independently?
No — they always occur simultaneously as a redox reaction.

What is an oxidant?
The species that causes oxidation and is itself reduced (electron acceptor).

What is a reductant?
The species that causes reduction and is itself oxidized (electron donor).

What is the ultimate source of biological energy generation?
Movement (flow) of electrons.
What is electromotive force (emf) in biological redox reactions?
The energy released from electron transfer between species with different electron affinities, which can be used to perform work.
Foods such as glucose are considered what type of compounds in redox chemistry?
Reduced compounds
Why is electron flow from glucose to oxygen exergonic?
Because O2 has a higher affinity for electrons than the electron-carrier intermediates.
What type of energy is stored in the mitochondrial proton gradient?
Potential energy
What is the proton-motive force?
The potential energy stored in a proton gradient across a membrane.
How can an overall redox reaction be represented?
As two half reactions: one oxidation and one reduction.
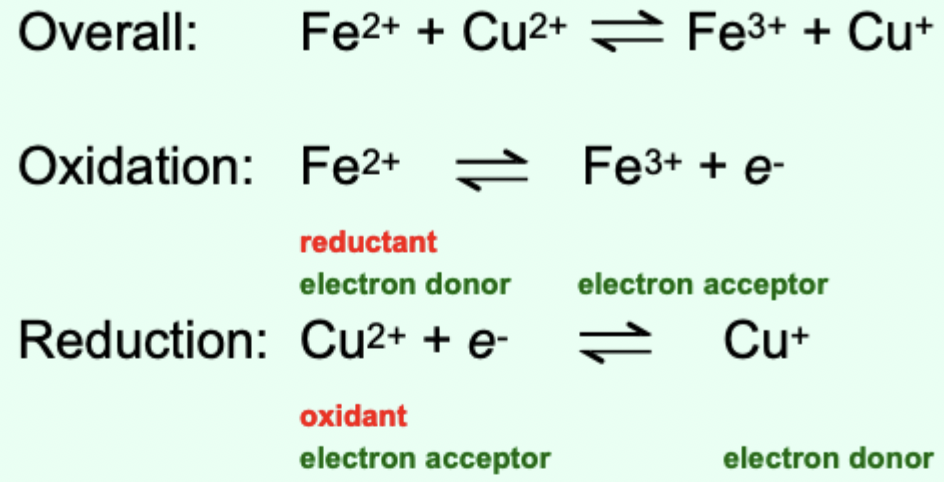
What is a conjugate redox pair?
The electron donor and electron acceptor involved in a half-reaction.
What is the order of increasing electronegativity relevant for carbon oxidation states?
H < C < S < N < O
What is the atomic number of carbon, and why is it tetravalent?
Carbon has an atomic number of 6 and is tetravalent because it has 4 valence electrons and can form four covalent bonds.

Why can carbon form four covalent bonds?
Its valence shell contains 4 electrons, allowing four shared electron pairs.
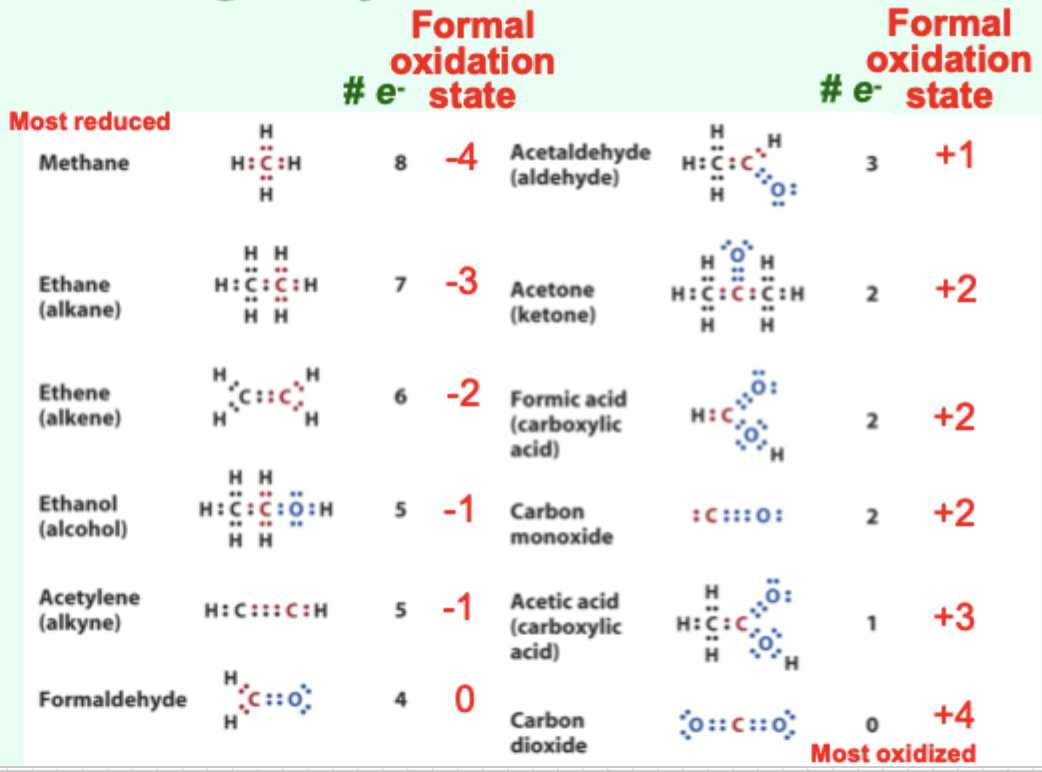
What does the formal oxidation state of carbon represent?
The formal number of electrons carbon “owns“ based on electronegativity.
How does bonding carbon to oxygen affect oxidation state?
It increases carbon’s oxidation state (carbon is oxidized).
How does bonding carbon to hydrogen affect oxidation state?
It decreases carbon’s oxidation state (carbon is reduced).
In biological systems, what does oxidation of an organic substrate usually involve?
Loss of electrons, often occurring concurrently with loss of hydrogen.
Why does loss of hydrogen usually indicate oxidation in biochemistry?
Because hydrogen carries electrons; losing hydrogen corresponds to loss of electrons.
What does direct addition of oxygen atoms to an organic substrate indicate?
Loss of electrons and oxidation.
In biological systems, electrons are transferred between what two types of molecules?
From an electron donor to an electron acceptor.
What are the different ways electrons be transferred in biological systems?
Directly as electrons
As hydrogen atoms
Directly as hydride ions
Through direct combination with oxygen
What does a hydrogen atom consist of?
One proton (H+) and one electron (e-).

What does it mean when electrons are transferred as hydrogen atoms?
Both a proton and an electron are transferred together.
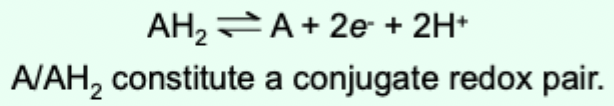
What is a hydride ion (H-)?
A hydrogen species that carries two electrons.
How are electrons transferred in hydride ion transfer?
Transferring two electrons simultaneously.
Hydride ion transfer occurs in which type of biochemical reactions?
NAD-linked oxidation–reduction reactions.
What does electron transfer through direct combination with oxygen mean?
Oxygen combines with an organic reductant and is covalently incorporated into the product.
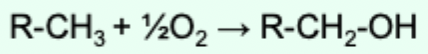
Why does direct combination with oxygen indicate oxidation?
Because oxygen is highly electronegative and causes loss of electrons from the organic substrate.
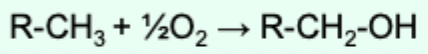
What is a reducing equivalent?
A single electron equivalent in a redox reaction, whether transferred as an e-, H atom, hydride ion, or oxygen.
What is the reduction potential (E)?
A measure (in volts) of the affinity of an electron acceptor for electrons in a conjugate redox pair.
What does a higher (more positive) reduction potential indicate?
Greater affinity for electrons.
What is the biochemical standard reduction potential (E°′)?
The reduction potential of a conjugate redox pair measured under biochemical standard conditions.
What are the biochemical standard conditions for measuring E°′?
55 M water
1 M each solute
101.3 kPa each gas
pH 7
298 K
In a redox reaction, electrons flow between half-cells in what direction?
From the half-cell with the lower E°′ to the half-cell with the higher E°′.
Which half-cell has the greater affinity for electrons?
The half-cell with the more positive E°′ value.
What does a more positive E′° value indicate?
Greater affinity for electrons.
How is ΔE′° calculated for a redox reaction?
ΔE′° = E′°(electron acceptor) − E′°(electron donor)

What does a positive ΔE′° indicate?
Electron flow is favorable (spontaneous).
What is the relationship between ΔG′° and ΔE′°?
ΔG′° = −nFΔE′°
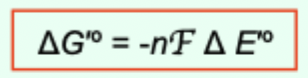
In the equation ΔG°′ = −nFΔE°′, what does n represent?
The number of electrons transferred in the reaction.
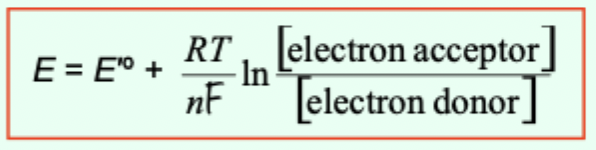
What does the Nernst equation describe?
How the actual reduction potential depends on the concentrations of participating species.
Why does biological oxidation of glucose occur in stages rather than one step?
To release free energy in manageable amounts that can be efficiently captured as ATP.
What is a coenzyme?
A low molecular weight compound required for catalytic activity.
What are the two universal sets of coenzymes that undergo reversible oxidation–reduction?
NAD+ / NADP+ and FMN / FAD.
What are the major non-NAD/FAD specialized electron carriers in biological systems?
Lipid-soluble quinones: membrane-embedded electron carriers
Iron–sulfur proteins: transfer electrons via iron–sulfur clusters
Cytochromes: electron transfer via heme groups
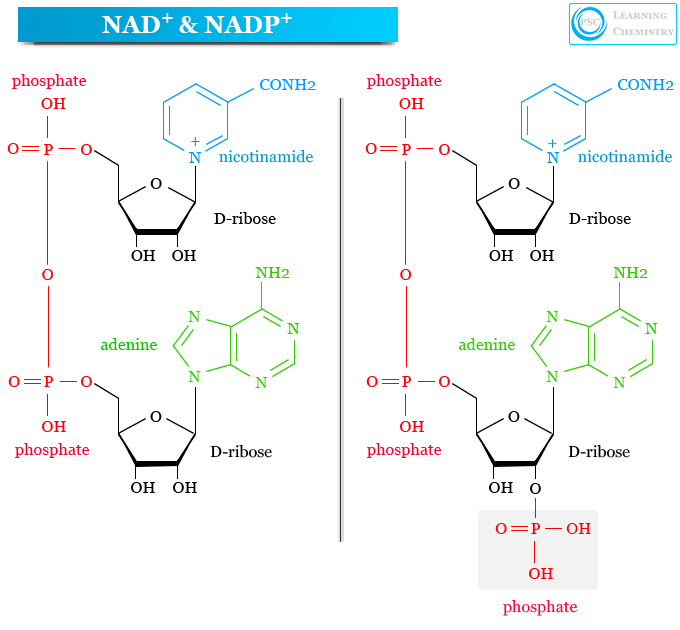
What do NAD⁺ and NADP⁺ stand for, and how do they differ?
NAD+: nicotinamide adenine dinucleotide
NADP+: phosphorylated form of NAD⁺ (extra phosphate on the ribose)
What structural feature distinguishes NADP⁺ from NAD⁺?
NADP+ has an additional phosphate group esterified to a ribose hydroxyl group; NAD+ does not.
What is the difference between oxidized and reduced forms of NAD(P)?
Oxidized: NAD⁺ / NADP⁺
Reduced: NADH / NADPH
Reduction adds a hydride (2 electrons + 1 proton).
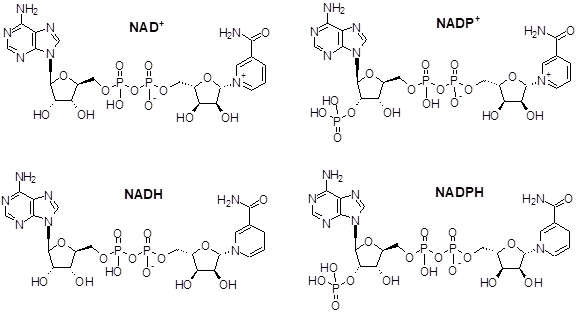
What vitamin are NAD⁺ and NADP⁺ synthesized from?
Niacin (vitamin B₃)
How does reduction of NAD⁺ or NADP⁺ occur?
Reduction occurs via hydride ion (H⁻) transfer (two e- simultaneously).
Do NAD⁺ and NADP⁺ remain bound to enzymes after a reaction?
No. NAD⁺ and NADP⁺ dissociate from the enzyme after each reaction cycle.
What is the primary metabolic role of NAD⁺?
Generally functions in catabolism, driving the oxidative breakdown of complex organic compounds.
Where are NAD⁺ and NADH predominantly located in the cell?
In the mitochondria
What is the primary metabolic role of NADPH?
Generally functions in anabolism, supplying reducing power for biosynthesis of complex biological molecules.
Where are NADP⁺ and NADPH predominantly located in the cell?
In the cytosol
What do FAD and FMN stand for?
FAD = flavin adenine dinucleotide
FMN = flavin mononucleotide
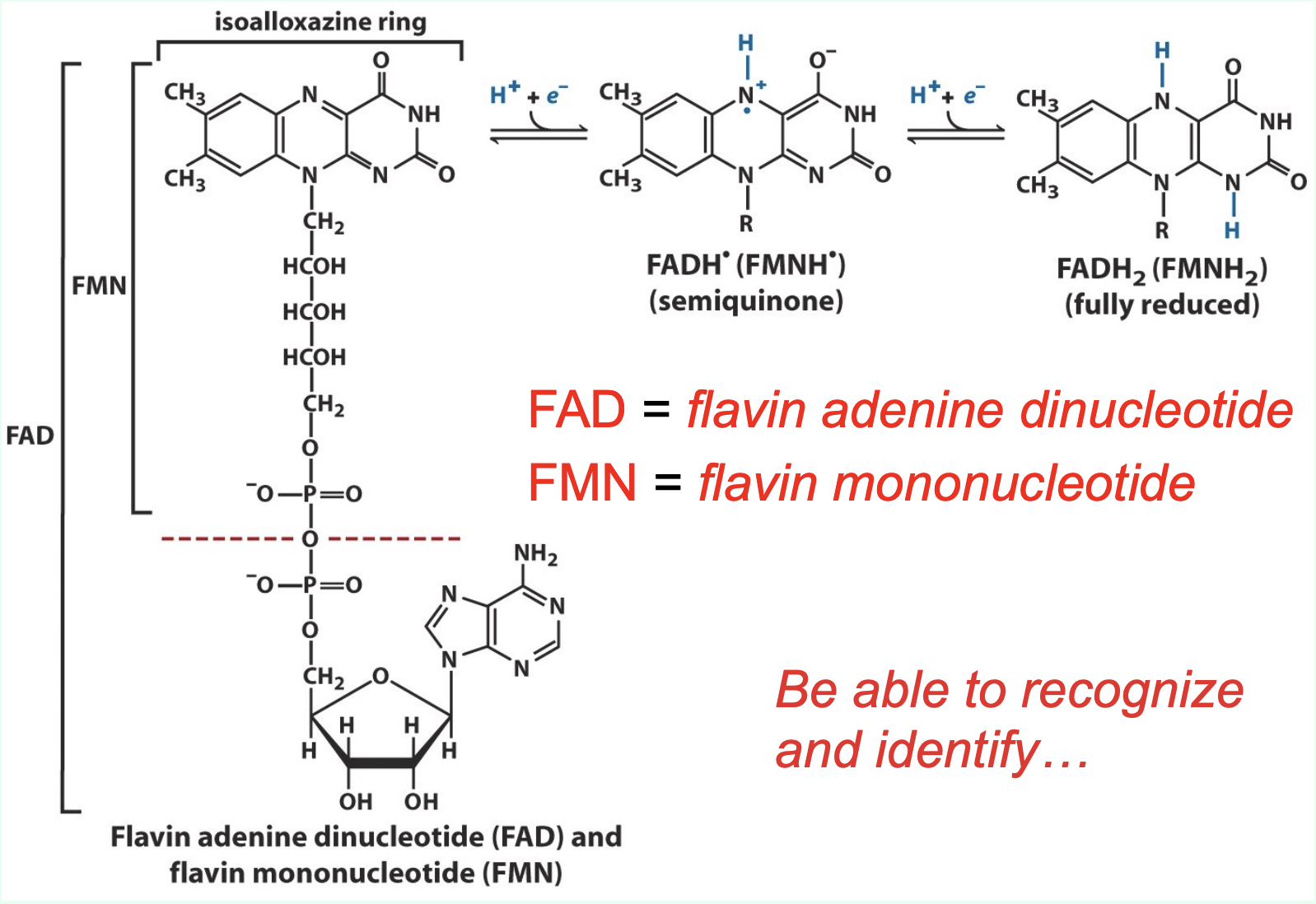
What key structural feature allows you to recognize FAD and FMN?
Both contain an isoalloxazine (flavin) ring, which is the redox-active portion of the molecule.
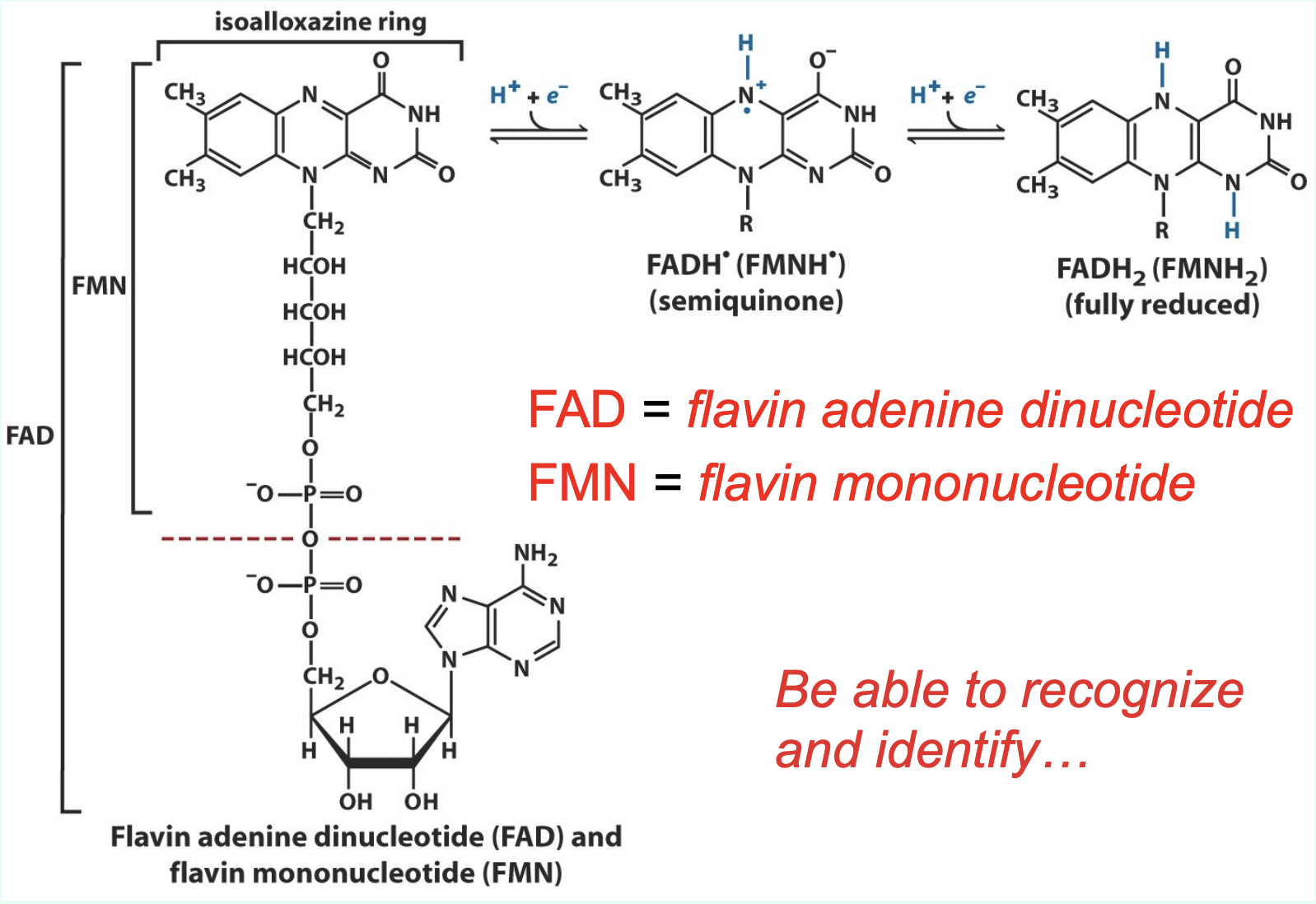
How are FAD and FMN associated with enzymes?
FAD and FMN are tightly bound coenzymes of flavoproteins and act as prosthetic groups (do not dissociate between redox cycles).
What is a key difference between NAD(P)⁺ and FAD/FMN as electron carriers?
NAD(P)+: soluble, dissociates after each reaction
FAD/FMN: tightly bound prosthetic groups, remain attached to enzymes