NST103_Part4_Lecture5_ROS_VitaminC_and_E
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
what are Reactive oxygen species ROS
hydroxide radical>> superoxide >> hydrogen peroxide
formed by 1e- transfer reaction from water redox state to oxygen redox state
damage DNA, enzymes, and membranes
used to kill microbes by neutrophils
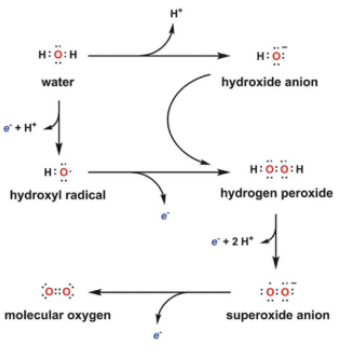
hydroxyl radical
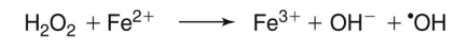
formed from Fe2+ and H2O2 during fenton reaction
why cells try to keep low Fe2+ and H2O2
Fe2+ + H2O2 —> Fe3+ + OH- + OH•
very reactive!
superoxide
O2•– can form upon 1e reduction of O2 by FADH2 or Fe2+
reacts with certain enzymes selectively
FeS cluster susceptible to damage by O2• and produce dangerous Fe2+ as product
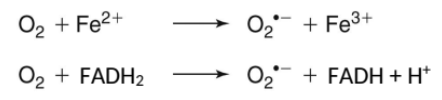
inactivation of FeS cluster containing enzymes is why E. coli/yeast cannot grow at high [O2]
hydrogen peroxide
formed from many sources (enzymes, O2•- detoxification
least reactive ROS
fenton reaction
ROS defense systems in cells
superoxide dismutase (SOD)*
catalase (Cat)*
glutathione peroxidase (GPX)*
peroxiredoxin (PRDX)*
vitamin C, E
vitamin C deficiency
scurvy
bleeding under skin, joint pain, corkscrew hair growth
caused by disfunction of collagen hydroxylases in absence of vitamin C
vitamin C synthesis ( animals vs humans )
can be synthesized by mammals
humans are missing only 1 enzyme for vitamin C biosynthesis: L-gulonolactone oxidase (GULO)
also absent in other primates that eat vitamin C rich fruits
vitamin C transport across cell membranes
ascorbic acid is absorbed through sodium-coupled transporter SVCT1/2
dehydroascrobic acid (DHA) is absorbed through diffusion driven glucose transporters (GLUT1/3)
DHA reduced to ascorbic acid inside cells
vitamin C absorption
absorption is saturable due to limited expression of transporters in enterocytes
taking more vitamin C does not increase blood Vit C after a certain threshold
vitamin C distribution across tissus
levels are 10x higher inside cells compared to blood, driven by sodium-coupled intake
concentration varies across tissues
vitamin C functions
ROS scavenger
required in catalytic amounts for repair of iron oxygenases
collagen biosynthesis
DNA demethylation
histone demethylation
co substrate for several copper oxygenase enzymes
conversion of Fe3+ to Fe2+ to facilitate Fe transport across membranes
vitamin C mononuclear iron oxygenases
use oxygen to hydroxylate substrates (add hydroxyl group)
enzyme contains Fe coordinated with His, Asp, and a-KG
rxn mech similar to CyP450 enzymes, but Fe bound to heme in Cyp450 instead of directly coordinated with other res (his, Asp)
ascorbate needed to reduce Fe3+ —> Fe2+
vitamin C reactivation of enzymes stuck in FeIV=O state
mononuclear iron oxygenases can get stuck in FeIV=O state without a substrate present
ascorbate serves as substrate to recycle enzyme back to Fe2+ active state
collagen hydroxylases and vitamin C
vit C required to reactivate enzyme that hydroxylates proline and lysine res on collagen
scurvy caused by disfunction of collagen hydroxylases in absence of vitamin C
function of collagen hydroxylase
collagen: most abundant animal protein, main protein in connecitve tissue
proline is second most abundant AA in collagen, and most prolines are hydroxylated
hydroxylated lys allow for attachment of oligosaccharides
Pro and Lys hydroxylation essential for producing functional collagen chains
HIF1α
hypoxia inducible factor 1 alpha
transcription factor whose levels are regulated by [O2]
activates transcription of genes required for survival under low [O2]
glycolytic genes for fermentative ATP prodiction
EPO for increased RBC production
histone demethylases
epigenetic mark that controls gene expression
2 types of enzymes catalyze
KDM1A/LSD1: FAD dependent
JHDM: Fe2+, O2, α-KG dependent, require catalytic amounts of vitamin C
DNA demethylases
DNA methylation at promoter elements for epigenetic modification
TET: DNA demthylase that Fe2+, O2, α-KG dependent, require catalytic amounts of vitamin C
ROS defense and vitamin C
ascorbate can react with radicals and quench radical chain reactions
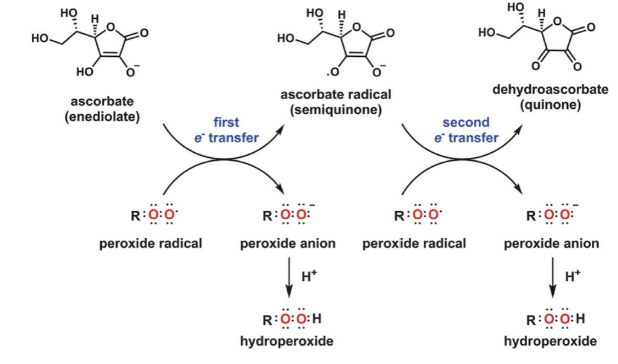
ascorbate radical is stable and doesn’t spontaneously react with other molecules
DHA reduced back to ascorbate by NADPH-utilizing glutathione reductase
vitamin C is water soluble so it can react with water soluble radicals
radical chain reactions in polyunsaturated lipids (and vit E)
polyunsaturated lipids can react with hydroxyl radical and cause chain rxn that damage lipid molecules
vit E intercepts ROO• radical to stop chain rxn
poly- and mono-unsaturated lipids are more susceptible to chain reactions because it is easier to subtract e from unsaturated carbon
vitamin E and ROS defense
vit E can react with radicals to form a stable radical that stops chain reactions
stable due to possibility of resonance states above where electron density of radical can be delocalized
Vit E is lipophillic, so it reacts with lipid radicals unlike vit C
vitamin E transport
lipophilic: transported across tissue with other lipids in LDL and CM
vit E protects lipids in LDL from oxidation as there is high [O2] in blood which promotes oxidation (O2 more soluble in lipids than water)
vitamin E tissue distribution
most of body Vit E is accumulated in adipose tissue where it helps to minimize stored lipid oxidation