oxidation of alcohols
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
11 Terms
what oxidising agent is used for the oxidation of alcohols?
acidified potassium dichromate (VI)
what can 1o alcohols be oxidised to?
aldehydes (which can be further oxidised to carboxylic acids)
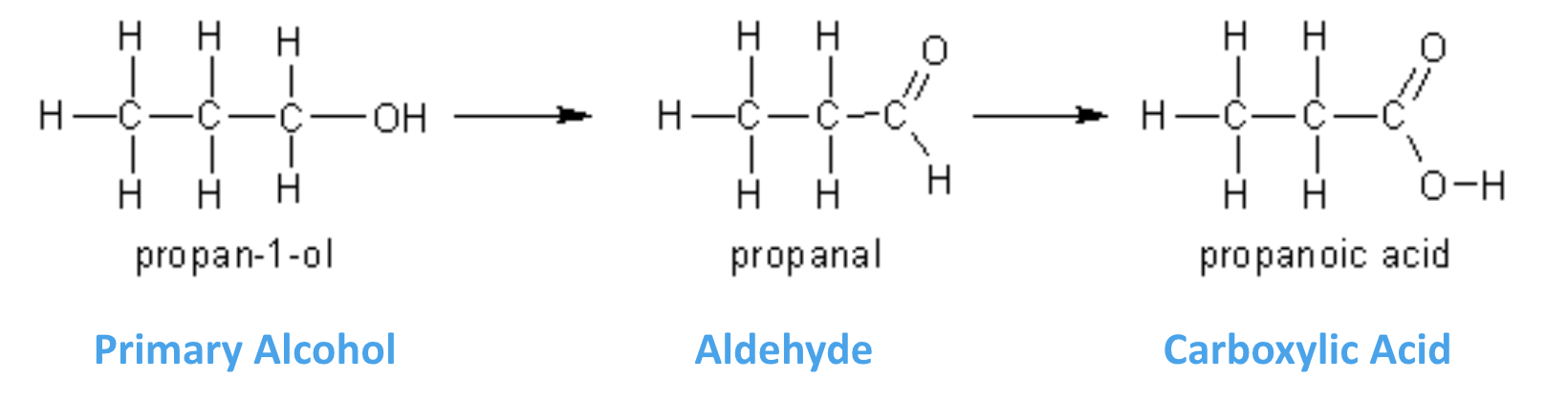
what would happen if you added an XS of the oxidising agent to a 1o alcohol?
1o alcohol would be fully oxidised to carboxylic acid (1o alcohol → aldehyde → carboxylic acid)
what can 2o alcohols be oxidised to?
ketones
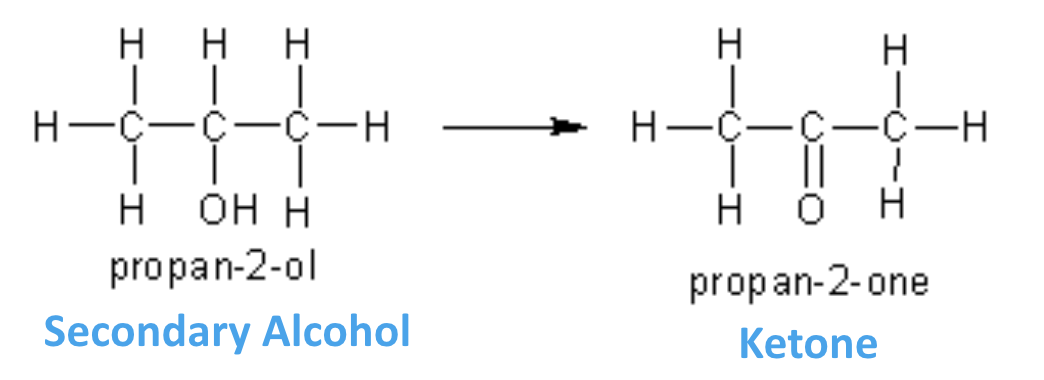
what can 3o alcohols be oxidised to?
nothing …
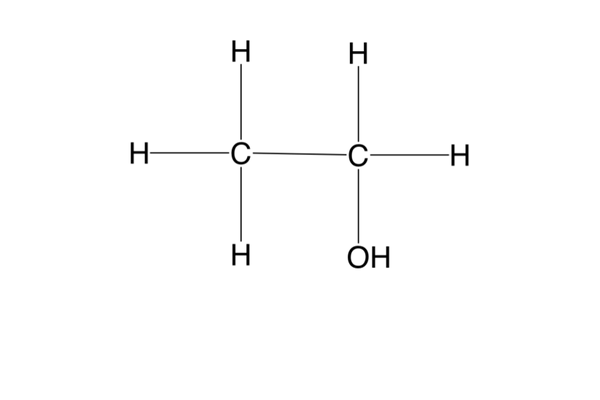
what is a primary (1o) alcohol?
an alcohol w/ only 1 C bonded to the C bonded to the functional group -OH (e.g. ethanol)
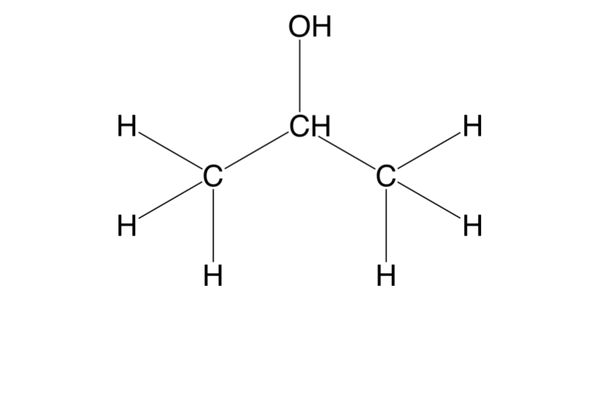
what is a secondary (2o) alcohol?
an alcohol w/ 2 Cs bonded to the C bonded to the functional group -OH (e.g. propan-2-ol)
what is a tertiary (3o) alcohol?
an alcohol w/ 3 Cs bonded to the C bonded to the functional group -OH (e.g. 2-methyl-propan-2-ol)
how do we represent the oxidising agent in eqns?
[O]
name and describe the method for the oxidation of a primary alcohol to an aldehyde:
heating under reflux:
keeps reactant in flask to allow further oxidation
anti-bumping granules used to prevent formation of large bubbles
name and describe the method for the further oxidation of an aldehyde to a carboxylic acid:
distillation:
product is distilled off as it forms to prevent further oxidation