Periodicity
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
How are Elements classifies in the Periodic Table?
They are arranged due to proton number. Elements in a period have the same number of electron shells. Elements in a group have the same number of outer electrons. Elements are also classified into s-, p-, d- and f- blocks to show electron configuration.
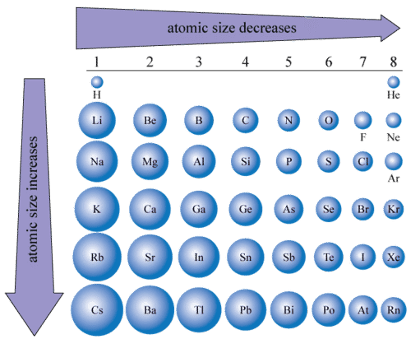
Explain the trend in Atomic Radius across a Period and down a Group?
Across a Period:
Atomic Radius decreases. This is due to an increased nuclear charge of the elements with the same number of electron shells. This means that there is a greater attraction from the nucleus so the outer electrons are pulled in closer, reducing the atomic radius.
Down a Group:
Atomic Radius Increases. This is due to an increasing amount of electron shells. This increases the distance between the outer electrons and the nucleus, reducing the attraction. More shells also increase nuclear shielding by blocking the attractive forces, reducing the nuclear attraction further, increasing the atomic radius.
Explain the trend in Ionisation Energy across a Period and down a Group?
Across a Period:
Ionisation Energy generally increases. This is due to the decreasing atomic radius and increasing nuclear charge, causing electrons to be attracted more to the nucleus, so more energy is required to overcome the attraction and remove the electron.
Down a Group:
Ionisation Energy decreases. This is due to the nuclear attraction between the nucleus and outer electrons reducing and the increasing amounts of shielding means that less energy is required to remove an outer electron.
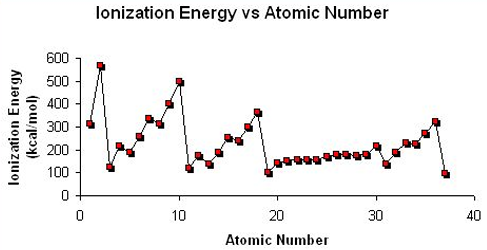
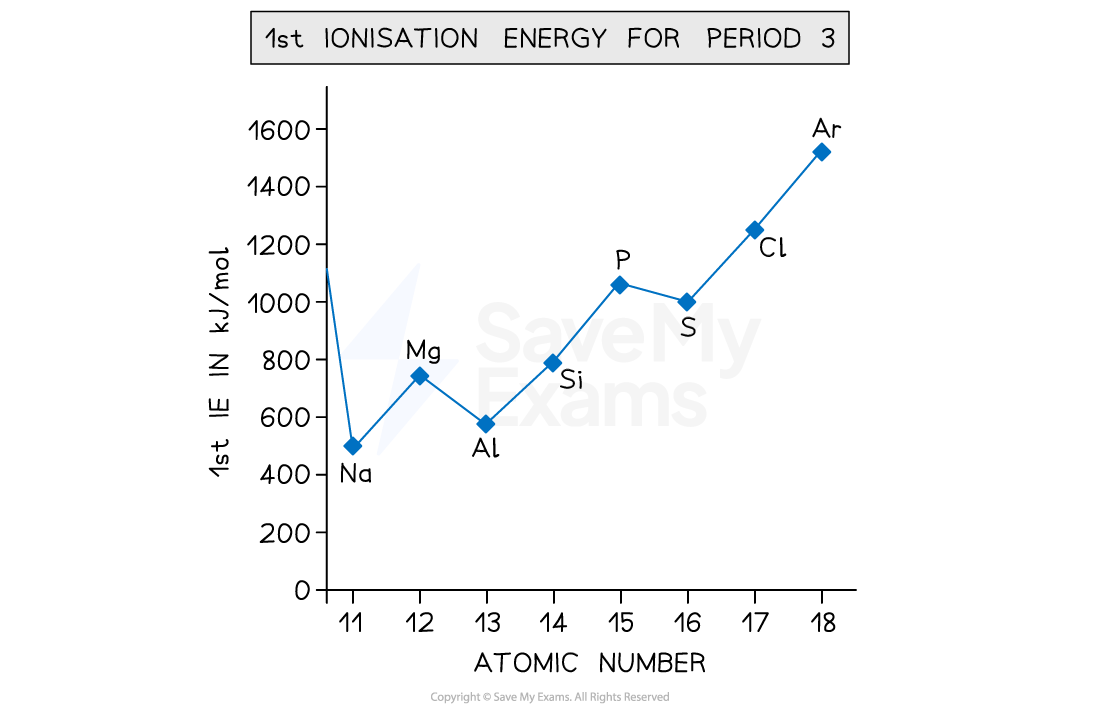
Explain the trend in Ionisation Energy across Period 3?
Ionisation Energy generally increases along Period 3. This is due to the decreasing atomic radius and increasing nuclear charge, causing electrons to be attracted more to the nucleus, so more energy is required to overcome the attraction and remove the electron.
There is a drop in First Ionisation Energy between groups 2 and 3 as it is now removing a p-block electron which is in a higher energy level compared to the s-electrons so less energy is required to remove it.
There is a drop in First Ionisation Energy between groups 5 and 6 as electrons are now being added to half-filled orbitals. This means that due to two electrons being in the same orbital, there will be electron-electron repulsion form the two negative charges, so it is easier to lose.
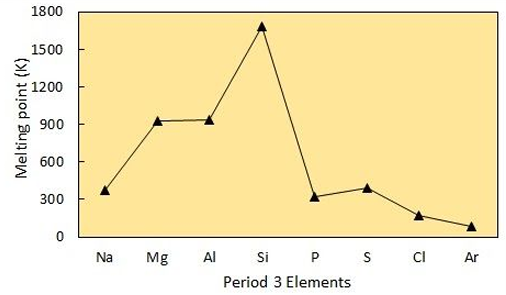
Explain the trend in Melting Point across Period 3?
Sodium - Aluminium:
These 3 elements all have metallic bonding. This means that their melting point increases based on charge. As your go along the period, the charge on the ion increases. This means that there are more electrons released so join the sea of delocalised electrons. As a result, the electrostatic forces increase so the metallic bond becomes stronger.
Silicon:
Silicon has a macromolecular structure. This means that it contains very strong covalent bonds which require lots of energy to overcome giving it the highest melting point.
Phosphorus - Chlorine:
These 3 elements are all simple covalent molecules (P4, S8, Cl2) held together by weak van der Waals forces. As these don’t require much energy to overcome their melting points are lower. The molecules with more electrons have more van der Waals forces so it has a higher melting point, making the order S8 > P4 > Cl2.
Argon:
Argon is a noble gas that exists as an individual atom due to its full outer shell. This makes it very stable but with very weak van der Waals forces. As a result, it has the lowest melting point causing it to exist as a gas at room temperature.