L22-reductions
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
9 Terms
What is a good reducing agent for aldehydes and ketones
Sodium borohydride (mild reducing agent )
What are the two steps/reagents needed when carrying out a reduction of aldehydes and ketones
Mild or strong reducing agent and a solvent e.g. ethanol
Acid
What is needed when doing a reduction involving carboxylic acids, esters amides and what happens in the reaction
Strong reducing agent (as they have a higher oxidation level )
No acid as the intermediate contains a leaving group that can leave
remember that the product an aldehyde/ketone is more reactive than the reactant so it also reacts with the reducing agent
Forms alcohol
What type of carbonyl compounds can mild reducing agents react with
Only the most reactive ones, the ones less reactive than ketones need a strong reducing agent
What product is formed when nitriles are reduced
Amines
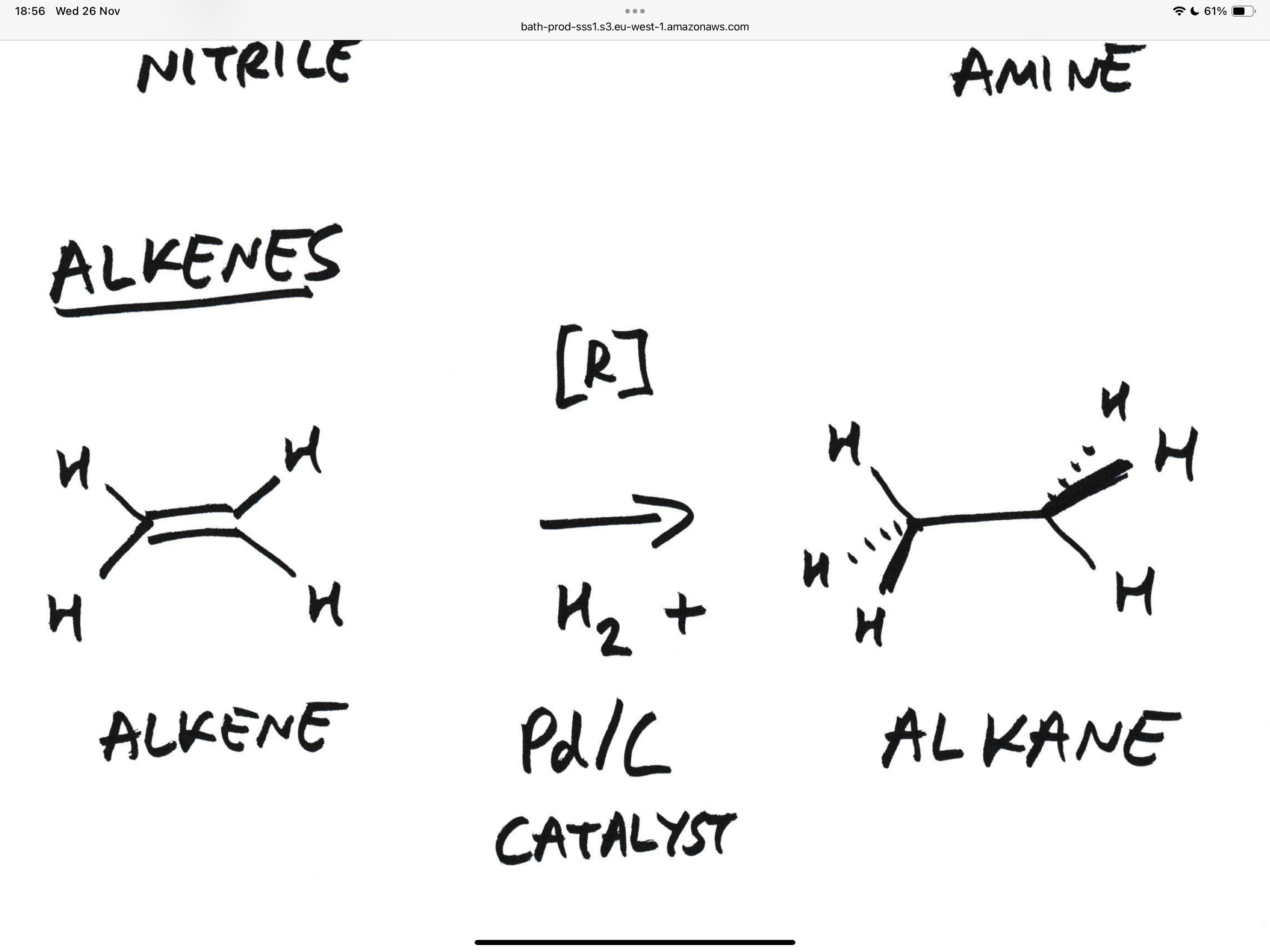
What is the reaction for the reduction of alkenes (Ethene for example ) +what are the catalysts
H2 and pd/c
Both hydrogens are delivered in the same face
What is the product and reagents of the reduction of an alkyne
Alkyne → alkene → alkane
H2 and pd/c (or Lindlar catalyst which is just poised pd/c )
when lindlar is used reduction is stoped at the alkene (cis only as both hydrogens are delivered at the same face )
What sort of compounds use hydrogen and pd/c as their reducing agent
nitro
Alkynes
Alkenes
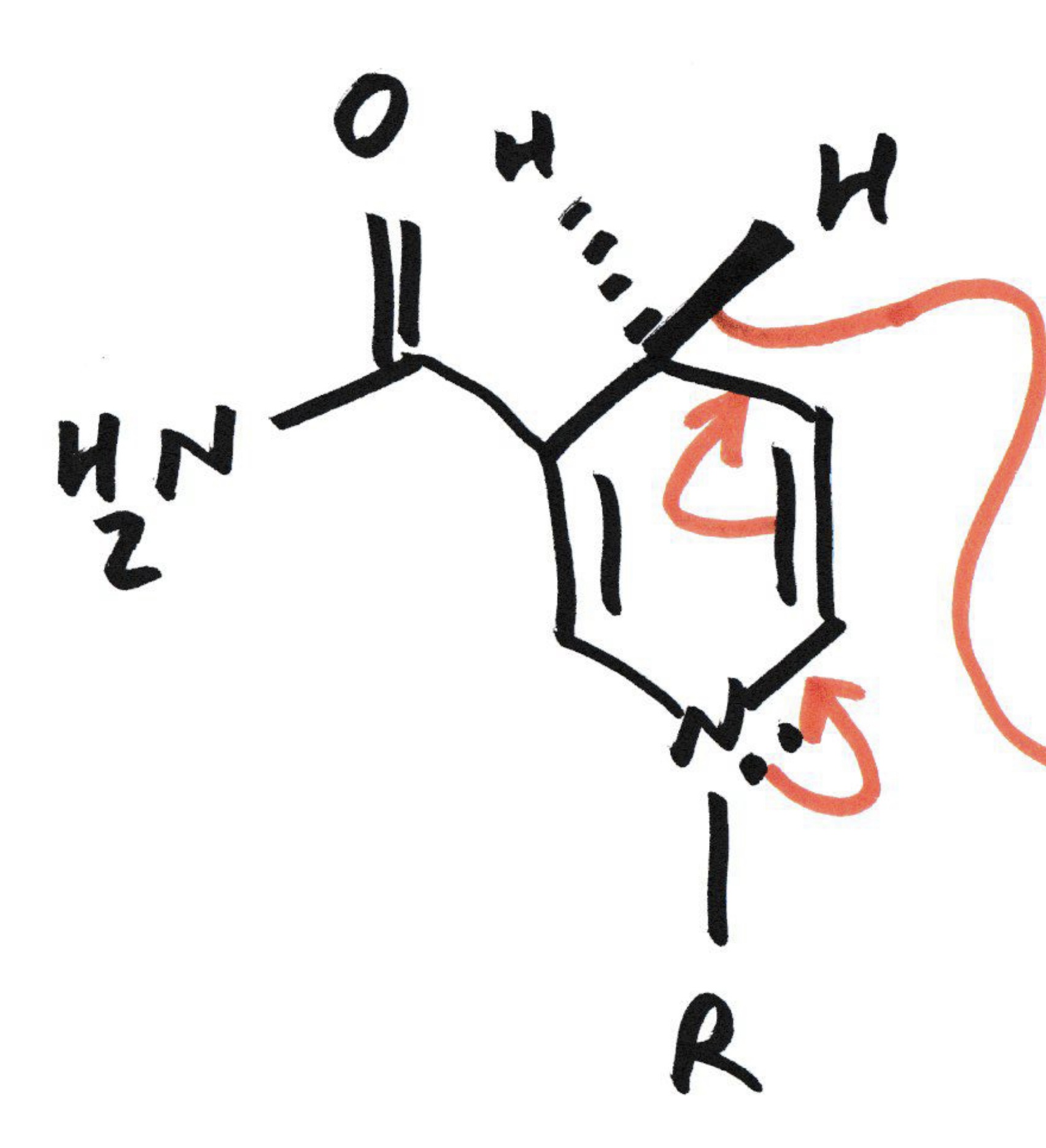
What does nature/ our bodies use to make reduction (oxidation ) reactions occur
NADH/NADPH and its reduced into NAD+
(nicotineamide adenine dinucleotide )