Chapter 23: Polymers and Alcohols
23.1-Addition Polymers
Polymers
- Polymers are long molecules made up of hundreds or thousands of repeating subunits.
- They are really useful materials for everyday items, such as plastic containers and clothing, but they are a nuisance when it comes to throwing them away because they aren’t broken down easily.
Addition Polymerisation
- Addition polymers are formed by joining lots of alkene molecules together.
- The single alkene is called a monomer and when several monomers are connected together, we call this a polymer.
- Polymerisation happens when the double carbon bond breaks, allowing another alkene to connect to the carbon.
- This happens multiple times until you end up with a polymer made up of hundreds or thousands of monomers.
- Instead of drawing out a really long chain, it’s much easier to represent polymers by drawing a single subunit (the monomer) inside square brackets, with a little ‘n’ in the right hand corner to show that we have lots of them joined together.
- Remember to draw bonds sticking outside the square brackets to make it clear that the chain continues.
- We name the polymer depending on the type of monomer it is made from and stick the word poly at the front.
- Let’s say we have a polymer made up of lots of ethene molecules joined together - this would be called poly(ethene), which we also refer to as polythene.
- Polythene is everywhere - you’ll find it in things like plastic water bottles, bin liners and hose pipes.
23.2-Alcohols
Alcohols
- Functional Groups
- The ‘O-H’ bond is the functional group of alcohols. Alcohols contain a bond between O-H, where the oxygen is bonded to a carbon. This is their functional group, used in reactions.
- As we have seen with alkanes and alkenes, alcohols can also be named. The first four alcohols in the homologous series are called methanol, ethanol, propanol and butanol.
Representing Alcohols
- Alcohols can be represented. We can represent alkenes very easily, by using the number of carbons each molecule contains. Ethanol can be represented as CH3CH2OH.
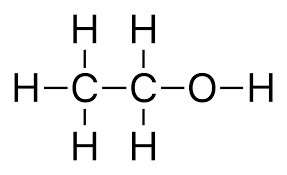
- Alcohols can be drawn out. We can also represent alcohols using their displayed formulae. For example, the displayed formulae of ethanol is shown below.
Production of Ethanol by Fermentation
- Ethanol (C2H5OH) is one of the most important alcohols
- It is the type of alcohol found in alcoholic drinks such as wine and beer
- It is also used as fuel for cars and as a solvent
- It can be produced by fermentation where sugar or starch is dissolved in water and yeast is added
- The mixture is then fermented between 15 and 35°C with the absence of oxygen for a few days
- Yeast contains enzymes that break down sugar to glucose
- If the temperature is too low the reaction rate will be too slow and if it is too high the enzymes will become denatured
- The yeast respire anaerobically using the glucose to form ethanol and carbon dioxide:
Practice Questions:
- Give an example of an alkene polymer.
- Polythene/propene/butene
- How are polymers made?
- Monomers join together by polymerisation
- Define condensation polymerisation.
- Condensation polymerisation involves monomers with two functional groups
- When these monomers react they join together usually lo sing small molecules, so are called condensation polymerisation reactions