4.1.1 Basics of Organic Chemistry
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
What does nomenclature mean?
The system used for naming organic compounds
What does the term empirical formula mean?
Simplest whole number ratio of atoms in a molecule
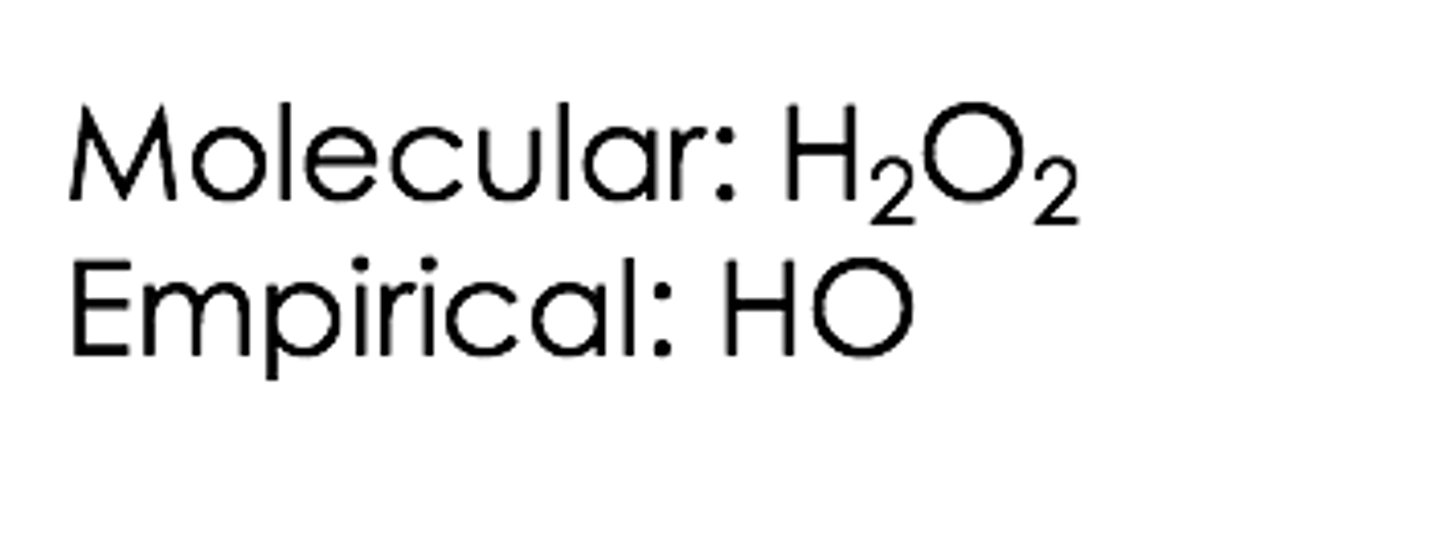
What does the term molecular formula mean?
It provides the actual number of atoms of different elements in a molecule
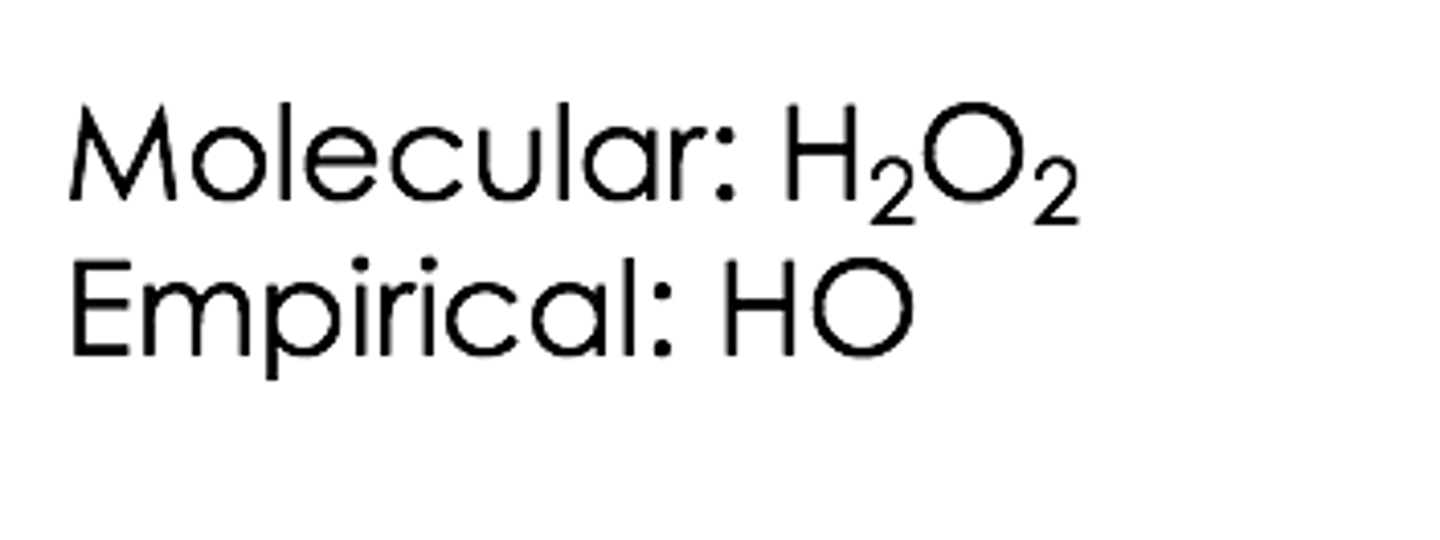
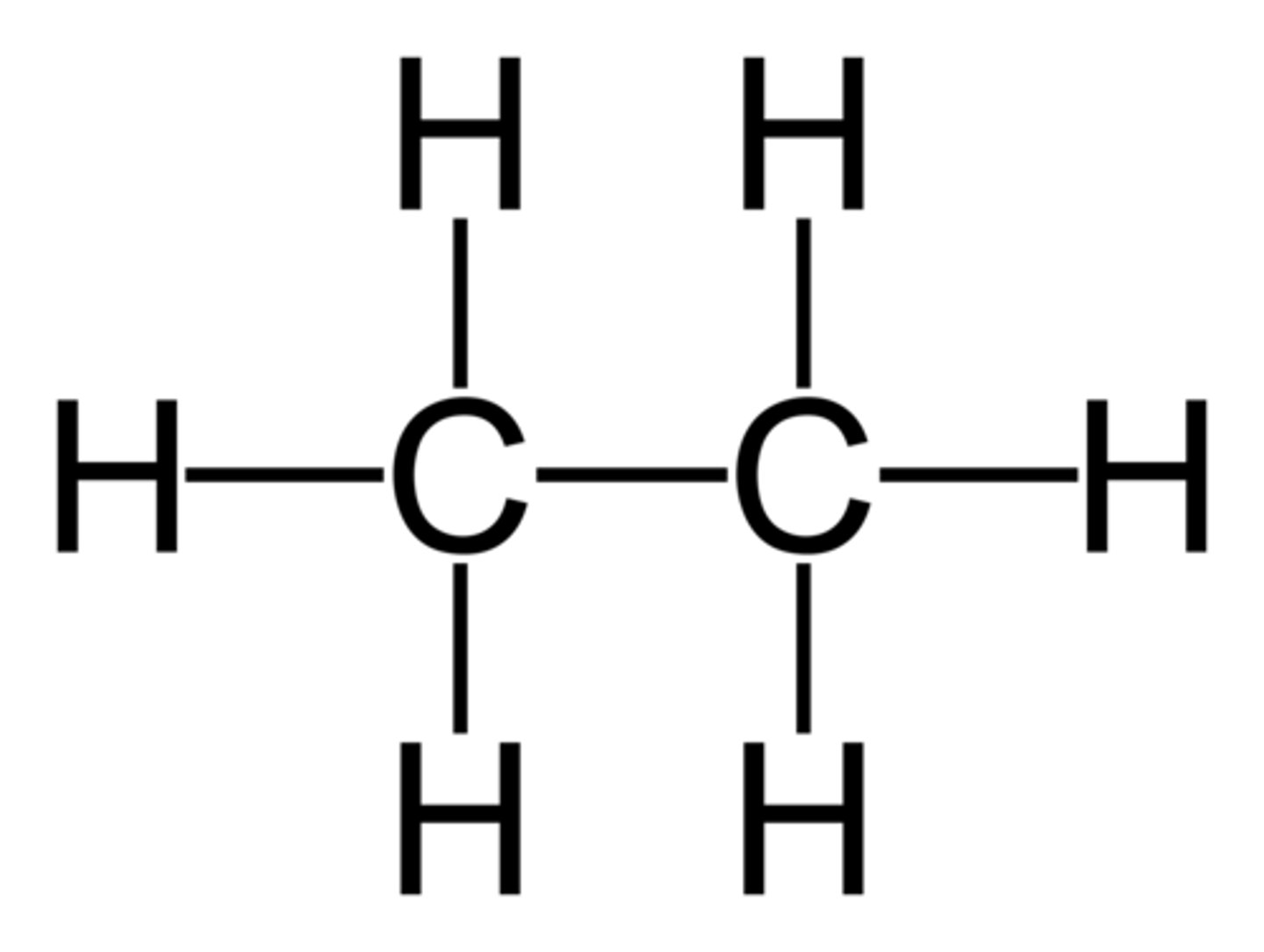
What does the term displayed formula mean?
It shows every atom and every bond in a molecule
What does the term structural formula mean?
It shows the arrangement of atoms in a molecule without showing every bond
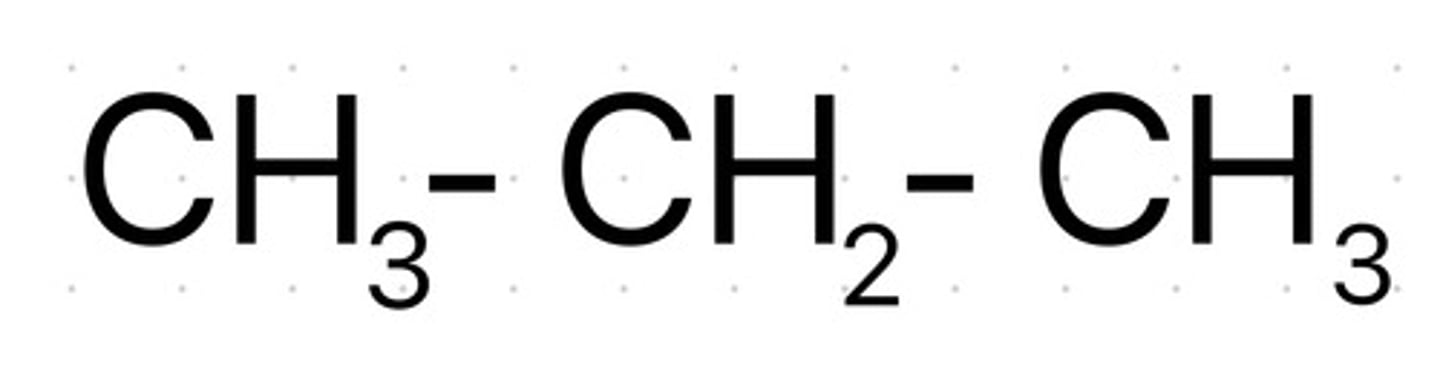
What is a skeletal formula?
A type of formula drawn as lines with each vertex being a carbon atom; carbon atoms are not drawn, and each C atom is assumed to have all unspecified bonds as C-H
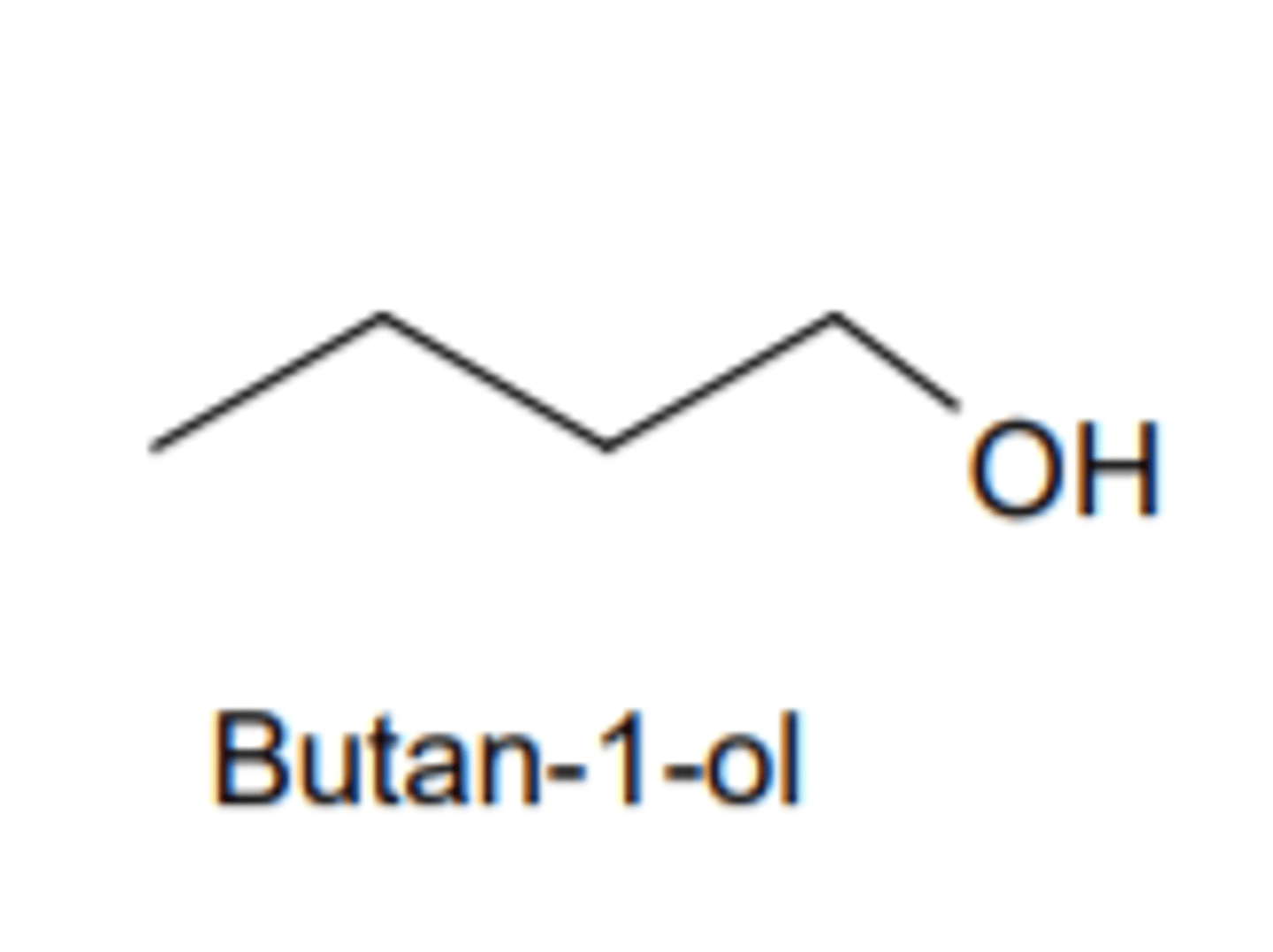
What is a homologous series?
A series of organic compounds having the same functional group but with each successive member differing by CH2
What is a functional group?
A group of atoms responsible for characteristic reactions of a compound
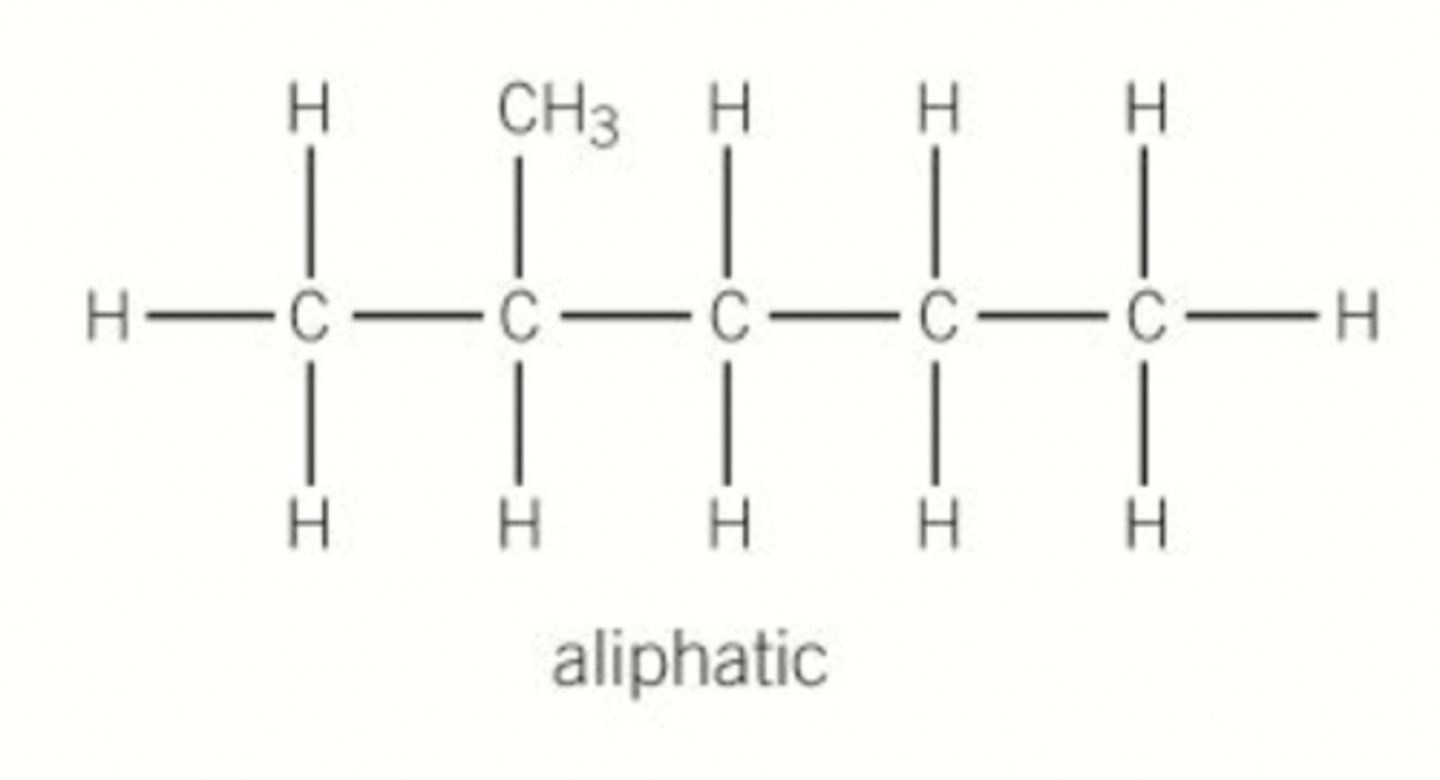
What is an aliphatic hydrocarbon?
Hydrocarbon with carbon atoms joined together in a straight line or branched chain
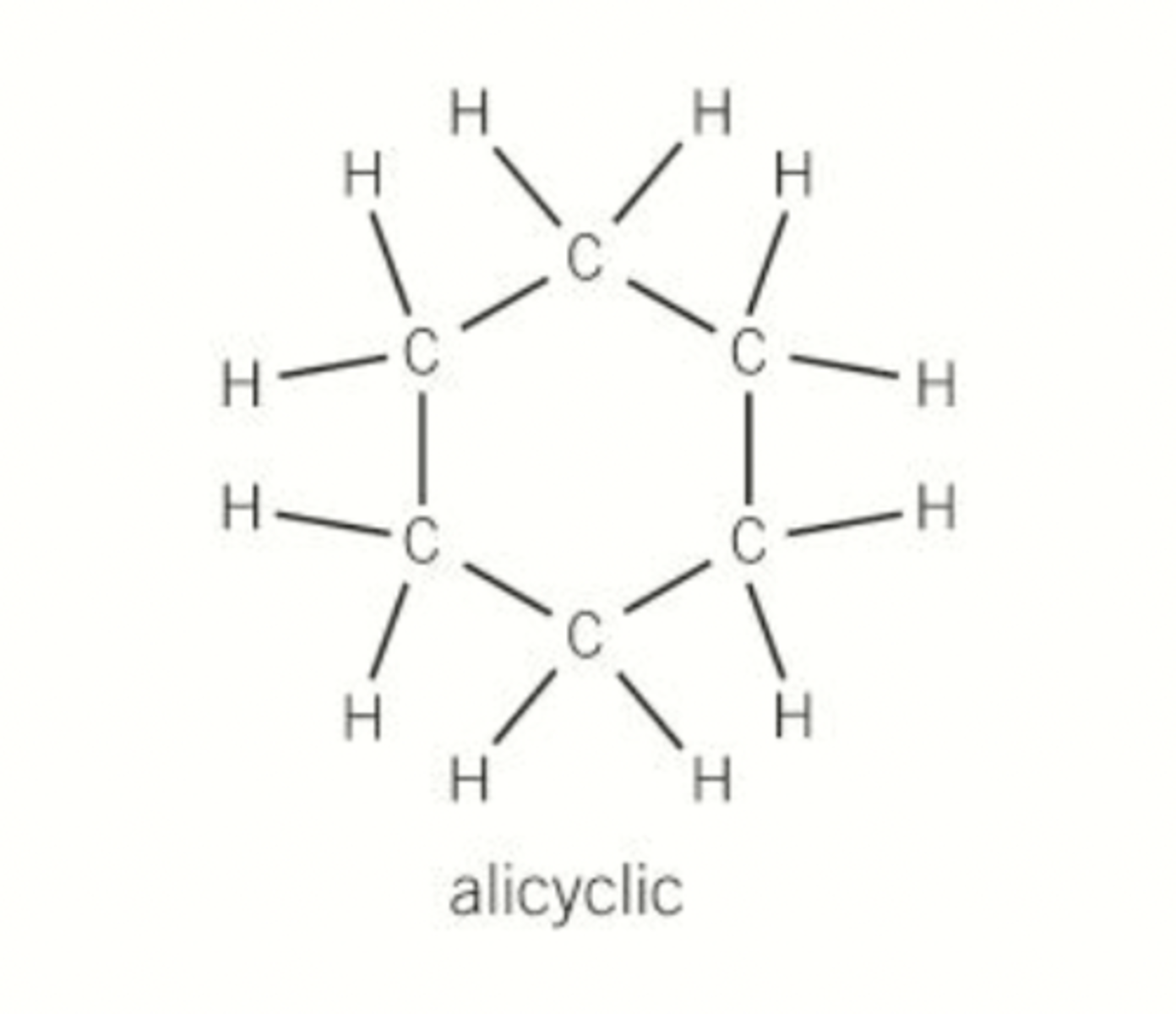
What is an alicyclic hydrocarbon?
Hydrocarbons arranged in non-aromatic rings with or without side chains
What is an aromatic hydrocarbon?
Hydrocarbon that contains at least one benzene ring

What are the suffixes for: a) No double bonds, b) At least one double bond, c) Alcohol, d) Aldehyde, e) Ketone, f) Carboxylic acid?
a) -ane, b) -ene, c) -ol, d) -al, e) -one, f) -oic acid
What are the prefixes for: a) CH3, b) C2H5, c) C3H7, d) C4H9, e) Cl, f) Br, g) I?
a) methyl-, b) ethyl-, c) propyl-, d) butyl-, e) chloro-, f) bromo-, g) iodo-
What is the general formula of alkanes?
CnH2n+2
What is the general formula of alkenes?
CnH2n
What is the general formula of alcohols?
CnH2n+1OH
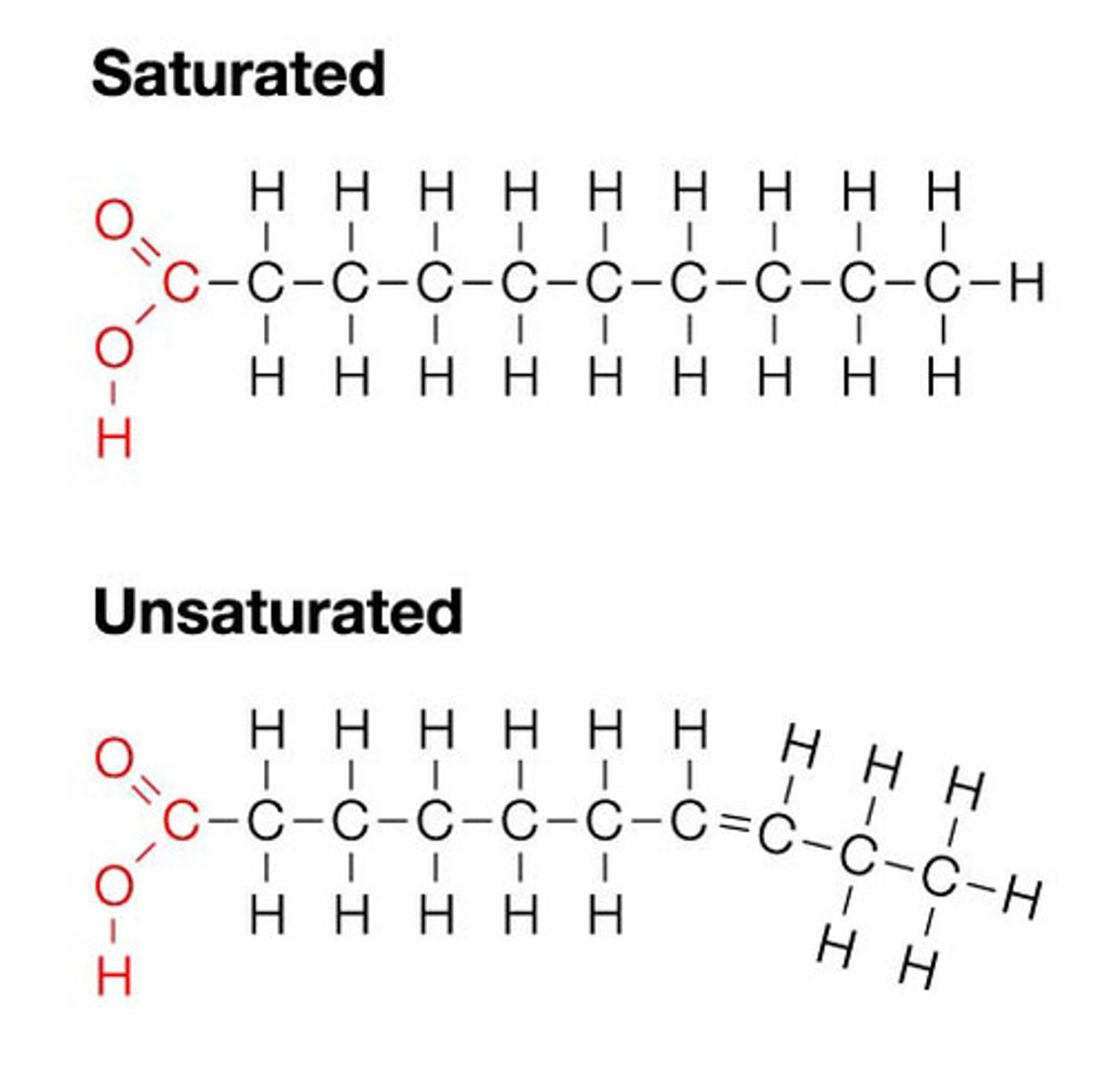
What does saturated mean?
Organic compounds which only contain single bonds
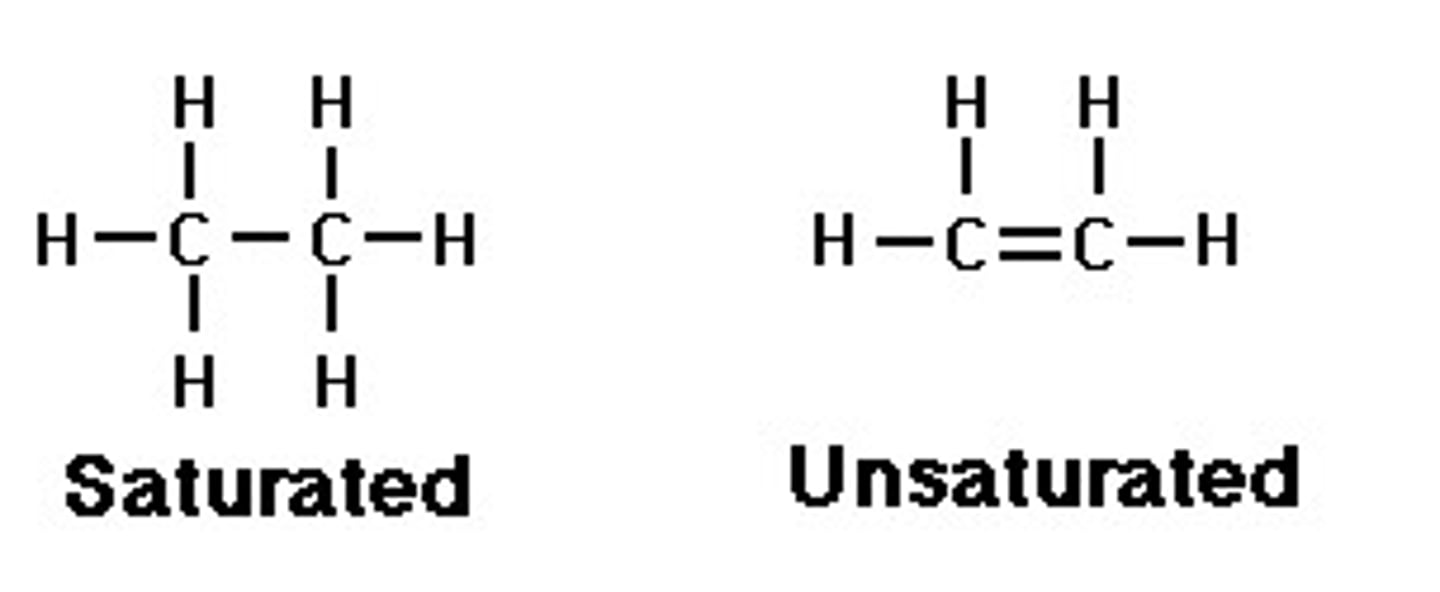
What are unsaturated compounds?
Organic compounds that contain at least one carbon-carbon double covalent bond
Define structural isomerism.
When molecules have the same molecular formula but different structural formula
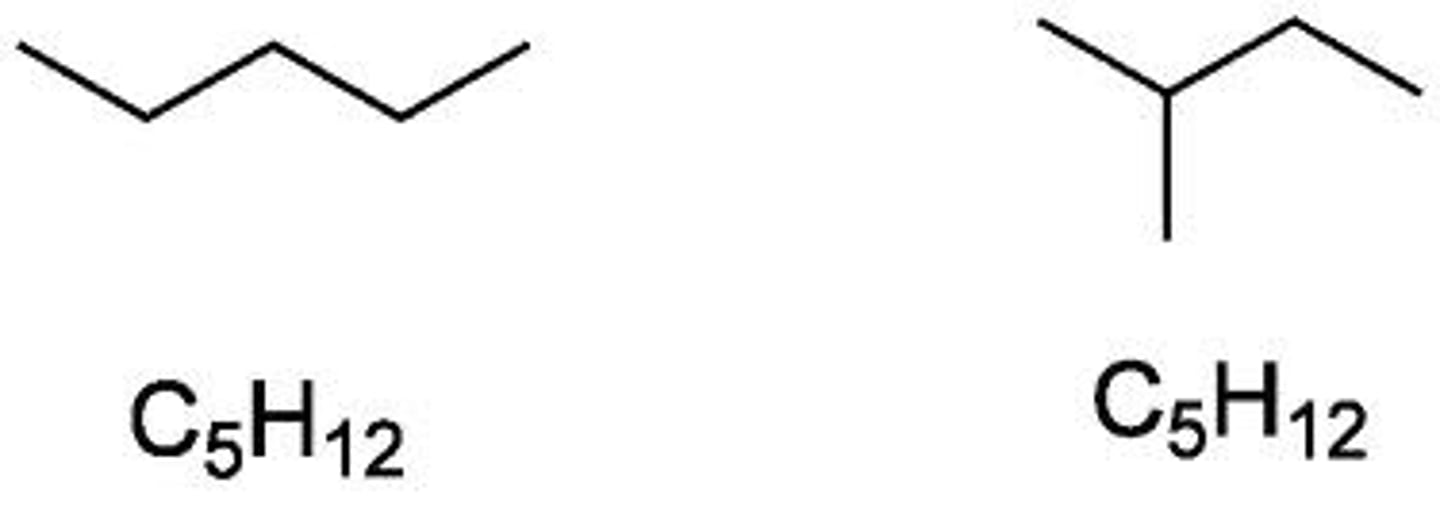
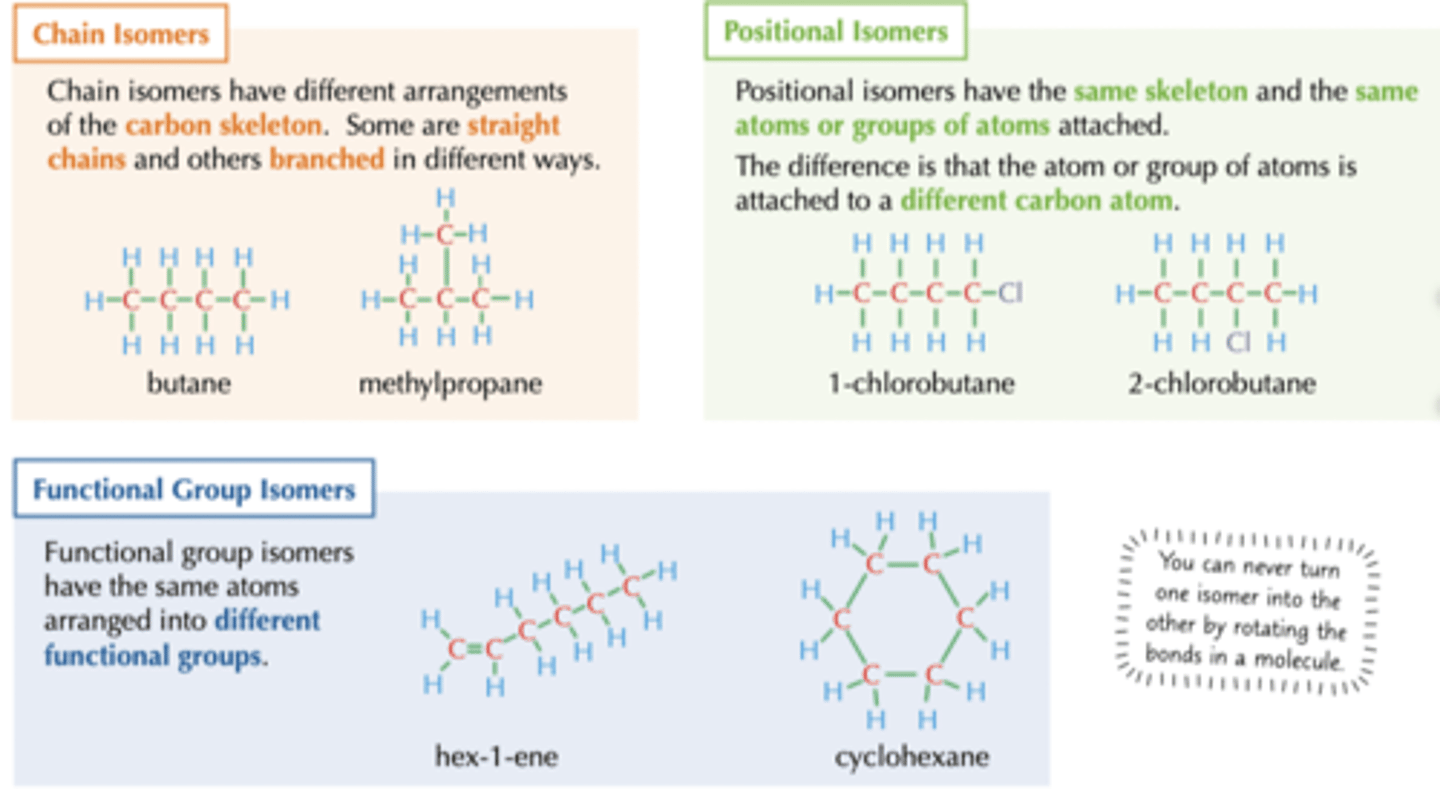
What are the 3 ways in which structural isomers can be formed?
1. different arrangement of carbon skeleton (chain)
2. Functional groups/atoms bonded to different carbons (positional)
3. Different functional groups (functional)
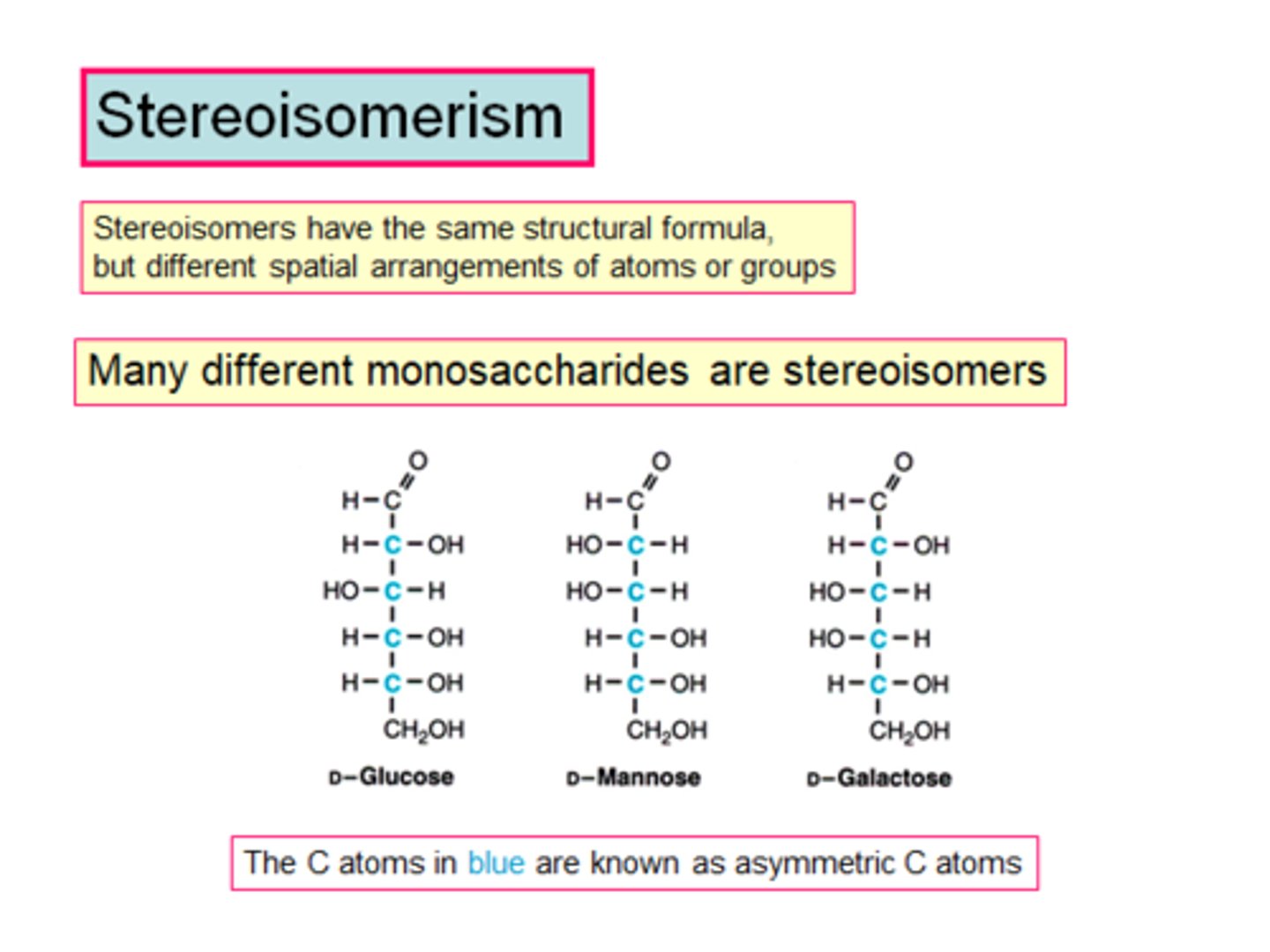
What are stereoisomers?
Organic compounds with the same molecular formula but different arrangement of atoms in space
What is E-Z isomerism and how are the isomers decided?
Caused by limited rotation about C=C bonds.
- Z isomer has high-priority substituents on the same side
- E isomer has them on different sides

What is Cis-trans isomerism?
A special type of E/Z isomerism where the two substituents on each carbon atom are the same
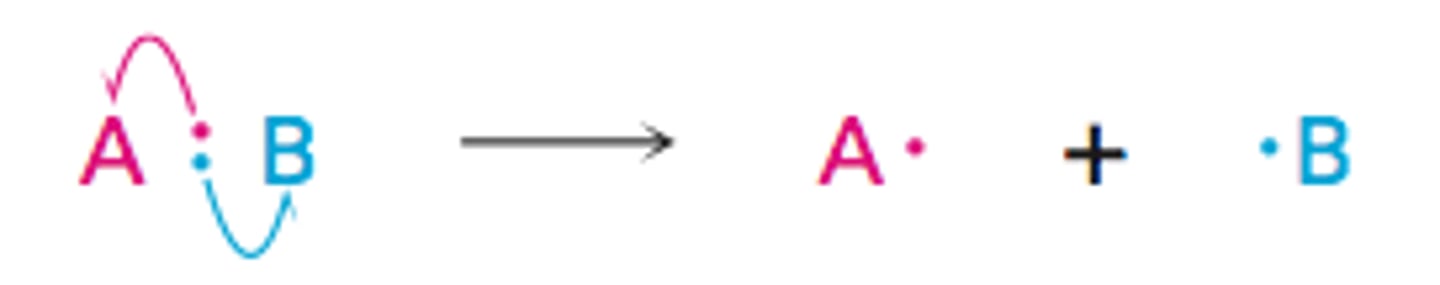
What is homolytic fission?
When each bonding atom receives one electron from the bonded pair, forming two radicals
What is heterolytic fission?
When one bonding atom receives both electrons from the bonded pair
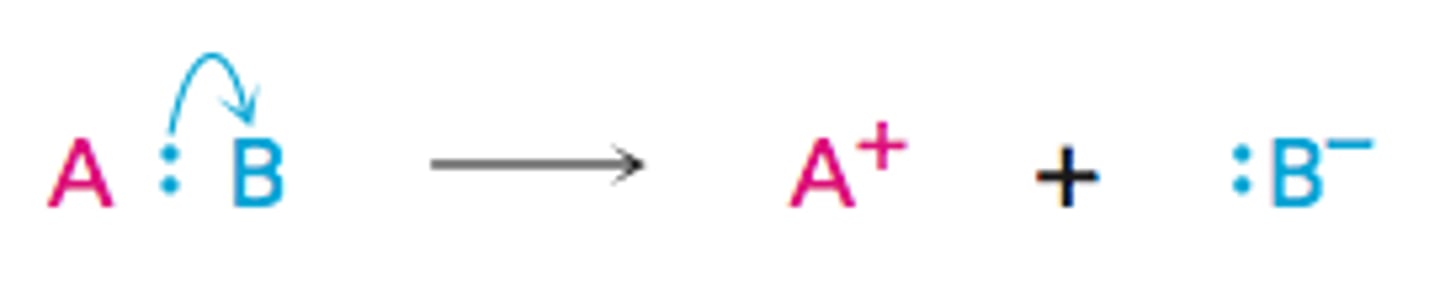
What are radicals?
Highly reactive, neutral species
How is a covalent bond formed from two radicals?
The radicals collide and the electrons are involved in bond formation