[CC] Drug Testing in the Philippines
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
80 Terms
R.A. 9165
_______OTHERWISE KNOWN AS “COMPREHENSIVE DANGEROUS DRUGS ACT OF 2002” IS THE LEGAL BASIS OF DRUG TESTING.
DSWD
THE POWER TO MONITOR AND REGULATE WAS GIVEN TO DOH WITH COORDINATION TO _____ AND OTHER AGENCIES (PDEA, ETC.)
DOH/BHFS
FORMULATE AND ESTABLISH THE STANDARDS FOR REGULATIONS OF HOSPITALS, CLINICS AND OTHER HEALTH FACILITIES
DOH/BHFS
ENFORCE THE STANDARDS THAT SHALL BE THE BASIS OF ISSUANCE OF LICENSE AND ACCREDITATION OF A FACILITY
DOH/BHFS
DEVELOP POLICIES AND GUIDELINES ON ESTABLISHMENT, OPERATION AND MONITORING OF DRUG TESTING LABORATORIES.
DOH/BHFS
CONDUCT ACCREDITATION OF ALL PRIVATE AND GOVERNMENT DRUG TESTING LABORATORIES.
DOH/BHFS
CONDUCT TRAINING OF DOH REGULATORY OFFICERS.
DOH/BHFS
CONDUCT RESEARCH AND DEVELOPMENT OF DRUG TESTING AND OTHER RELATED ACTIVITIES.
EAMC
DEVELOP COMPREHENSIVE PROCEDURAL AND SCIENTIFIC STANDARDS FOR ALL ASPECTS OF DRUG TESTING PROGRAM.
EAMC
PROMOTE QUALITY ASSURANCE PROGRAM IN ALL DRUG TESTING LABORATORIES.
EAMC
ASSIST DOH DESIGNATED AGENCIES IN THE CONDUCT OF ACCREDITATION OF CONFIRMATORY LABORATORIES FOR DRUG TESTING AND IN THE EVALUATION OF DRUG TEST KITS AND REAGENTS.
EAMC
RESOLVE CONFLICTING/CHALLENGE RESULTS AMONG CONFIRMATORY LABORATORIES
EAMC
TRAIN TECHNICAL STAFF OF DRUG TESTING LABORATORIES
EAMC
CONDUCT RESEARCH AND DEVELOPMENT OF DRUG TESTING AND RELATED ACTIVITIES.
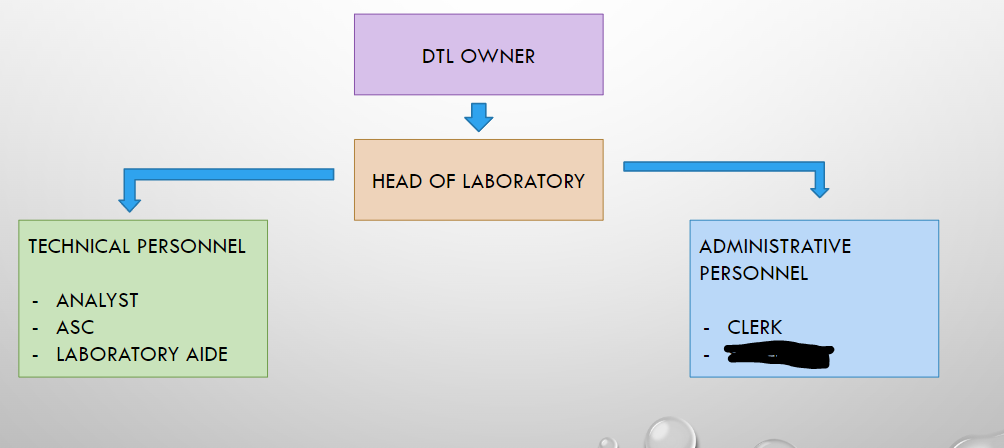
DTL OWNER
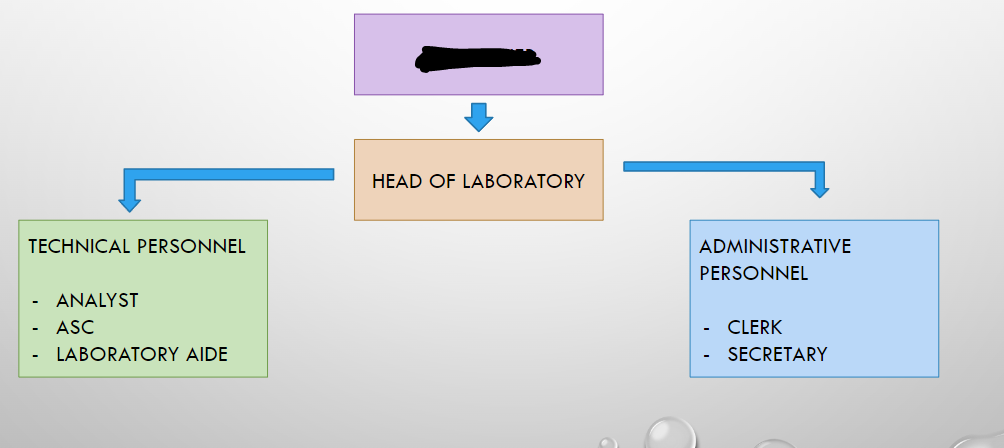
HEAD OF LABORATORY
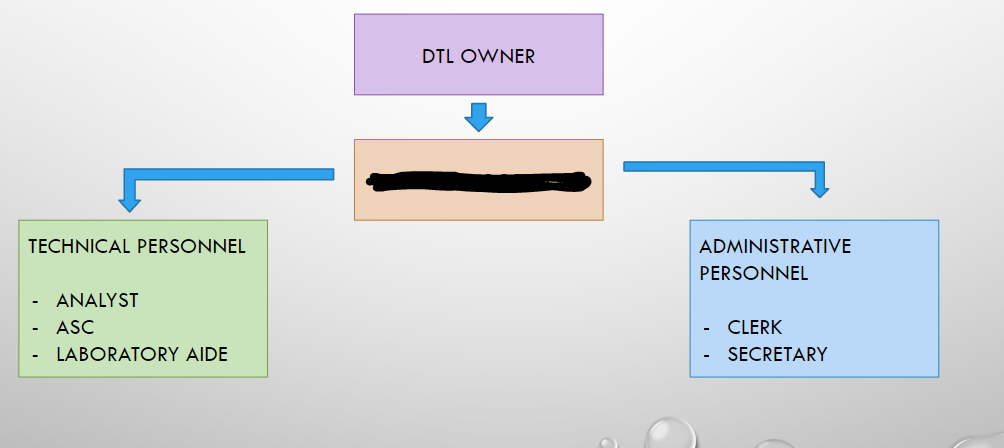
ANALYST
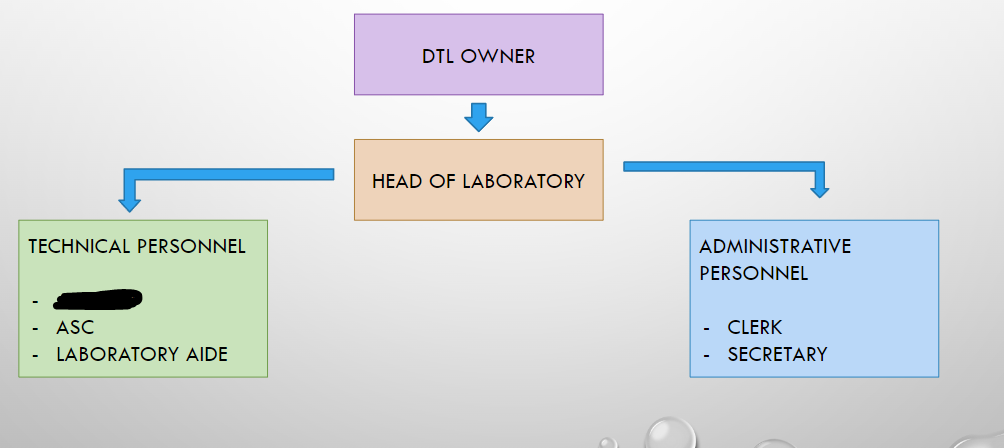
ASC
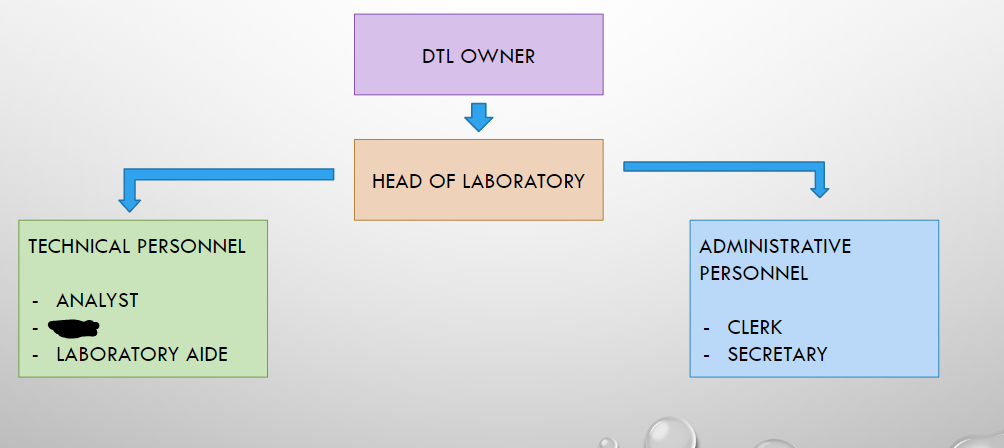
LABORATORY AIDE
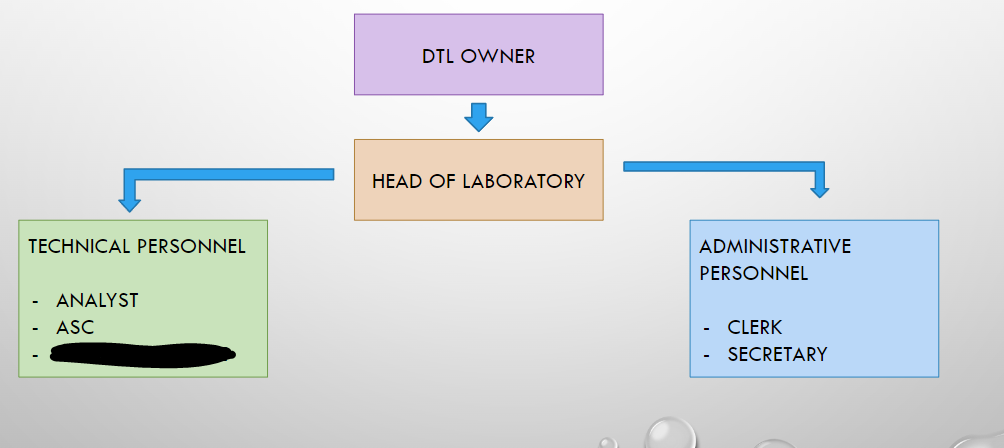
CLERK
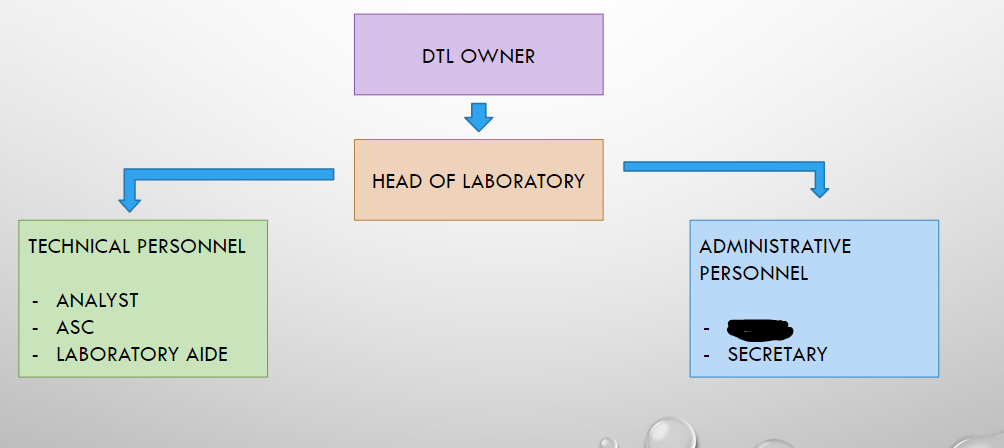
SECRETARY
SCREENING LABORATORY
A LICENSED PHYSICIAN CERTIFIED IN CLINICAL PATHOLOGY BY THE PHILIPPINE BOARD OF PATHOLOGY HEADS WHAT LABORATORY
SCREENING LABORATORY
A LICENSED PHYSICIAN TRAINED IN LABORATORY MANAGEMENT AND DRUG TESING OPERATION HEADS WHAT LABORATORY?
CONFIRMATORY LABORATORY
A LICENSED PHYSICIAN CERTIFIED IN CLINICAL PATHOLOGY BY THE PHILIPPINE BOARD OF PATHOLOGY WITH AT LEAST 2 YEARS OF ACTIVE LABORATORY EXPERIENCE IN ANALYTICAL TOXICOLOGY HEADS WHAT LABORATORY?
CONFIRMATORY LABORATORY
A LICENSED CHEMIST WITH MSTER’S DEGREE IN CHEMISTRY, BIOCHEMISTRY OR A BRANCH OF CHEMISTRY WITH AT LEAST 2 YEARS OF ACTIVE LABORATORY EXPERIENCE IN ANALYTICAL CHEMISTRY HEADS WHAT LABORATORY?
CHEMIST
CHEMICAL ENGINEER
MEDICAL TECHNOLOGIST
PHARMACIST
TO BE ANALYST THEY MUST BE REGISTERED
ANALYST
FINISHED THE TRAINING UNDER THE NRL AND MUST BE A DOH CERTIFIED DRUG ANALYST.
AUTHORIZED SPECIMEN COLLECTOR
THE DESGINATED PERON TO COLLECT SAMPLES WITHIN THE LABORATORY
AT LEAST HIGH SCHOOL GRADUATE
MUST HAVE UNDERGONE APPROPRITAE TRAINING.
AUTHORIZED SPECIMEN COLLECTOR MUST BE AT LEAST
LABORATORY AIDE
EACH PERSONNEL MUST HAVE THE EDUCATIONAL BACKGROUND APPROPRIATE FOR THE TASKS ASSIGNED.
THEORY AND PRACTICE OF THE DRUG TESTING PROCEDURES USED IN THE LABORATORY.
CHAIN OF CUSTODY, REPORTING AND RECORD KEEPING
REVIEW AND INTERPRETATION OF TEST RESULTS
QUALITY ASSURANCE PROGRAM
DANGEROUS DRUGS REGULATIONS AND POLICIES.
TRAINING REQUIREMENTS FOR HEAD OF LABORATORY
ANALYTICAL METHODS AND PROCEDURES.
MAINTENANCE OF CHAIN OF CUSTODY.
REVIEWING AND REPORTING TEST RESULTS.
PROPER REMEDIAL ACTION IN RESPONSE TO PROBLEM THAT MAY ARISE.
QUALITY CONTROL PROCEDURES AND PRACTICES
DANGEROUS DRUGS REGULATIONS AND POLICIES
TRAINING REQUIREMENTS FOR ANALYST
COLLECTION PROCEDURE FOR EACH TYPE OF SPECIMEN
CHAIN OF CUSTODY AND RECORD KEEPING
SPECIMEN INTEGRITY AND SECURITY
DANGEROUS DRUGS REGULATIONS AND POLICIES
TRAINING REQUIREMENTS FOR ASC
THE COLLECTION PROCEDURES CHANGES SIGNIFICANTLY.
THE ASC MADE A MISTAKE THAT CAUSED A TEST TO BE CANCELLED.
RETRAINING ASC SHALL BE REQUIRED UNDER THE FOLLOWING CONDITIONS
EMPLOYER OF THE CLIENT/DONOR/SUBJECT.
INVESTIGATOR AT THE CRIME SCENE
COMPLAINANT
OWNER/ADMINISTRATOR OF ESTABLSIHMENT
THE FOLLOWING PERSONS ARE NOT AUTHORIZED TO COLLECT SPECIMEN
SCREENING
AT LEAST HAVE 20 SQUARE METERS IS FLOOR AREA AND 10 SQUARE METERS WORK AREA AND MUST HAVE EXHAUST FAN, SINK AND STORAGE CABINET.
CONFIRMATORY
AT LEAST 60 SQUARE METERS FLOOR AREA AND 30 SQUARE METERS WORKING AREA WITH EXHAUST FAN, SINK, FUME HOOD, STOCK ROOM AND INSTRUMENTATION ROOM
BLOOD
10 ML CAPACITY PLAIN TEST TUBE.
TISSUE
SPECIMEN IMMEDIATELY FROZEN AND TRANSPORTED PLACED IN A CLEAN, DRY, TIGHTLY CAPPED PLASTIC CONTAINER WITH NO ADDITIVES.
SCALP HAIR
SELF-SEALING TRANSPARENT PLASTIC BAG, 200 MG CAPACITY
REASONABLE SUSPICION/CAUSE
A DRUG TESTING LABORATORY MAY COLLECT: BLOOD
REASONABLE SUSPICION/CAUSE
A DRUG TESTING LABORATORY MAY COLLECT: FINGERNAILS
PRE-EMPLOYMENT, RANDOM, REASONABLE SUSPICION/CAUSE
A DRUG TESTING LABORATORY MAY COLLECT: SALIVA (ORAL FLUID)
PRE-EMPLOYMENT, RANDOM, RETURN TO DUTY, FOLLOW-UP
A DRUG TESTING LABORATORY MAY COLLECT: SCALP HAIR
RETURN TO DUTY, FOLLOW-UP
A DRUG TESTING LABORATORY MAY COLLECT: SWEAT
REASONABLE SUSPICION/CAUSE
A DRUG TESTING LABORATORY MAY COLLECT: TISSUE
PRE-EMPLOYMENT, RANDOM, REASONABLE SUSPICION/CAUSE, MANDATORY
A DRUG TESTING LABORATORY MAY COLLECT: URINE
MINIMUM OF 5ML
HOW MUCH OF BLOOD
TO BE DETERMINED #1 [QUANTITY]
HOW MUCH OF FINGERNAILS
100 MG HAIR
HOW MUCH OF SCALP HAIR
TO BE DETERMINED #2 [QUANTITY]
HOW MUCH OF TISSUE
2 ML COLLECTED AS A NEAT SPECIMEN
HOW MUCH OF SALIVA
1 PATCH WORN FOR 7 TO 14 DAYS
HOW MUCH OF SWEAT
60 ML IN SINGLE CONTAINER OR 30 ML IN TWO SEPARATE CONTAINERS.
HOW MUCH OF URINE
SEPARATE THE SERUM IMMEDIATELY AND FREEZE THE SAMPLE AFTER USE.
HOW TO HANDLE AND STORAGE OF BLOOD
TO BE DETERMINED #1 [STORAGE]
HOW TO HANDLE AND STORAGE OF FINGERNAILS
STORED AT COOL AND DRY PLACE.
HOW TO HANDLE AND STORAGE OF SCALP HAIR
MACERATED AND FROZEN
HOW TO HANDLE AND STORAGE OF TISSUE
DEEP-FROZEN AT LEAST -8 TO -10 DEGREE CELSIUS.
HOW TO HANDLE AND STORAGE OF SALIVA
TO BE DETERMINED #2 [STORAGE]
HOW TO HANDLE AND STORAGE OF SWEAT
PROLONGED STORAGE AT -20 DEGREE CELSIUS. (2 – 6 FOR NO MORE THAN 1 DAY)
HOW TO HANDLE AND STORAGE OF URINE
UNIQUE ACCESSION NUMBER
THE LABORATORY MUST HAVE A _________ UPON ENTRY OF THE SPECIMEN TO THE LABORATORY.
CCF FORM
INSPECT THE SPECIMEN SUBMITTED AND THE _________ TO VERIFY THE INTEGRITY AND IDENTITY OF THE SPECIMEN.
IMMUNOASSAY
THESE METHODS ARE USED FOR PRELIMINARY SCREENING, BASED ON ANTIGEN-ANTIBODY REACTION. SMALL AMOUNTS OF THE DRUG AND/OR METABOLITES MAY BE DETECTED.
CHROMATOGRAPHY
SEPARATION OF MIXTURE IN THE MAIN OUTCOME OF THIS METHOD. IN THIS PROCESS, A MIXTURE OF SUBSTANCES IS SEPARATED IN A STATIONARY MEDIUM.
HYPHENATED TECHNIQUE
COMBINATION OF TWO SOPHISTICATED TECHNOLOGIES
VALIDITY TEST
__________ IS A TEST TO DETERMINE THE INTEGRITY OF THE SPECIMEN.
OBSERVED
UNOBSERVED
THERE ARE TWO TYPES OF URINE SAMPLES
ADULTERATED
SUBSTITUTED
DILUTED
IMPROPERLY COLLECTED, HANDLED AND STORED
IMPROPERLY DOCUMENTED
INVALID URINE SAMPLES
COC
CHAIN OF CUSTODY
MEMORANDUM FOR RECORDS
IT IS A RECORD TO DOCUMENT SPECIMEN THAT HAD BEEN REJECTED OR CANCELLED. IT INCLUDES REASON AND CORRECTIVE MEASURES DONE.
Integrated Drug Test Operation Management Information System
IDTOMIS
2
THE LABORATORY MUST RETAIN ALL RECORDS GENERATED TO SUPPORT TEST RESULTS FOR AT LEAST _____ YEARS.
INDEFINITELY
HOWEVER, POSITIVE RESULTS ARE STORED _________
THE CLIENT MUST GET HIS/HER OWN RESULTS
LABORATORIES MUST STRICTLY ADHERE TO CONFIDENTIALITY OF THE RESULT AND MAINTAIN THE COC OF THE TESTING. #1
THE CLIENT MUST INFORM THE LABORATORY IN ADVANCE
LABORATORIES MUST STRICTLY ADHERE TO CONFIDENTIALITY OF THE RESULT AND MAINTAIN THE COC OF THE TESTING. #2
THE AUTHORIZED PERSON MUST HAVE A GOVERNMENT ISSUED I.D., AUTHORIZATION LETTER FROM THE CLIENT AND A COPY OF THE CLIENT’S I.D.
LABORATORIES MUST STRICTLY ADHERE TO CONFIDENTIALITY OF THE RESULT AND MAINTAIN THE COC OF THE TESTING. #3
INTERNAL QC
PERFORMED BY USING A POSITIVE AND NEGATIVE CONTROL SAMPLES/KITS PROVIDED BY THE MANUFACTURER OR SUPPLIER PER BATCH/BOX OF TEST KITS IN SDTL.
EXTERNAL QC
GIVEN BY THE NRL/PROFICIENCY TEST
7
NEGATIVE URINE SAMPLES ARE RETAINED FOR _____ DAYS BEFORE DISPOSAL.