Practical 23: The Molecular Basis of Cancer
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Vogelstein's original 1990 model
1. Mutations in the APC gene were almost equally frequent in all adenomas and adenocarcinomas;
2. The frequency of KRAS mutations was low in early adenomas, but high in later adenomas and adenocarcinomas
3. Mutations in p53 and deletions of chromosome 18 were uncommon except in late adenomas and adenocarcinomas.
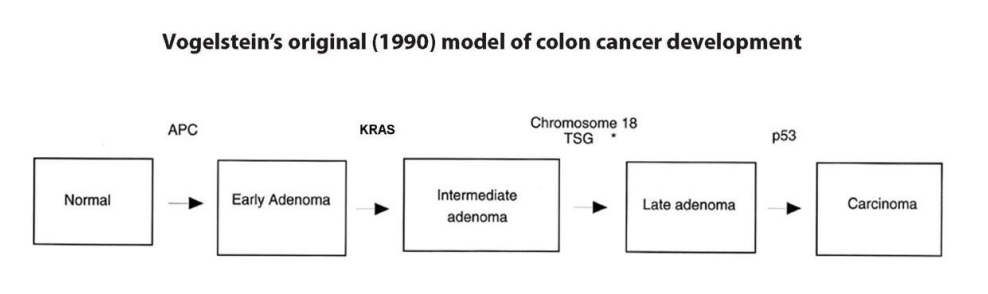
Vogelstein's 2008 model
Adenomatous Polyposis Coli / Familial Adenomatous Polyposis
APC is very important in regulating cell cycle progression. The APC gene is so named after the hereditary condition Adenomatous Polyposis Coli, which is also known as Familial Adenomatous Polyposis (FAP) or polyposis coli.
What would we expect to find if we sequenced the APC gene in germ-line DNA of a patient with FAP, and a colonic adenoma or cancer from that patient?
FAP is a classic example of Knudson’s “Two Hit Hypothesis” for tumour suppressor genes like Rb-1, the gene for inherited susceptibility to Retinoblastoma. FAP patients inherit a mutated APC gene in every cell in the body so they have a very high probability of having colorectal epithelial cells that by losing or mutating their normal copy of APC end up with only mutant APC and are on the road to an adenoma.
So in the germ-line of an FAP patient we should find one mutant and one normal copy of the APC gene, while in an adenoma or cancer we would find only the mutant form.
If we sequenced germ-line DNA in a more typical sporadic colon cancer patient, how would the result be different?
In a more typical sporadic colorectal cancer patient, the germ-line DNA would show no mutations in APC.
It is important to note that all patients with sporadic colorectal cancer have NOT inherited an APC mutation (only those with FAP inherit a germ-line mutation in one copy of APC).
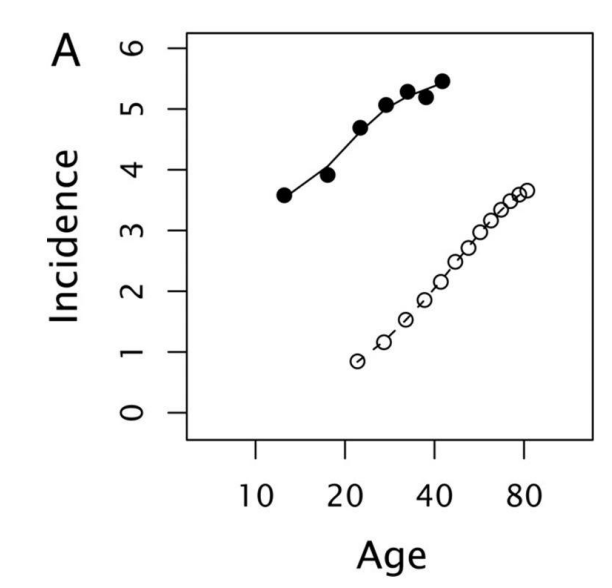
The graph compares the incidence (per 106 population on a log10 scale) of colorectal cancer in patients with FAP (black dots) and typical, sporadic cases (white dots).
What differences between the two populations do you notice?
Patients with FAP have a higher incidence of colorectal cancer and develop it earlier than sporadic cases.
With one copy of the tumour suppressor gene APC already mutated, they only require loss/mutation of one further copy to develop the first step in the multistep progression to cancer. Sporadic cases, however, must sustain mutations to both copies within the same cell genome. Therefore, FAP patients are more likely to develop colorectal cancer and will do so at an earlier age, in keeping with the graph findings.
Suppose someone is discovered to have Familial Adenomatous Polyposis, and there is concern that some of their close relatives may also be affected. How would you identify the affected relatives?
If someone is suspected of belonging to an FAP family they would usually be examined by colorectal endoscopy, which reveals hundreds of adenomatous polyps in the large intestine (usually in the late teens or twenties – see below). They might have a family history.
To identify the affected relatives, testing of their germ-line DNA (extracted from blood) can be performed for the known mutation in their family, but for this you would need to know where the mutation is, by sequencing the APC gene in the known FAP case in the family, and this is technically difficult even today, though maybe not in future.
Suppose a family has a clear history of inherited predisposition to colorectal cancer. Are they likely to have FAP, i.e. an APC mutation, or what other explanation would you consider first?
Not all hereditary predisposition to colorectal cancer is due to inherited APC mutations; in fact, far commoner is Lynch Syndrome (which used to be called HNPCC: Hereditary Non-Polyposis Colorectal Cancer) which is inheritance of a mismatch repair deficiency.
These patients do not show polyposis (the presence of very large numbers of polyps in the intestines). They may show only a few polyps or no polyps prior to colorectal cancer formation, so endoscopy wouldn’t pick up affected relatives.
48 year old male has a 5 week history of abdominal pain and intermittent rectal bleeding. the gentleman's sister, aged 39, is being treated for endometrial cancer. A colonoscopy detects a single focus of narrowing in the ascending colon (first part of the colon), which is biopsied. In the uninvolved areas of bowel, no significant inflammatory changes are identified.
What is your favoured diagnosis on the basis of the clinical history and colonoscopy findings?
The most likely diagnosis is a primary colorectal adenocarcinoma.
48 year old male has a 5 week history of abdominal pain and intermittent rectal bleeding. the gentleman's sister, aged 39, is being treated for endometrial cancer. A colonoscopy detects a single focus of narrowing in the ascending colon (first part of the colon), which is biopsied. In the uninvolved areas of bowel, no significant inflammatory changes are identified.
What unusual features of the patient's history can you identify so far?
1. This patient is rather young to have developed such a disease sporadically, although this is not impossible.
2. His sister has endometrial cancer at a rather young age.
3. Most colorectal cancers are in the descending colon (the last part of the colon).
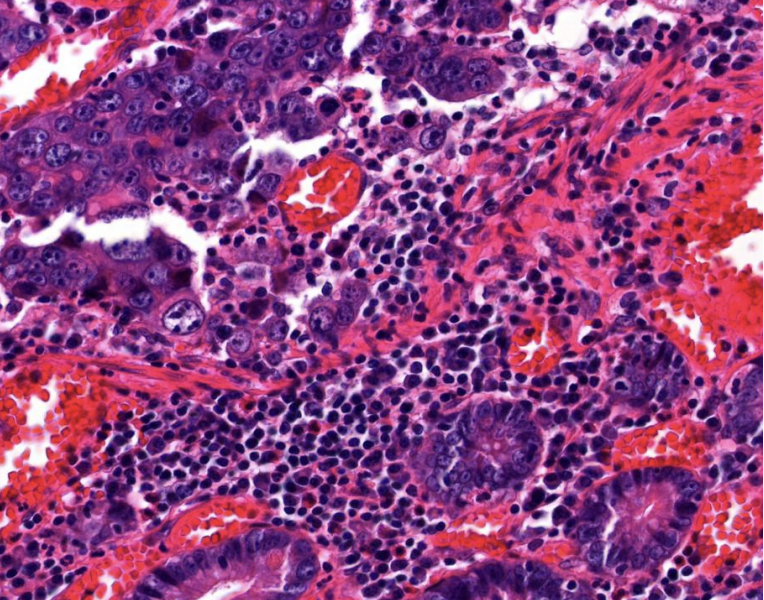
the patient's biopsy histology, what is your favoured histological diagnosis?
Colonic adenocarcinoma
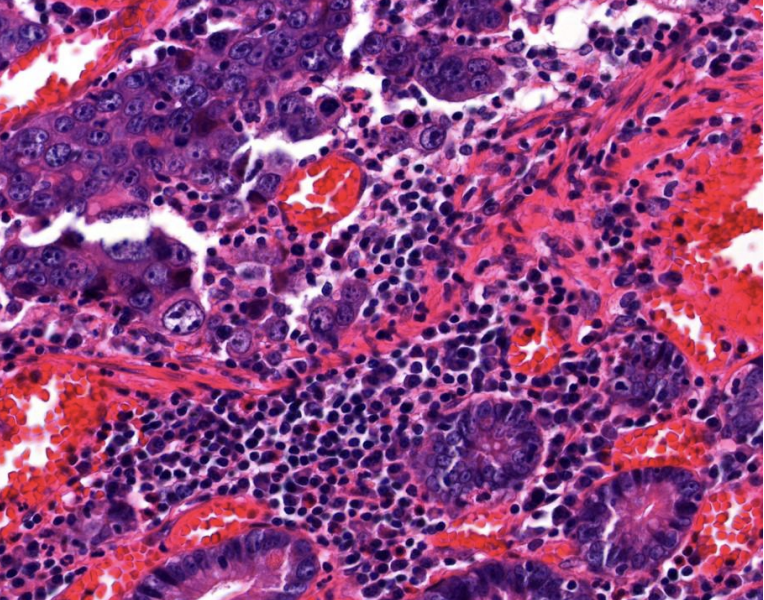
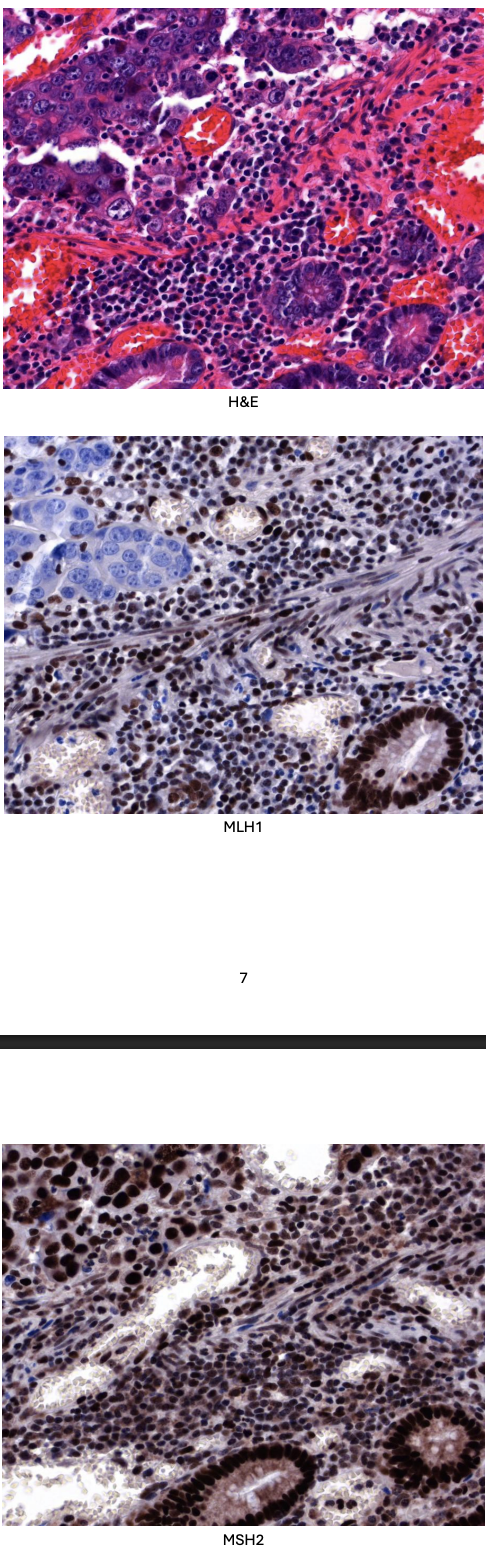
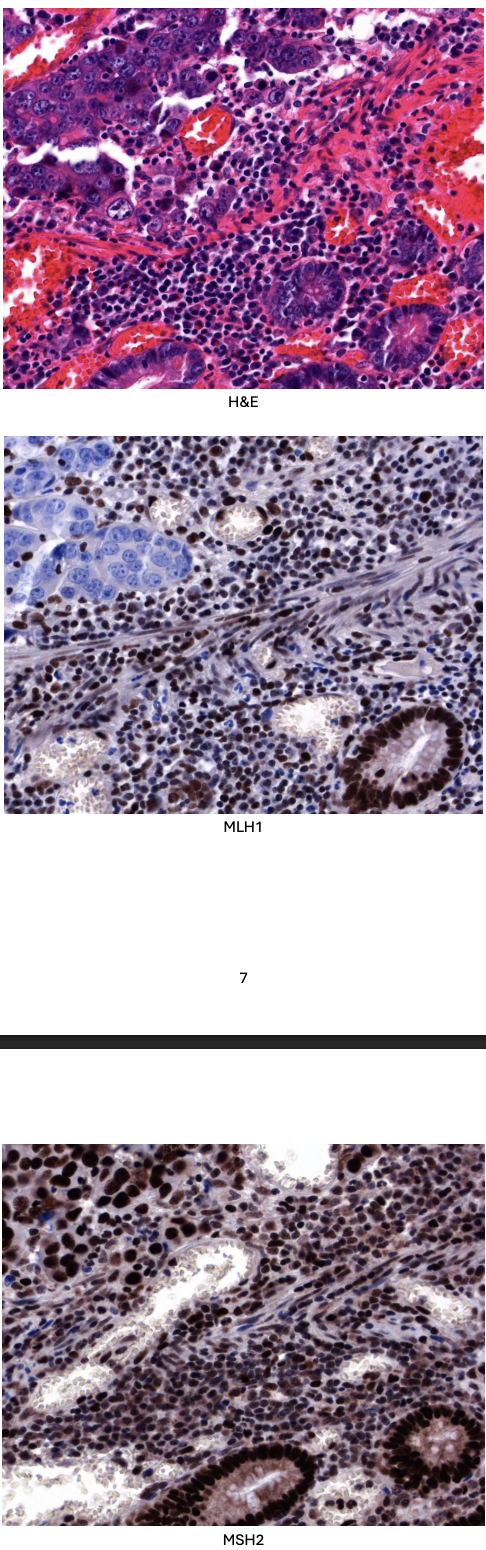
Biopsy tissue was sent to the histopathology department for analysis and a moderately differentiated adenocarcinoma was diagnosed. Immunohistochemistry for MLH1 and MSH2 was performed, and the results are shown below. (Positive staining for MLH1 and MSH2 is brown.)
There is loss of MLH1 immunostaining within the cancer cells but preservation of MSH2. MLH1 protein is missing.
The underlying MLH1 gene is mutated, with likely deficiency in mismatch repair with microsatellite instability at a molecular level.
The patient's father was diagnosed with adenocarcinoma of the caecum at the age of 45 years and his sister was diagnosed with endometrial carcinoma aged 39. What should the treating clinicians explain to the patient and his family about the implications for other relatives?
This clinical and laboratory information suggests that the patient has Lynch syndrome (a hereditary nonpolyposis colorectal cancer syndrome), caused by a pathogenic germline variant (a hereditary mutation) in a mismatch repair gene, in this case MLH1. In individuals with a mismatch repair gene mutation, the risk of developing colorectal cancer is 80% by age 75.
Patients with Lynch syndrome more commonly develop cancer in the ascending (first part) of the colon, compared with the general population. Lynch syndrome is also 9 associated with a predisposition to the development of other malignancies (e.g., endometrial, ovarian, and stomach).
Identifying Lynch syndrome in affected individuals is important, as close surveillance (colonoscopy every 1–2 years) of at-risk family members can reduce the rate of colorectal cancer.
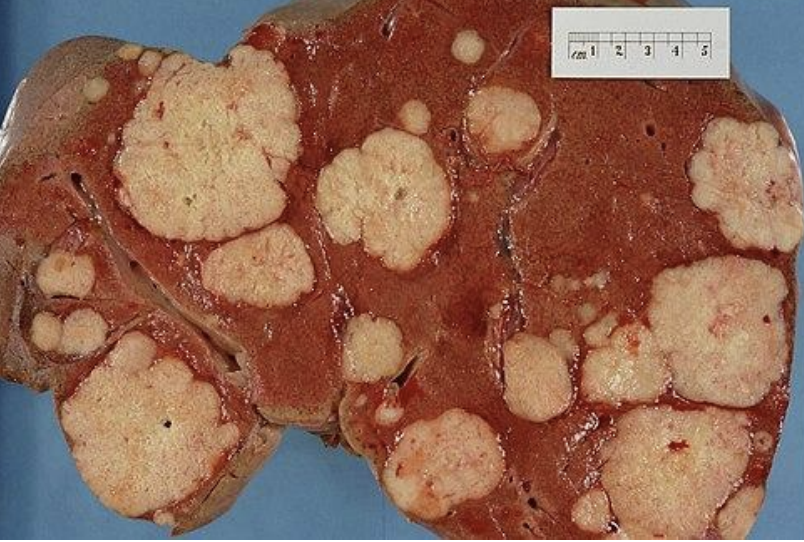
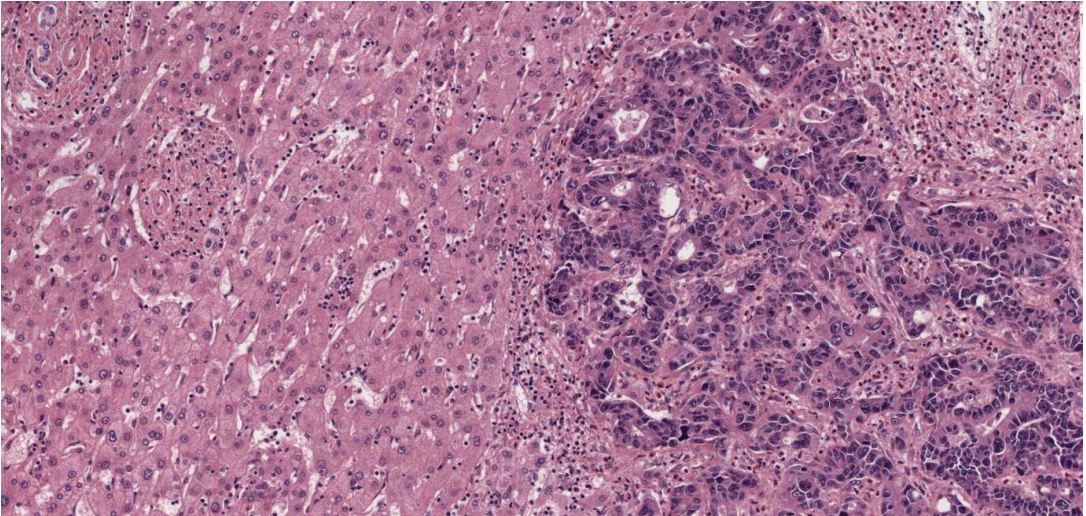
The liver abnormalities are metastases originating from the colon. Below is a post mortem example of a liver in which there are multiple metastases of colonic adenocarcinoma. Our patient might develop additional liver metastases like this in due course.
Lymph nodes are likely ‘bulky’ due to tumour spread via the lymphatic system.
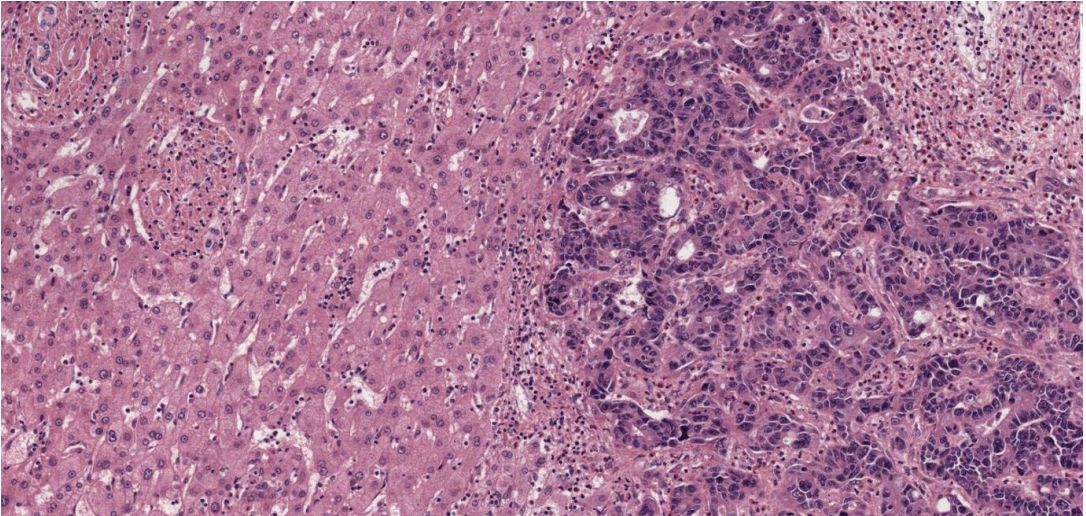
Liver tissue infiltrated by irregular and sometimes cribriform glands, the nuclei of the cells are pleomorphic and hyperchromatic. Necrotic debris is evident. There is a desmoplastic (densely fibrotic) response.
Morphologically, there is gland formation, typical for colonic mucosa, but not for the liver. Immunohistochemistry for markers characteristic of particular tissues (e.g., colon) could also be used. This is common practice in pathology.
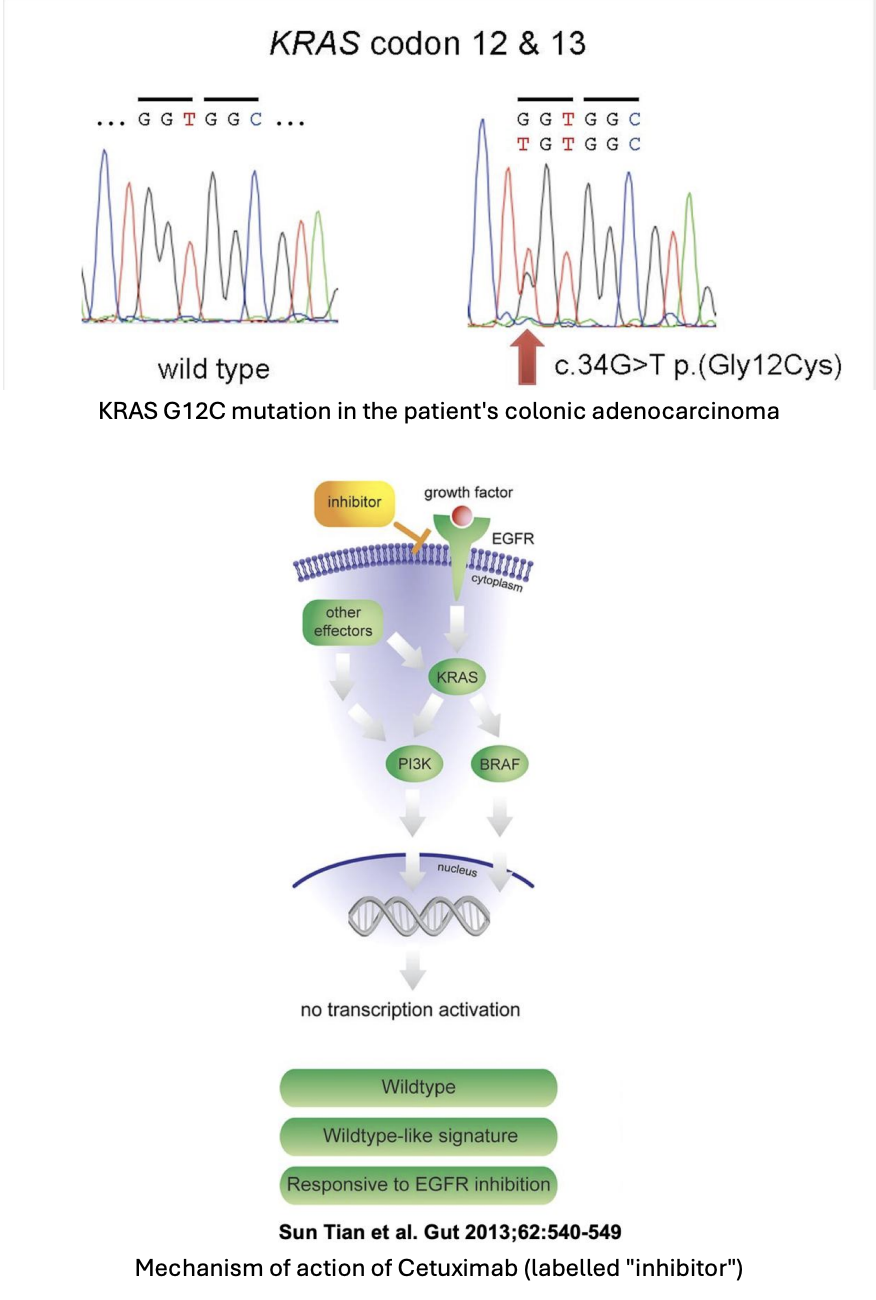
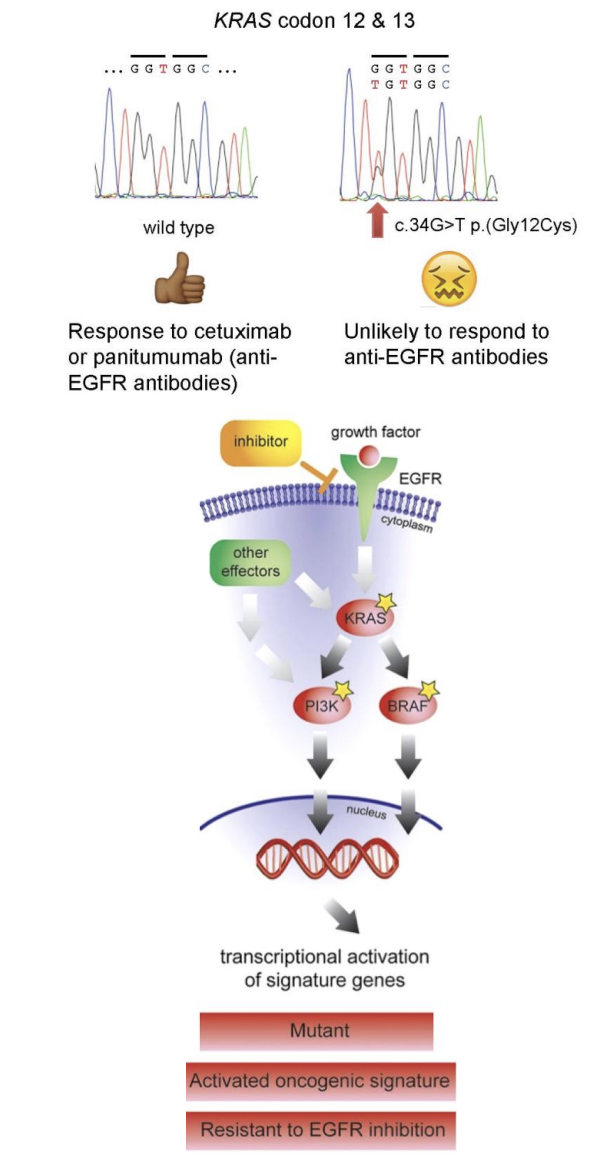
What is your conclusion about whether cetuximab would be a suitable treatment?
The patient's activating KRAS G12C mutation will lead to activation of pathways downstream of KRAS, regardless of whether the EGF receptor is inhibited by Cetuximab. Therefore, Cetuximab treatment is unsuitable. It can be seen that activating mutations in PI3K and BRAF would have similar effects.
This underlines the importance of considering genes as part of pathways, rather than just in isolation, when we are considering the effect of particular mutations in cancer.
Problem-Solving Exercise 3 Part 1: APC Mutations in Cancer
Look at hand out to do from page 13-22
Answers attached here:

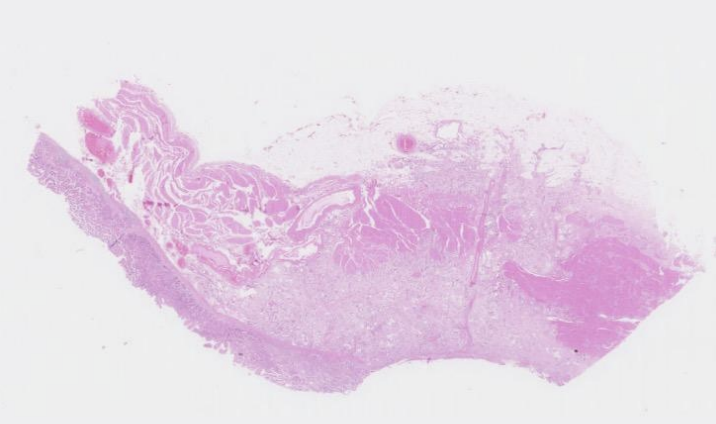
stomach from a 71 year old male
This section of stomach shows extensive ulceration, which appears to be due to an invasive adenocarcinoma. The adenocarcinoma is composed of irregular glands and tubules formed from neoplastic cells (with enlarged nuclei showing marked pleomorphism and hyperchromasia with some mitotic figures) invading into the main layers of the stomach wall including the mucosa, submucosa and muscularis propria, with invasion through the muscular wall into the serosa.
There is reasonably close resemblance to the parent gastric glandular epithelium, so this is graded as a well to moderately differentiated adenocarcinoma. The process is malignant neoplasia of the gastric glandular epithelium
Stomach: Well-to-moderately differentiated adenocarcinoma
This disease is often fatal because gastric cancers tend to be diagnosed at an advanced stage and have already spread beyond the stomach to form peritoneal metastases, lymph node metastases and spread to other organs (e.g. liver, lung, ovary) and are difficult to treat successfully.
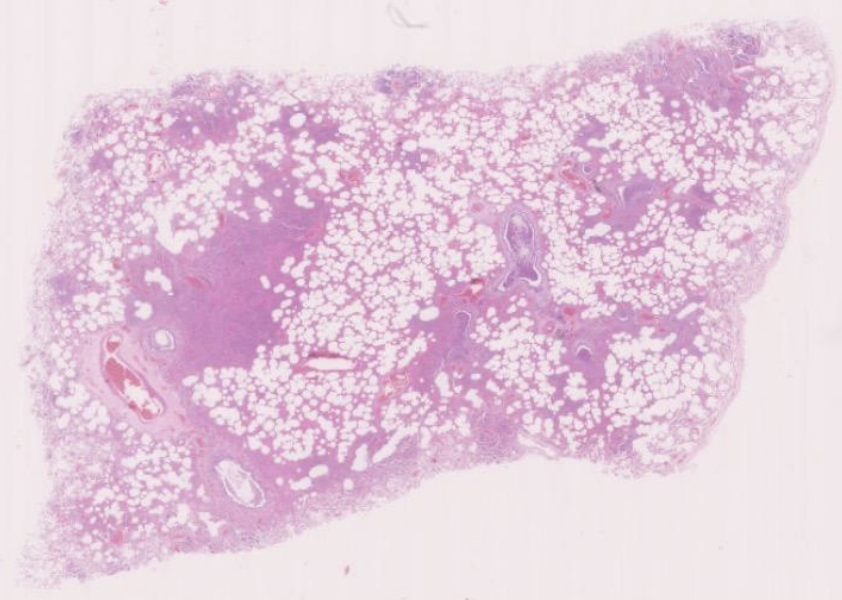
lung from an 80 year old female, who had had a previous stroke, who was found dead at home.
This is lung tissue in which some normal alveolar airspaces are seen. These appear white, as they would have contained air in life. In other alveolar airspaces, particularly those centred around the bronchi, there are abundant acute inflammatory cells (neutrophils) associated with fibrin. Smaller numbers of macrophages are also seen. The capillaries are very dilated. The bronchi also contain large numbers of neutrophils. In a few places, these can be seen infiltrating the respiratory epithelium lining the bronchi.
In some of the airways, aspirated food (plant) material can be seen. This may be because the previous stroke had cause impairment of swallowing reflexes.
There is no evidence of pulmonary thromboemboli, infarction or malignancy.
The large numbers of neutrophils favour a bacterial aetiology. A Gram stain could be performed on the tissue in order to try and visualise and subclassify the causative organism.
The features are those of acute bronchopneumonia