4. Models of Bonding and Structure // S2.1, S2.2
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Ionic compounds
Formed when electrons are transferred from one atom to another
Forms ions with complete valence shells
The cations and anions are attracted to each other by strong electrostatic forces
Have high melting opints
Naming binary ionic compounds
Cation first, Anion second, add -ide suffix
Covalent bonds
Sharing one or more pairs of electrons so each atoms achieves a noble gas configuration
Coordinate bonds
Both shared electrons come from the same atom
2 Electron domains
Linear, 180°
3 electron doains
Trigonal planar, 120°'
bent or V-shaped (smaller than 120°)
4 electron domains
Tetrahedral, 109.5°
Trigonal pyramidal, 107°
Bent, or V-shaped 105°
Differences in single, double. triple bonds
The more bonds in one, the shorter and stronger
Boiling point
Liquid to gas
All attractive forces between particles are broken
Good indication of the strength of intermolecular forces
Melting point
Crystal structure is broken down, but attractive forces between particles exist
Impurities impact
Impact structure and result in lower melting points
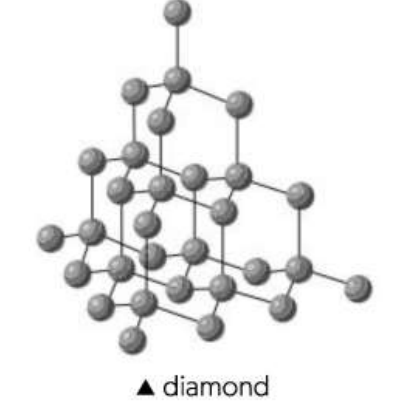
Diamond properties
Each carbon covalently bonded to four others
Giant tetrahedral diamond structure'
109°
1.54/1010 m long lengths.
All electrons are localized = Doesn’t conduct electricity
All bonds equally strong, so hard to break and high melting point
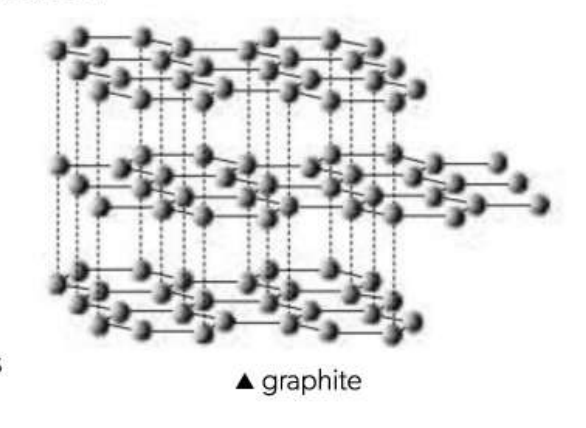
Graphite
Each Carbon is bonded to three other carbons
Layers of hexagonal rings
120°
1.42/1010 m
Weak attractive forces between layers = layers slide over each other = lubricant and waxy feeling
Between layers, electrons are delocalized = Good conducter of electricity
Graphene
One atom thick layer of Graphite
Extremely light
Semiconductor
200 times stronger than steel
Fullerenes
Large spheroidal molecules = hollow cage of sixty or more carbon atoms.
E.g. Buckminsterfullerene C60
Hexagons and pentagons → geodesic spherical structure similar to a football
Silicon
Giant tetrahedral covalent structure like diamond.
Si-Si bond length is 2.32x10-10 m
hard brittle solid
high melting point
metalloid
semiconductor.
Silicon dioxide (silica)
SiO2, quartz
Giant tetrahedral covalent structure similar to diamond and silicon.
Very hard and has a high melting point.
No delocalized electrons = does not conduct electricity.
unipositive cation
positively charged ion with a charge +1
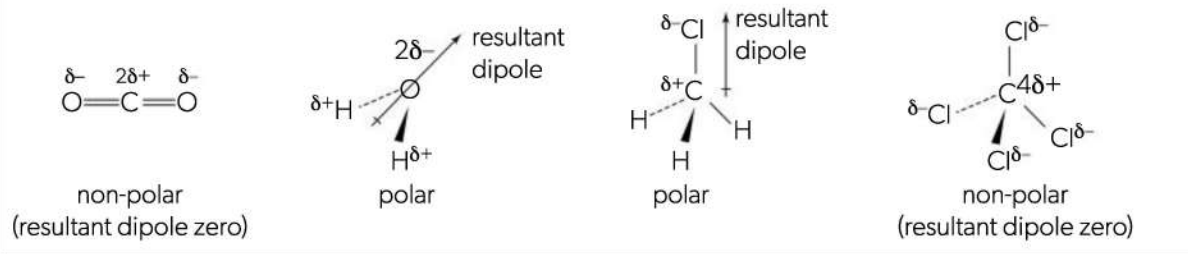
Bond polarity
More electronegative atoms exert greater attraction for electron pair(s)
One end of the bond will be more electron rich, resulting in bond dipole (polar bond)
Put number behind the atom with positive pole for how many bonds its pulling electrons with. E.g. NH3 would have Nitrogen with 3§+
Shape can also cause polarity.
Allotropes
Element can exist in different crystalline forms.
Different bonding and structural patterns, thus different chemical and physical properties
Intermolecular forces
Van Der Waals Forces
London Dispersion Forces
Dipole-Dipole Forces
Hydrogen Bonds
Van Der Waals Forces
More inclusive term for intermolecular forces where a temporary dipole occurs between both polar and non-polar molecules
London Dispersion Forces (LDP)
Temporary instantaneous dipole, which can induce another dipole in a neighbouring particle, resulting in a weak attraction between the two particles.
Increase with increasing mass.
Dipole-Dipole Forces
Polar molecules are attracted to each other by electrostatic forces.
Relatively weak, but the attraction is stronger than London dispersion forces.