Lab G: Reduction of Vanillin
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
17 Terms
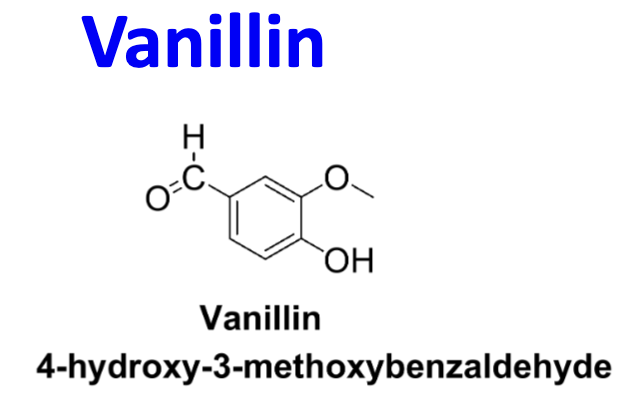
Structure of vanillin + name?
What are the pharmacological applications of vanillin?
Anticancer, antidiabetic
What is reduction at carbon?
Results in a decrease in # of C-Z bonds (Z = element more electronegative than carbon); usually C=O bonds.
OR
Results in an increase in # of C-H bonds.
What are reducing agents?
Provide the equivalent of 2 H atoms
What are the 3 types of reduction reactions?
1) Molecular H2
2) Dissolving metal reductions
3) Hydride reduction
What are the properties of the molecular H2 reaction?
Simplest reducing agent
Reductions carried out in the presence of a metal catalyst
What are the properties of the dissolving metal reductions reaction?
Using alkali metals (as a source of e^-1) and liquid NH3 (as a source of protons).
What are the properties of the hydride reaction?
Uses NaBH4 or LiAlH4 (delivers a H- ion)
Acidic workup (delivers an H+ ion)
What are hydride reducing agents?
Most useful for reduction of Aldehydes and Ketones
Contain a polar metal-hydrogen bond that serves as a source of the nucleophile hydride (H:-).
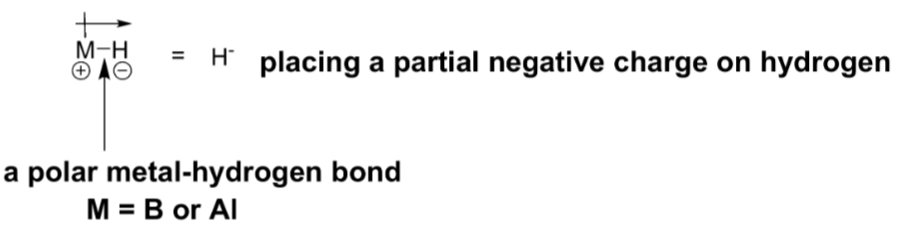
Give 2 examples of hydride reducing agents

Why is LiAlH4 a much stronger reducing agent than NaBH4?
Because the Al-H bond is more polar than the B-H bond.
What conditions must LiAlH4 be carried out in?
Under anhydrous reaction conditions - water reacts VIOLENTLY w/ this reagent. Water is added as a proton source after the reduction w/ LiAlH4 is complete.
What are the properties of NaBH4?
Milder + selectively reduced aldehydes + ketones in presence of most other functional groups.
Safer + easier to handle than LiAlH4.
What is the reduction of aldehydes + ketones?
An addition reaction – H2 added across the pi-bond.
Also a reduction reaction – Alcohol product has a fewer C-O bonds than the starting carbonyl compound
Draw the reaction scheme of reduction of aldehydes and ketones
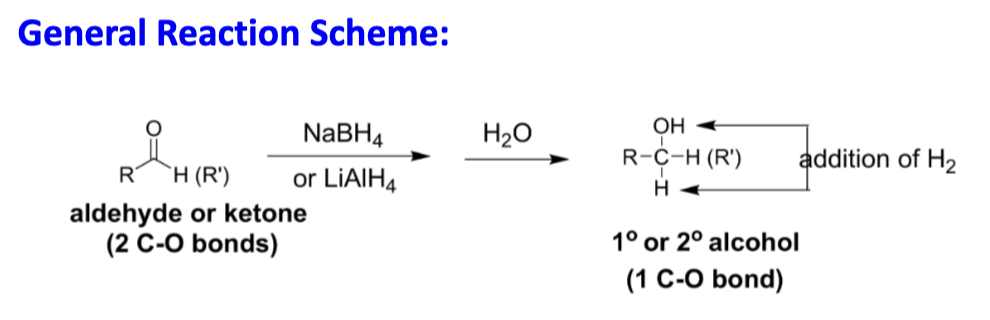
What are the products whe the starting material is an aldehyde or ketone?
Aldehyde: 1° alcohol
Ketone: 2° alcohol
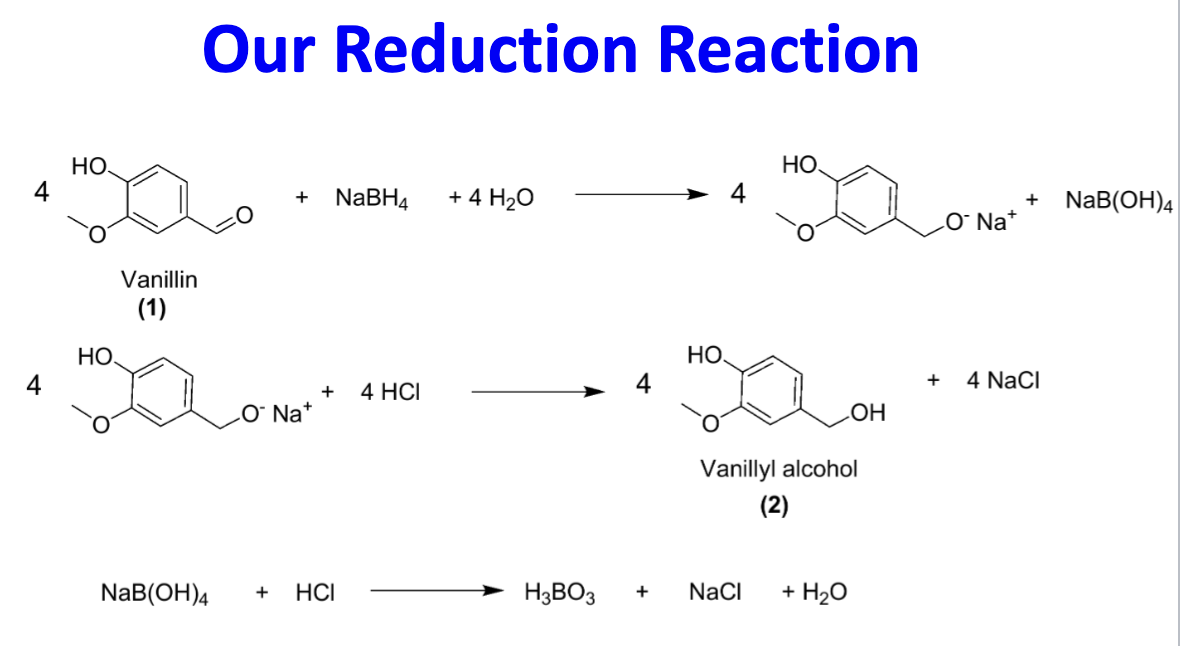
Draw the reduction of vanillin reaction scheme