06. Imports and Exports, Domestic and International : IATA. USDA. CDC guidelines
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
Which 2 laws identify standards for humane care during transportation?
1. AWA
-standards for animal care during transport
2.Lacey Act
-provides that wildlife be transported IAW IATA Live Animal Regs
Lacey Act (1900) authorizes ____ to regulate
Authorizes FWS to regulate
-Importation, exportation, transportation, sale, receipt, acquisition, or purchase of
-dead and live wild animals and their tissues and products imported into or exported from the United States
AWR- CFR, Title 9, Chapter 1 authorizes USDA -- ______ and ______(NCIE)
APHIS Veterinary Services and National Center for Import and Export (NCIE)
______ permits are required for receiver importing material derived from any animal potentially subject to USDA regulation
USDA permits
Does USDA permits cover importation of live lab animals?
The USDA has no regulatory authority of importing live lab animals that have NOT been inoculated with or exposed to livestock or poultry disease agents exotic to the United States. You must also make sure they are not coming from a facility that work using exotic disease agents are being used.
What doesn't USDA import permit cover?
This does not include; Primates, dogs, cats, livestock*, poultry, hedgehogs, tenrecs, minipigs, monoclonal antibodies, hybridomas, cell lines, and material for commercial purposes.
What does USDA import permit cover?
This also includes: Transgenic/knock-out mice and rats, hamsters, gerbils, guinea pigs, rabbits, ferrets, and their blood, tissue, DNA, extracts, antibodies, feces, sera, and antisera for research purposes. (blood, sera, antibodies, and antisera is limited to less than 1 liter)
Materials which require a permit include, animal tissues, blood, cells or cell lines of livestock or poultry origin, RNA/DNA extracts, hormones, enzymes, monoclonal antibodies for IN VIVO use in non-human species, certain polyclonal antibodies, antisera, bulk shipments of test kit reagents, and microorganisms including bacteria, viruses, protozoa, and fungi.
Various other animal materials which require a permit include dairy products (except butter and cheese), and meat products (e.g., meat pies, prepared foods) from countries with livestock diseases exotic to the U.S.
USDA __________ REGULATE the interstate movement of COVID-19 isolates.
USDA DOES NOT REGULATE the interstate movement of COVID-19 isolates.
USDA- Dogs for imported commercial Sale, what paper work needs to accompany the dog?
Health and rabies certificate
Import permit issued by APHIS
USDA- Dogs for Commercial Sale Coming from Countries affected with African Swine Fever, what additional requirements are needed?
1. no dirt or wooden shave bedding
2. dispose all bedding/ pad upon entry into US
3. microchip
4. animal to be bathed within 2 days of entry
USDA Health Certificate - Form _____
Form 7001
How long is the USDA quarantine period for poultry/swine and ruminants
30 days and 30 days
Carriers cannot accept any live animals for shipment greater than ____ hours before departure but can be extended to ____ hours if both shipper and carrier agree.
4hrs; 6 hrs
AWR delivery - Carrier/handler must attempt to contact the consignee (receiver) at least
Every 6 hours after arrival
AWR delivery - For NHP's and dogs/cats, the animal must be returned to the consignor if the consignee
Can't be reached within 24 hours or
Is reached but doesn't take delivery by 48 hours
Animals should be imported within _____ days after the proposed date of arrival stated in the import permit
Must be imported within 30 days after the proposed date of arrival stated in the import permit
No more than ______ live dogs or cats, 8 weeks of age or older, that are of comparable size, may be transported in the same primary enclosure (ground transport, boat, private jet)
four
CDC - PHS import - Dogs rabies vaccine: after ___ months old and >____ weeks prior to travel; Dogs must have rabies vaccine _____ days prior to entry UNLESS they are < ____ mo, from a rabies free country for at least 6 months, or it will be taken straight to a research facility; what about cats?
after 3 months old and >4 weeks prior to travel
Dogs must have rabies vaccine 30+ days prior to entry UNLESS they are < 6mo, from a rabies free country for at least 6 months, or it will be taken straight to a research facility.
Cats no rabies vaccine requirement (state requirement)
What else is required for dogs from high-risk countries?
-Dogs must appear healthy upon arrival.
-Dogs must be at least 6 months old at time of entry or return to the U.S.
-Dogs must have a microchip
-The CDC Dog Import Form receipt
-A Certification of U.S.-issued Rabies Vaccination form or a USDA-endorsed export health certificate: These forms are filled out by a U.S. Department of Agriculture (USDA)-accredited veterinarian and endorsed by the USDA.
CDC - NHPs import: OWMs that die or euthanized during 31-day quarantine must be tested (Ag capture) for ____________
filovirus
CDC - NHPs import: NHP port entry ONLY at ________________, unless written approval is obtained in advance
CDC Quarantine Stations
CDC - NHPs import: During quarantine, all NHPs tested for ____ and monitored for _____ diseases
All NHPs tested for TB and monitored for zoonotic diseases
CDC - NHPs import: How often do institutions/individuals registered with the CDC to import NHP need to renew their registration?
Renew registration every 2 years
CDC - NHPs import: Importers must notify CDC within how many hour(s) of any illness or death of NHPs in quarantine facilities?
24 hrs
CDC - NHPs import: Importers must notify CDC at least how many days before importing a shipment of NHPs?
7 days
The Lacey Act authorizes which federal agency to regulate what activities?
a) CDC; Prohibit import, export, or interstate commerce of listed species
b) FWS; Transportation, importation, or sale or purchase of any fish, wildlife, or plant
c) PHS; Imported dogs and cats must be free of rabies
d) USDA; Transfer of toxins that have the potential to pose severe threat to animal health
b) FWS; Transportation, importation, or sale or purchase of any fish, wildlife, or plant
All of following apply to the CDC guidelines for importation of nonhuman primates EXCEPT?
a) Workers in direct contact with imported NHPs must wear, at minimum, a N95 respirator for respiratory protection
b) The minimum quarantine period for imported NHPs is 31 days
c) Animals with suspect positive TST reactions must remain in quarantine and receive at least 3 additional TSTs, administered 2 weeks apart
d) An importer must notify the CDC within 24 hours if any NHP tests positive for filovirus antigen or antibody
Must receive 5 addt'l TSTs
According to the Final Rule on Regulations for the Importation of Nonhuman Primates (42 CFR Part 71.53), which of the following is 𝐍𝐎𝐓 correct regarding quarantine of non-human primates (NHPs)?
a) All non-human primates that are imported from outside of the U.S. must be quarantined for at least 31 days after arrival at an HHS/CDC approved quarantine facility.
b) Any Old World NHPs that die or are euthanized during the 31-day quarantine period must be tested for filovirus antigen.
c) An importer registration certificate is valid for 3 years beginning from the date HHS/CDC issues the registration certificate.
d) An importer must contact CDC immediately (within 24 hours) by telephone, text, or e-mail to report any suspected zoonotic illness in an employee.
c. An importer registration certificate is valid for 3 years beginning from the date HHS/CDC issues the registration certificate. - 2 years
What does IATA stand for?
International Air Transport Association

What does IATA regulate?
-Global standard for transporting animals by commercial airlines
-Up-to-date airline and government requirements
-Requirements on handling, marking and labeling
Information on necessary documentation needed
-Classification of animal species and their container specifications
-Guidelines for non-air transport
What label wording and dimensions is required by IATA on the transport container?
At least one label must be attached
"Live Animals"
"Laboratory Animals"
Minimum dimensions:
10 x 15 cm label
Letter height
2.5cm (1 inch)
"This way up" labels
Must be placed on at least 2 opposite sides
Minimum dimensions:
7.4 x 10.5 cm
IATA- What are the three Health Status Classifications?
Conventional (unknown health status such as wild caught)
Conditioned Specific Pathogen Free (had testing, vaccine but not maintained in specialized/filtered housing such as dogs, cats and NHP)
Barrier raised SPF (rodent in ventilated housing)
BMBL - Appendix C: Transportation of infectious substances are regulated as hazardous materials by
-US Department of Transportation (DOT)
-Foreign governments
-International Civil Aviation Organization
BMBL - Category A Infectious Substance (UN 2814 / UN 2900) what is the different between the two ID number?
"UN2814" cause disease in humans or both humans and animals
"UN2900" cause disease in animals only
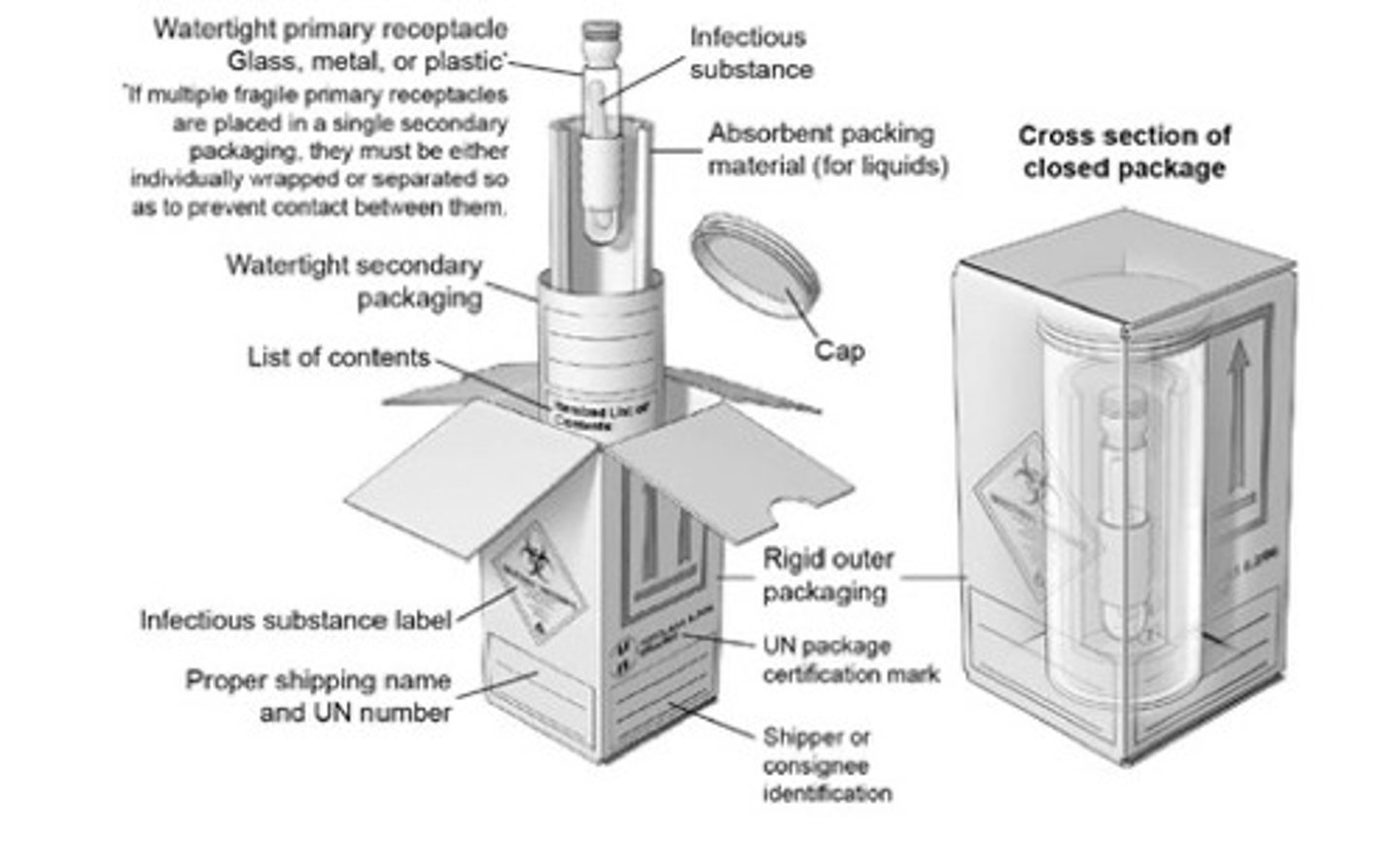
BMBL - Category A infectious substance packing requirement? ______package system
Triple packaging system
1. Watertight primary receptacle(s)
2. watertight secondary packaging (for liquids, secondary packaging must contain absorbent material to absorb all material); and rigid outer packaging. Each surface of the external dimension must be 100 mm (3.9 in) or more.
Completed packaging must pass specific performance testing
-Drop test and water-spray test
-Capable of withstanding an internal pressure producing a pressure differential of not less than 95 kPa (0.95 bar, 14 psi) and temps in the range of -40C to +55C (-40F to 131F).
BMBL - Category A infectious substance packing labeling requirement?
"Infectious substances, affecting humans, UN 2814" or
"Infectious substances, affecting animals, UN 2900"
Category A Packaging
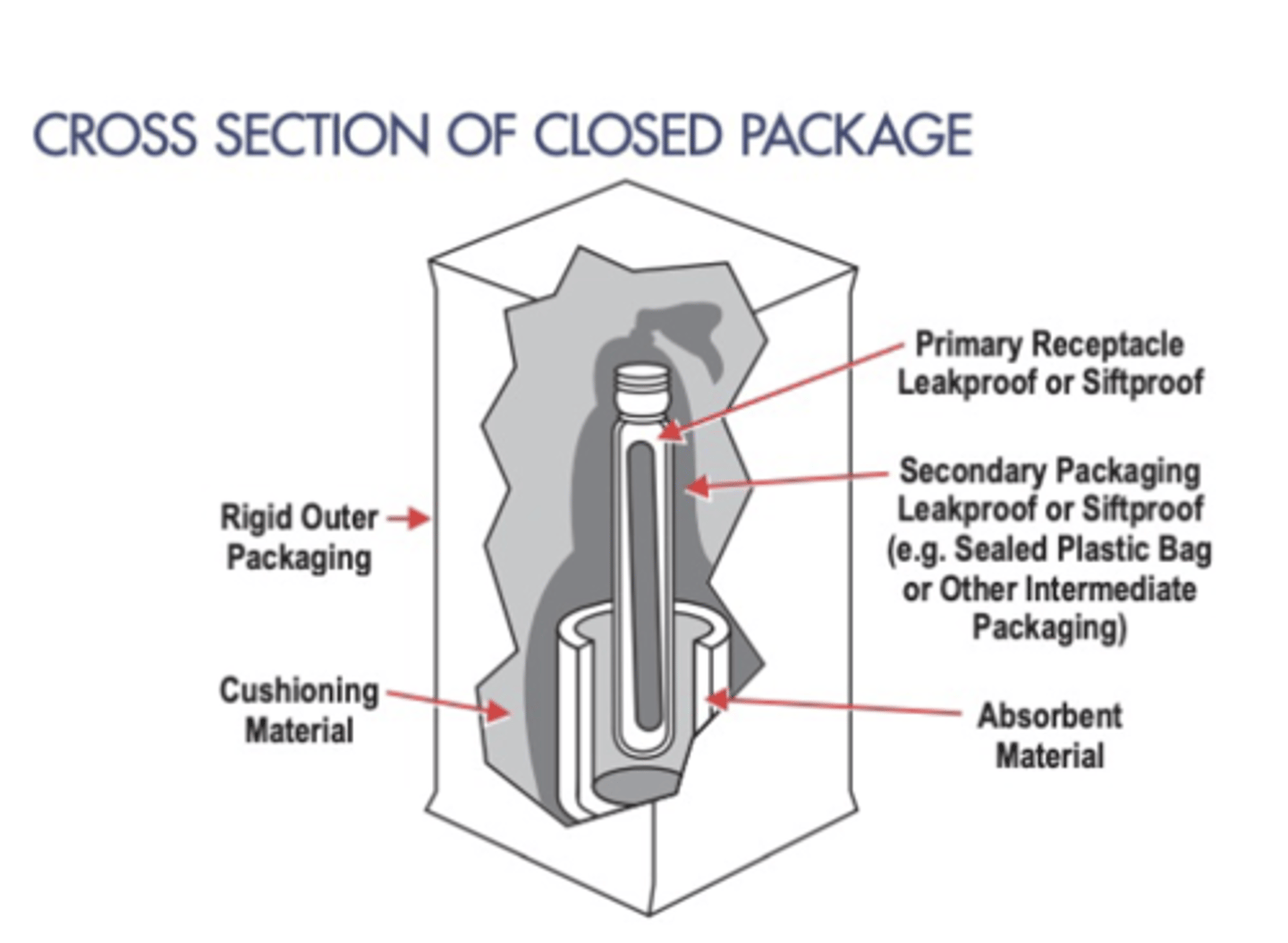
BMBL - Category B Biological specimen packing requirement? ______package system? UN _______
Triple packaging system for materials unknown or suspected of containing Cat B
Leak proof primary receptacle, leak proof secondary packaging (with absorbent material if liquids), and a rigid outer packaging. At least one surface of the outer packaging must have a minimum dimension of 100 mm by 100 mm (3.9 in).
The primary or secondary packaging must be able to withstand , without leaking, an internal pressure producing a pressure differential of 95 kPa.
The completed package must be able to pass a 1.2 m (3.9 feet) drop test.
UN 3733
Category B Packaging