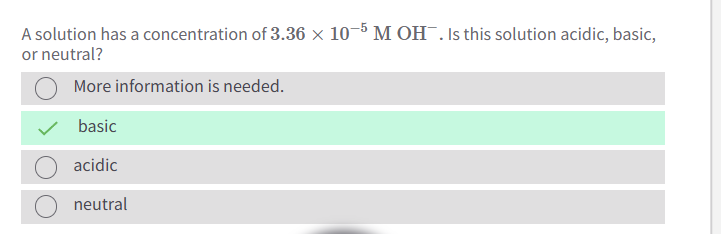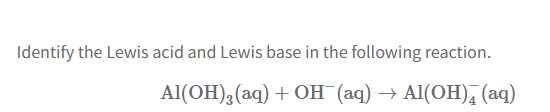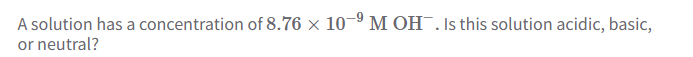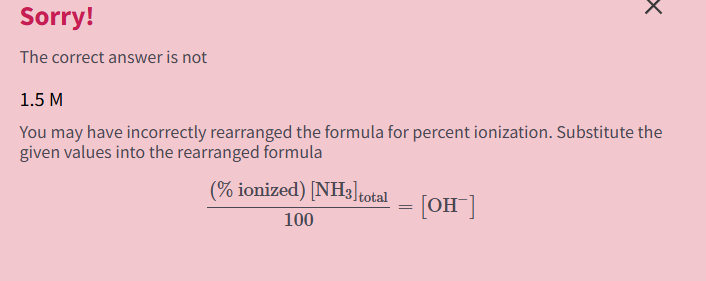Chem exam 2 chapter 16
1/103
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
104 Terms
What is an Arrhenius acid?
What is an Arrhenius base?
Arrhenius acid = Increases H+ conc. in solution
Arrhenius base = Decreases H+
Hydronium is same as proton
H3O and H+ yes
H+ and H2O = H3O
Whats important to note about id’ing strong vs weak acids?
Strong acids go to completion with —> arrow
weak acids don’t, they’re reversible with ←—> arrow

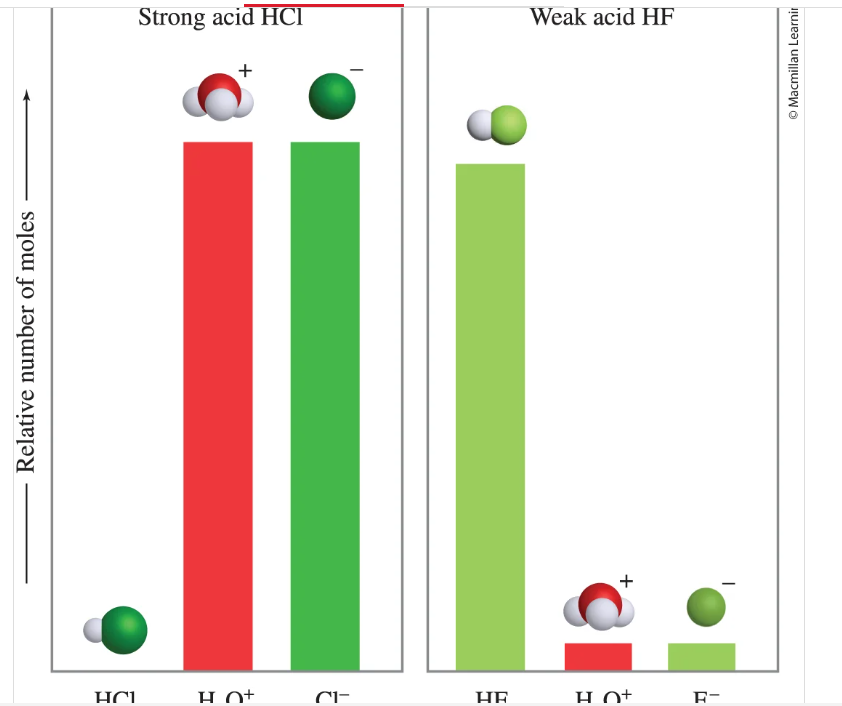
Given the same # of initial moles of a Strong acid and a weak acid, which will produce more hydronium H3O+ ions?
Strong acid will produce significantly more hydronium ions
What are the 7 strong acids and which ones sometimes considered a weak acid?
HClO4
HClO3 - sometimes a WA
H2SO4
HNO3
HCl
Hbr
HI


What are the strong bases?
Ba(OH)2
Sr(OH)2
Ca(OH)2
CsOH
RbOH
NaOH
KOH
LiOH
(Bsc)² CRNKL
bitch crinkl
What’s a very common example of a weak base?
Anything that is a derivative of NH3 , so NH3, CH3NH2, or anything that has a N surrounded by H’s or CH’s
weak bases don’t contain OH, but when they ionize they produce OH by taking H+ from water so remember that
whats rule for weak bases just like weak acids
won’t ionize completely, reversible and at EQ
Given same number of moles of a dissolved strong base, and a dissolved weak base, which will produce more hydroxide ions?
A strong base
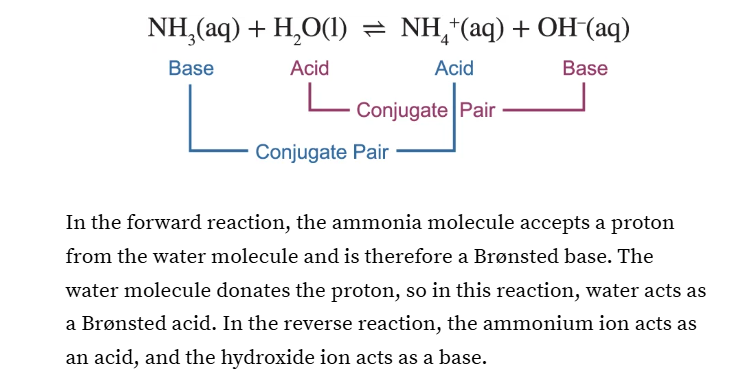
What is a bronsted acid?
What is a bronsted base?
Bronsted acid is a proton donor that releases H+
Bronsted base is a proton acceptor that makes bonds with H+
bronsted acid + base example
Why is water unique under the bronsted base definition
If H2O gets paired with a strong base, it’ll act as an acid
If H2O gets paired with a strong acid, it’ll act as a base
This makes it amphoteric
What’s acid and base strength?
What’s relationship?
acid strength = extent acid reacts with water to form hydroniums or H+ ions
base strength = extent base interacts with water to form ions
The stronger the weak acid or base, the weaker its conjugate base or acid
The weaker the weak acid or base, the stronger its conjugate base or acid


A solution of water is neutral because it contains an equal concentration of
Hydronium (H3O) and Hydroxide (OH-) ions


ID if it’s a strong acid or base. If it starts with a H it’s an acid.
ID if it’s a strong base,
Write out the equation with your molecule reacting with water, that will make it make more sense,
When you write out the bases, the ones that produce the MOST hydoxide (OH) will produce the least hydronium (H3O ions)
A weak base > strong base for producing H3O
Most Hydronium
Strong acid > Weak acid > weak base > strong base (with less moles) > strong base (with more moles, i.e (OH)2)

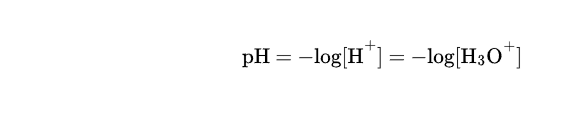

For C, this is finding pH given a base

How to calculate pH of a solution using pOH
1. find pH value using -log(value)
Subtract that value from 14
answer
A larger Ka value for ionization of a weak acid indicates
more complete ionization
i.e Ka = 1.8e-5 > 5.8e-10, even tho both are weak acids



Calculate pH by doing 14 - pOH (8.84) and you get 5.16
Calculate the OH- ions by doing (OH-) = 10-pOH
Calculate [H+] ions by doing [H+] = 10-pH

Look at the acid, if it’s strong or weak. Strong means a full disassociation so it’ll match
Assign the directly known value
Use Kw = 1e-14 to get other ion
Calculate pH/pOH
When a problem says “placed in” that means those are the……!!!
initial values

You’re calcing Qc here and comparing to value of 15 to see which direction it will move


You solve this without using B because B’s a solid.

For ICE tables, what does X represent? (example of A and B reactants
The amount of A, B, required to get the initial amount of A and B to its equalibrium concentration following reactions stochiometry
![<p>If K is very small, and you end up with an equation where you CANNOT use quadratics to solve like</p><p></p><p>K = [B]<sup>3</sup>/(A) (can’t solve this because you end up with x<sup>3</sup> + bx + c)</p><p></p><p>What do you do using the example problem shown?</p><p></p>](https://assets.knowt.com/user-attachments/f4e804a0-902d-49b9-9aed-c52596305335.png)
If K is very small, and you end up with an equation where you CANNOT use quadratics to solve like
K = [B]3/(A) (can’t solve this because you end up with x3 + bx + c)
What do you do using the example problem shown?
Set up Ice table
Write the K expression
IF THE K IS VERY SMALL VALUE, REMOVE THE X ONLY FROM THE DENOMINATOR
Now you will be able to easily solve for X, and then can plug it back into the ICE table to solve.
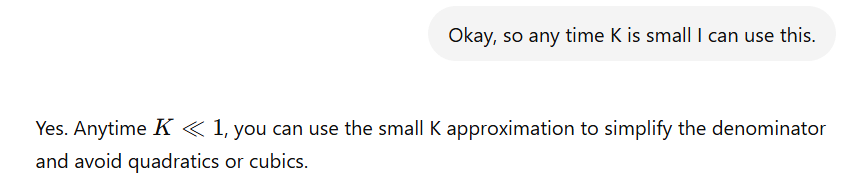
If given A + 2B —> 3C
and 3C has an initial value of 0.5M
When you make the ICE table, do you cube the 0.5M before putting it into the initial value?
NO, only when solving for Kc.
For the following
4 + 4×2 + -18x = (0.711 + 2x)2
How do you isolate for 0 to get the quadratic equation on right side?
remember a² + 2ab + b²
do 0.711²
then do 2 × 0.711 × 2x
then do b² which is 2x² —> 4x² (for this you’re basically just squaring the whole number while leaving the exponet on X, i.e (5x)² —> 25x²
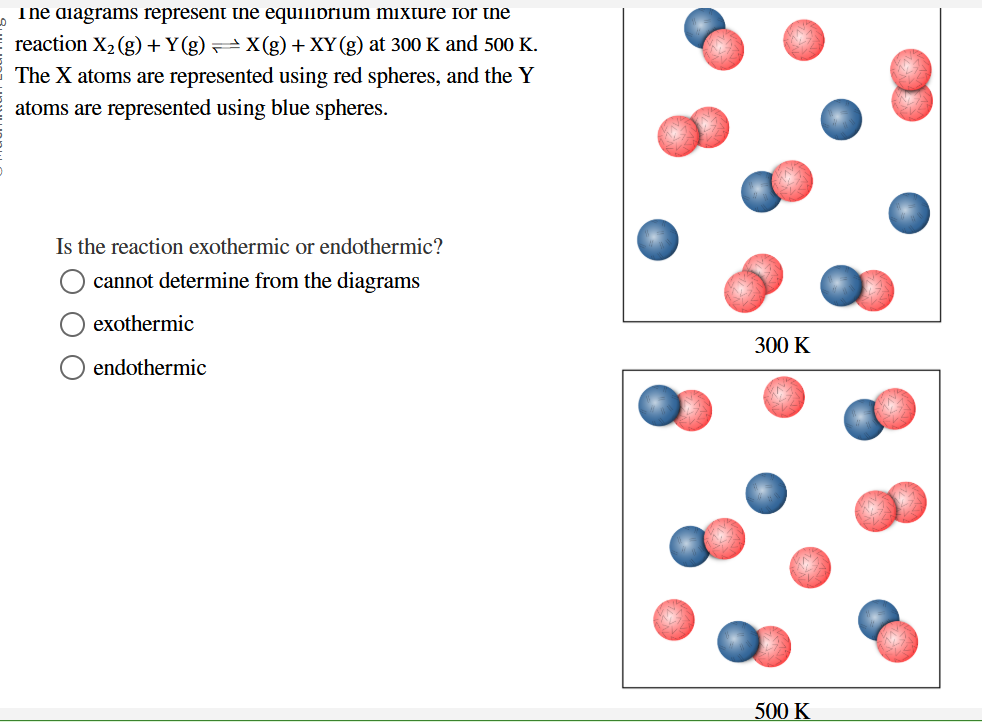
Temp + = Endothermic


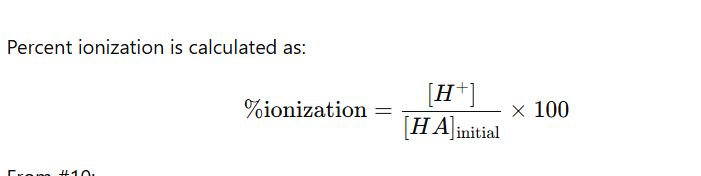
For percent ionization / percent error problems it will be
H+/initial HA concenentration
x 100

PLEASE ALWAYS WRITE THEM OUT THIS WAY IT WILL SIMPLIFY THINGS
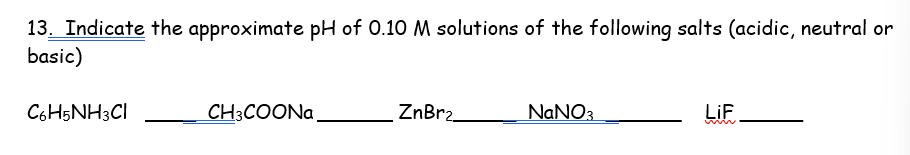
You can do this man, you just need to remember which are strong acids and which are strong bases, then do addition of both to determine if its acidic, basic, or neutral
Split the component into 2 based on the element at the end
i.e Znbr —> Zn+ Br-
The CATion will come from the base, while the ANION will come from an acid, this is why you need to remember the strong acids and strong bases so you know which element is similar to one of those strong acids and bases!
Zn is a cation, weak base, the reason it’s weak is because Zn isn’t found in any of the strong bases, Br comes from a strong acid
Add the effects (you’ll have to memorize these below)
Strong base (Ba, Sr, Ca, Cs, etc) : Neutral doesn’t affect pH
Weak base (NH4, C6H5NH3+) Acidic, lowers pH
Small metals: Zn, Al: Slightly acidic
Strong acid: Cl, Br, I : Neutral
Weak acid: F-, CH3COO, CN, HNO2, H2CO3: Basic : raises pH
Combine the effects of Zn (slightly acidic and Br-) neutral means the overall reaction is acidic

![<p>So the <strong>shortcut</strong>:</p><ul><li><p>Strong acid → [H₃O⁺] = initial</p></li><li><p>Strong base → [OH⁻] = initial</p></li><li><p>Only use Kw if you need the <strong>other ion</strong>.</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/743f1f12-e18b-494e-aa7d-05f6d688b54c.png)
So the shortcut:
Strong acid → [H₃O⁺] = initial
Strong base → [OH⁻] = initial
Only use Kw if you need the other ion.
This is an equation where you need kw, you know this because you’re given a Strong base (or if given a strong acid same idea), the (M) for the strong base will be the same for its relevant group (OH) , and you need to use kw to solve for the H3O group, it’s opposite ion

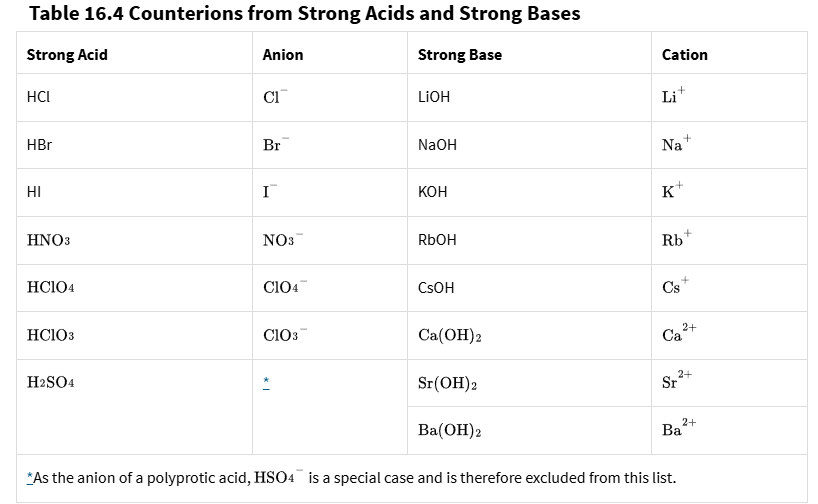
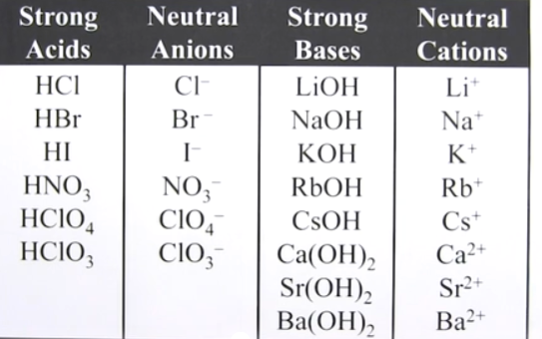
Remember this table
Strong base component (i.e Na+) + strong acid component (i.e Cl-)disassociating forms
a basic solution

ID the cation and Anion
ID if any component is a part of a strong acid or base
if you have a component thats a strong acid or base, regardless cation or anion, its neutral
If you have a component thats a weak base, i.e NH4, it’s acidic
If you have a component thats a weak acid, i.e PO4, its basic
example


For pH predictions like the following problem, what do we do, and what should we ignore?
It’s just like before, write out and disassociate the element.
If it’s a component of a strong acid or base, it will be Neutral.
JUST REMEMBER THESE
if it’s Fe³⁺, Al³⁺, Cr³⁺, Co³⁺
Cu²⁺, Zn²⁺, Ni²⁺, Pb²⁺, Sn²⁺
Be²⁺, Cr³⁺, Fe³⁺, Al³⁺
These will raise the pH and make it more basic
ignore water

Ranking questions
Calculate Kb for all of them
List the Kb’s
Rank the Kb’s from strongest to weakest (the one closest to 0 is the strongest with the lowest exponet i.e 1e-2 > 1e-4
ka x kb = kw
where ka is given
and kw = 1.00e-14

Write out disassociatoion
Look at the strength of the cation and anion
Cation (CH3NH3) - from a weak base = acidic
Anion (Br) - from a strong acid (neutral)
Solve for Ka using Kb + Kw
do Ka = Products/reactants (not including water)
Make an Ice table for the disassociation to get X
Solve for X using Ka
Remember you can simplify if Ka is very small
Once you solve for X, plug it into equation for H3O conc in ice table
You can use the H3O conc to find pH by doing the negative log one
****IMPORTANT
ANY COMPONENT OF A STRONG ACID OR BASE HAS NO EFFECT ON PH, SO WHEN YOU DO STEP 1 AND WRITE IT OUT, DON’T INCLUDE THE STRONG ACID OR BASE
i.e NaClO
Na + Cl is neutral
However NaClO is
Na+ ClO where Na doesn’t have an effect on pH
Therefore you write
ClO- + H2O ←→ HClO- + OH
ALSO VERY IMPORTANT
THE REASON you bother to identify them as cations and anions (cations = base) (anions = acids) is because it helps you to know whether to use Ka or Kb to solve.
For example in NaC2H3O2, Na is a strong base +, and neutral so disregard
C2H3O2 must be an anion acid, however its not a strong acid. its a weak acid, therefore it will increase pH and it will form an OH on the other side. This tells you YOU MUST USE KB, BECAUSE THE ONE YOU’RE SOLVING FOR IS KB.
this is important because if you set up the written out equation wrong, you’ll end up using wrong k value and your shit will be wrong.
Thats why sometimes you have to solve for Kb when given Ka, because the thing you’re trying to solve for (C2H3O2) is increasing pH, so its a weak acid acting as a base.
so in summary
ID the cation (base) and the anion(acid)
ID the strength of base and acid
if the acids weak, then you’re gonna use kb to solve because pH will increase. If the base is weak you’re gonna use ka to solve because the pH will decrease
set up ice table
solve for X
solve for pH

For acids with same central atom, the more oxygens attatched, the stronger it is

Rewrite each acid to id the alpha carbon
HC₂F₃O₂ → CF₃COOH
HC₂Cl₃O₂ → CCl₃COOH
HC₂BrH₂O₂ → BrCH₂COOH
HC₂H₃O₂ → CH₃COOH
Alpha carbon is always written in the middle, attatched to its RIGHT is a COOH group, and attatched to either its left, or its right is extra
Look at the extras, count the ones that are NOT hydrogens
F > Cl > Br > H
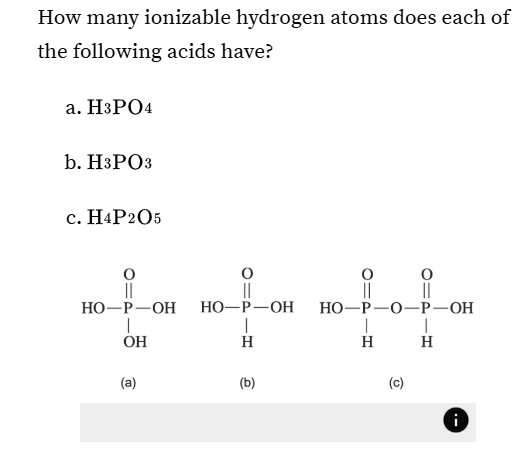
How many ionizable hydrogen atoms does each of the following acids have?
just count hydrogens that are bound to oxygens
for 1, there’s 3 oxygen bound to hydrogens
etc.
You might have to write them out
What’s a Lewis base?
What’s a Lewis acid?
Lewis Base = Electron pair doner
Lewis acid = Electron pair acid
How to tell if a Lewis Acid, Lewis Base, or Neither?
if lone pairs = lewis base
if no lone pairs, but + = lewis acid
If neutral and no lone pairs = neither
An acid and a base that differ by only one proton are called
conjugate

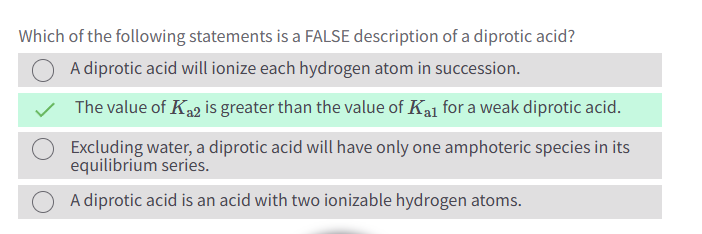


As the concentration of a weak base in water increases, what happens to percent ionization?
Decreases
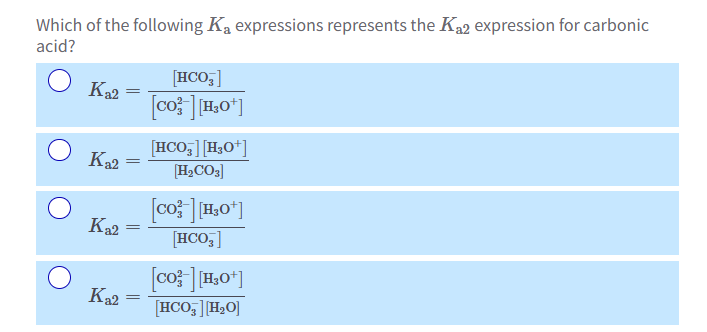

How to ID conjugate acid-base and conjugate base-acid
Conjugate acids and base pairs only differ by 1 hydrogen!