AP Chemistry Kinetics
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
23 Terms
The shorter the half life
the higher the value of k (constant) is
half-lives for zero order
decrease (as shown by arrows)
Half lives for first order
remain constant
half lifes for second order
increase
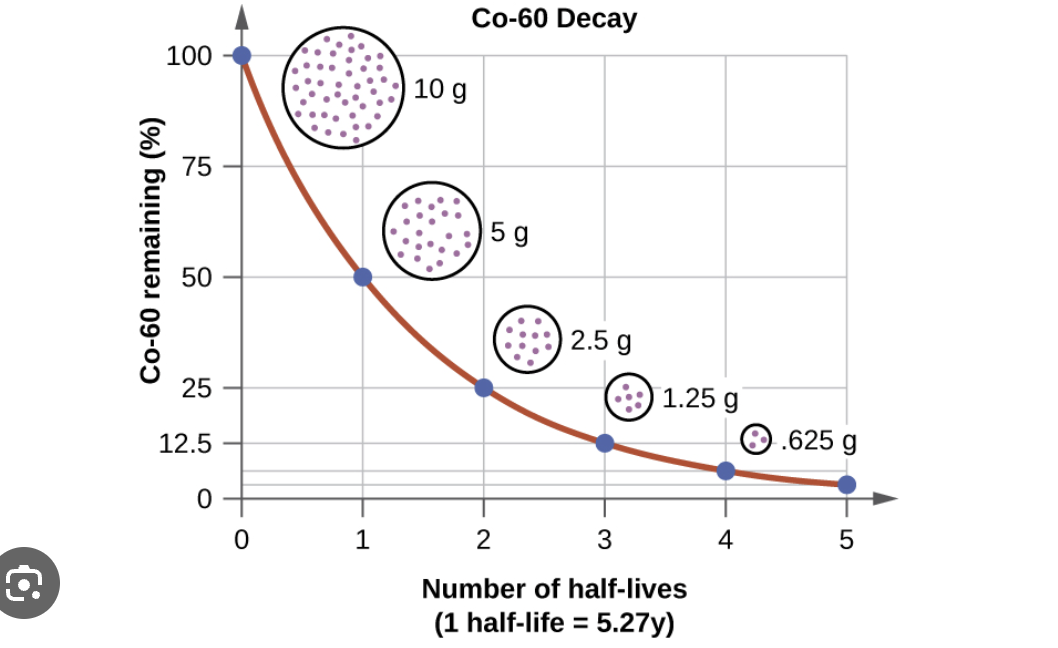
Nuclear decay
Is an example of a first order reaction
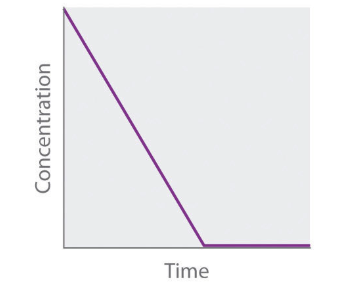
Concetration vs. Time Graph of Zeroth Order
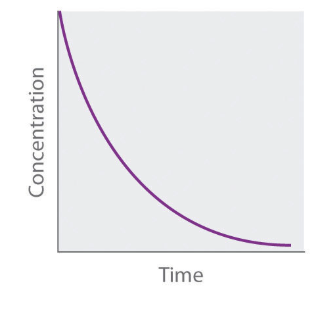
Concentration vs. Time Graph of First Order
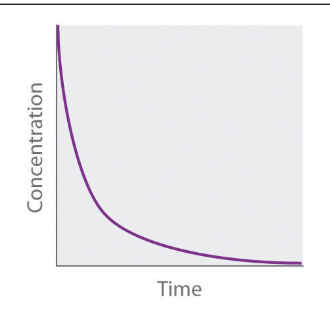
Concentration vs. Time Graph of Second Order
Straight line plot to determine rate constant zeroth order
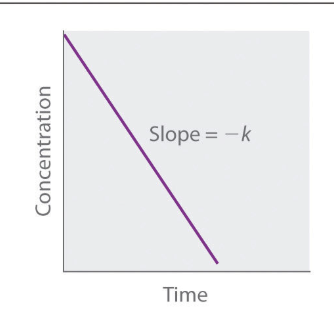
Straight line plot to determine rate constant first order
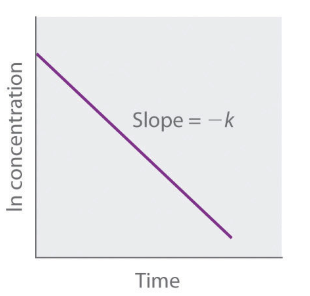
Straight line plot to determine rate constant second order
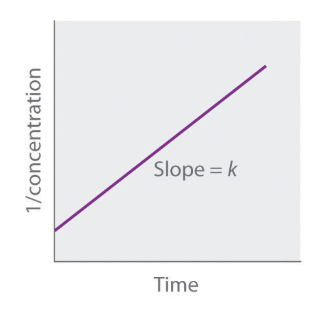
Relative rate vs. concentration of zeroth order
[A]t M Rate1 M/S
1 1
2 1
3 1
Relative rate vs. concentration of First Order
[A]t M Rate1 M/S
1 1
2 2
3 2
Relative rate vs. concentration of second order
[A]t M Rate1 M/S
1 1
2 4
3 9
half life for + unit k , rate constant for zeroth order
t12=[A]0/2k + M/s
half life for + unit k, rate constant for 1st order
t12= 0.693/k + 1/s
half life for + unit k, rate constant for second order
t12= 1/k[A]0 + M 1-s1
1st order reactions always stay
constant
[A]t
the concentration at a time t (what we want to find)
[A]0
the initial concetration
k
rate constant
t
the time elapsed
you can rearrange a formula to solve for
[A]t