Organic Chemistry Reactions
1/80
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
81 Terms
H-X
X adds Markovnikov
H-Br and ROOR
X adds anti-Markovnikov
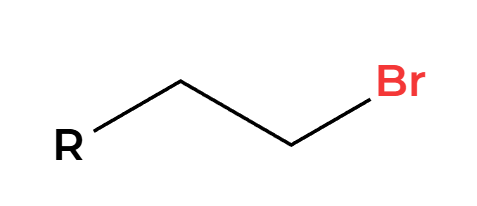
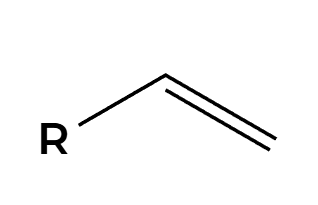
H+ and H2O
OH adds Markovnikov
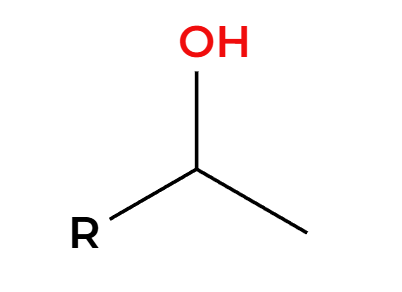
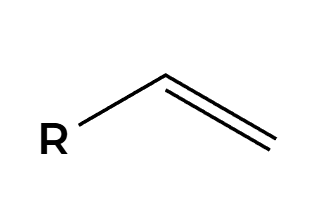
H3O+
OH adds Markovnikov
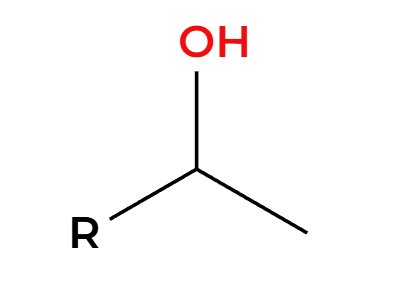
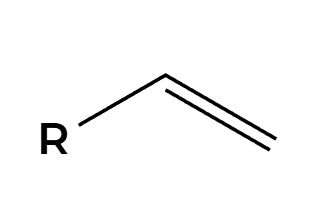
H2SO4 (dilute)
OH adds Markovnikov
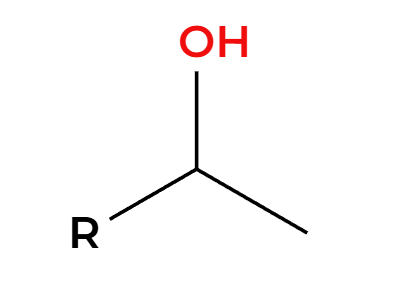
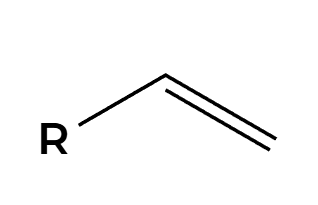
Hg(OAc)2, H2O
NaBH4
OH adds Markovnikov, cannot rearrange
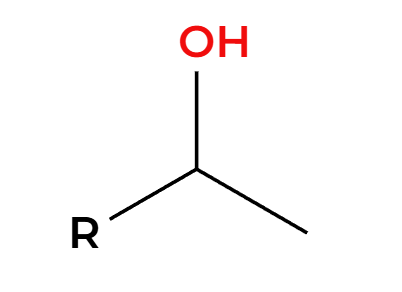
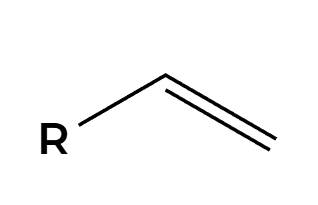
BH3
H2O2, NaOH
OH adds Anti-Markovnikov, groups add syn
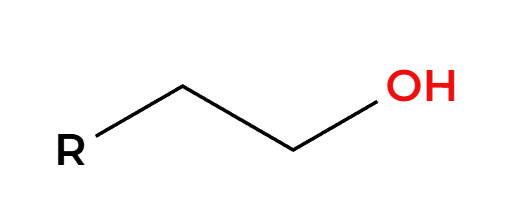
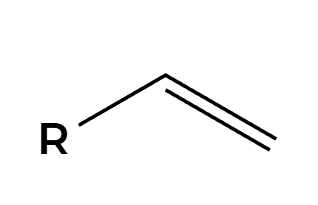
H2, Pt
H’s add syn
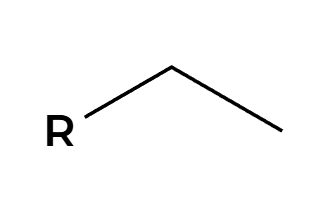
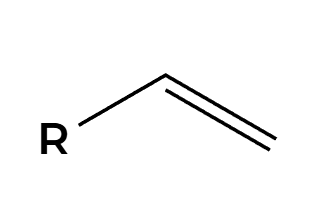
X2
X’s add anti
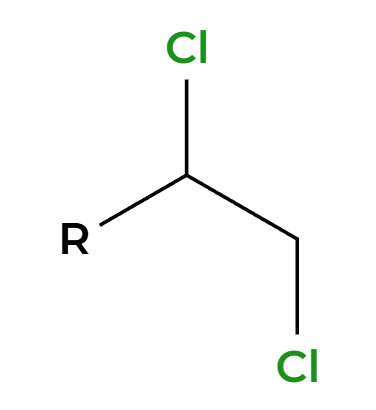
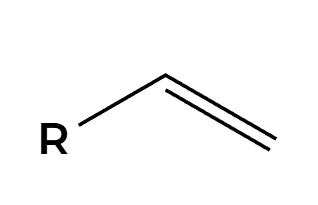
X2, H2O
OH adds Markovnikov, add anti
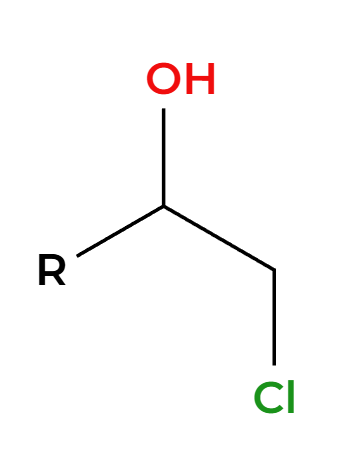
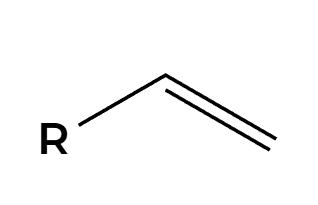
RCO3H
H3O+
OH’s add anti
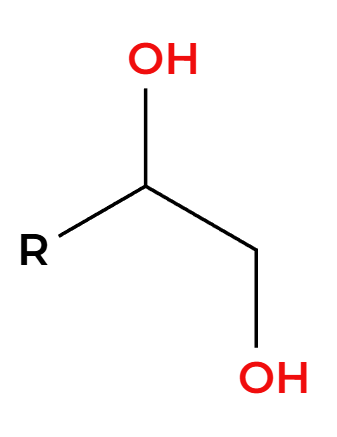
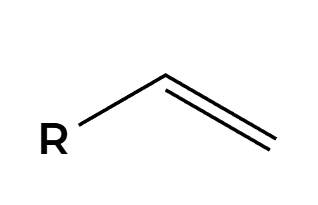
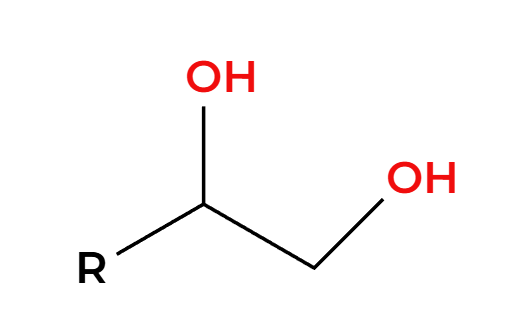
OsO4
NaHSO3, H2O
OH’s add syn
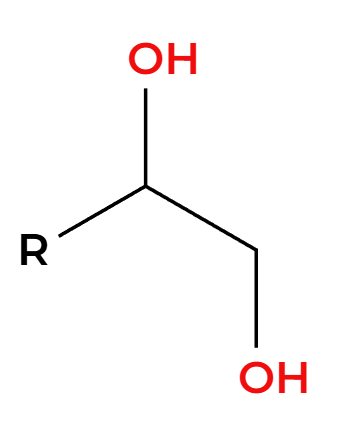
MCPBA
H3O+
OH’s add anti
O3
DMS
cleavage
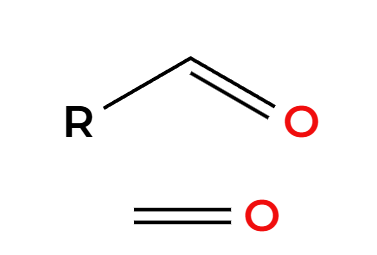
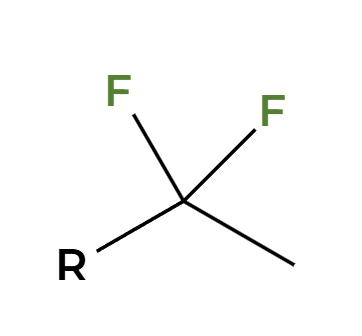
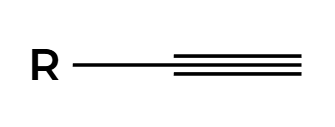
NaNH2
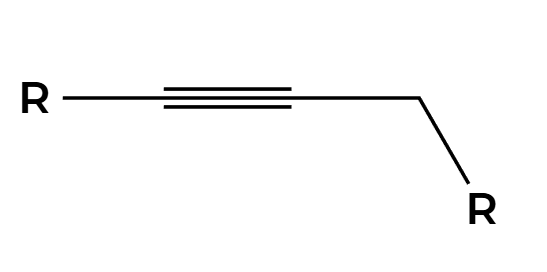
alkyne
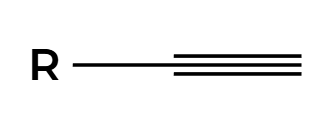
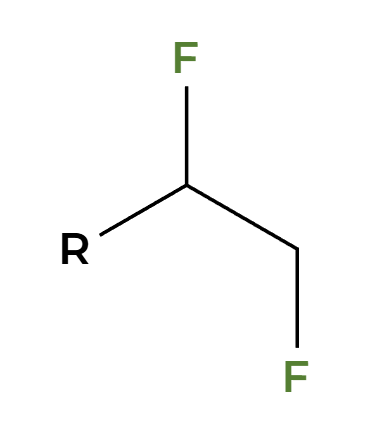
NaNH2
alkyne
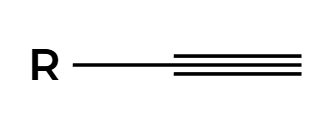
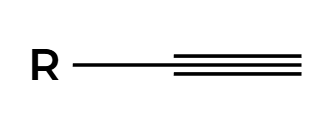
HX
X adds Markovnikov
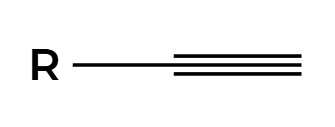
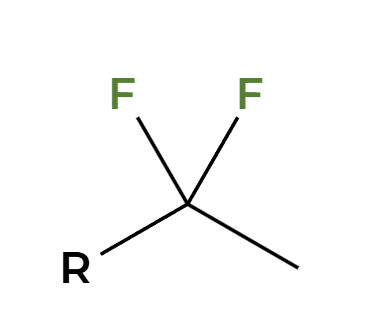
2HX
2X adds Markovnikov
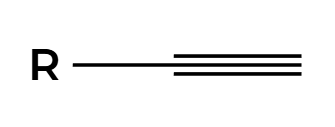
2X2
add 2 to each side of pi bonds
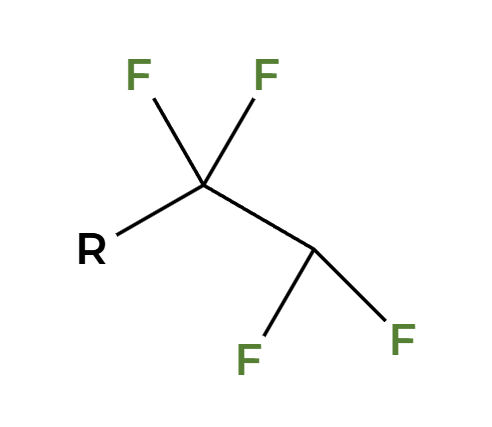
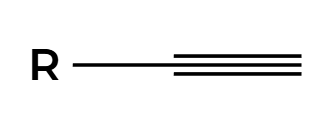
X2
Xs add anti
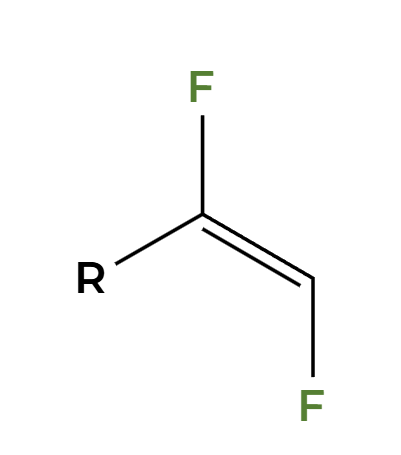
HgSO4, H2SO4, H2O
OH adds Markovnikov to make enol then tautomerization to ketone
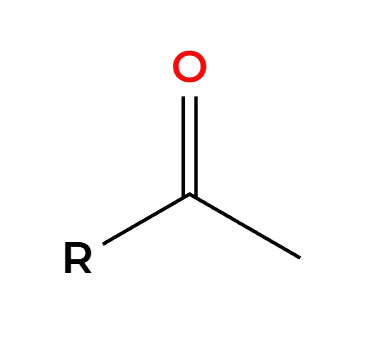
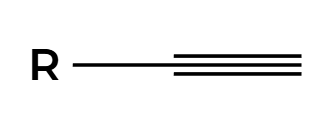
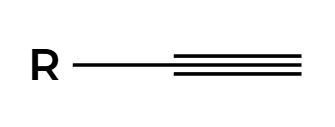
R2BH
H2O2, NaOH
OH adds anti Markovnikov to make enol then tautomerization to aldehyde
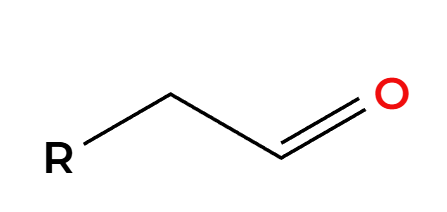
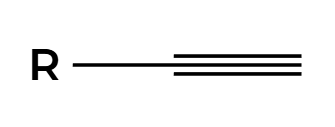
H2, Pt
cannot stop at alkene, goes all the way to alkane
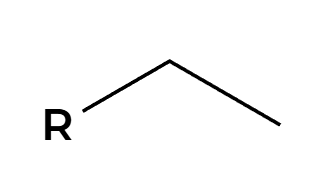
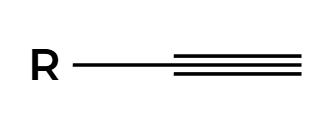
H2, Lindlar
H’s add syn
Na, NH3
H’s add anti
H2, Lindlar
only cis product is made
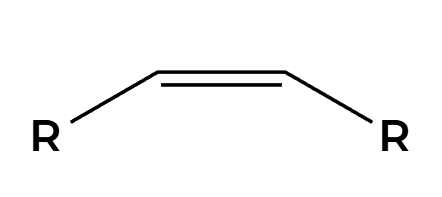
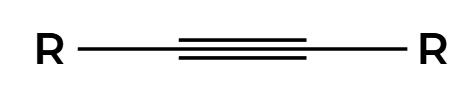
Na, NH3
only trans product is made
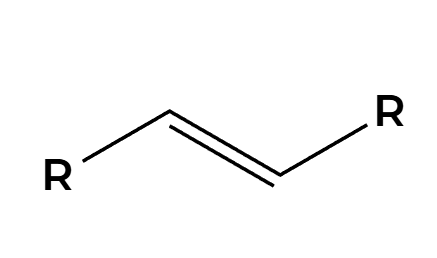
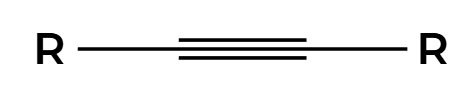
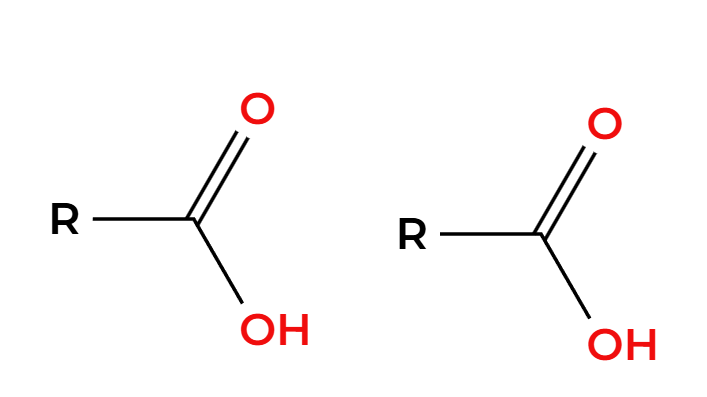
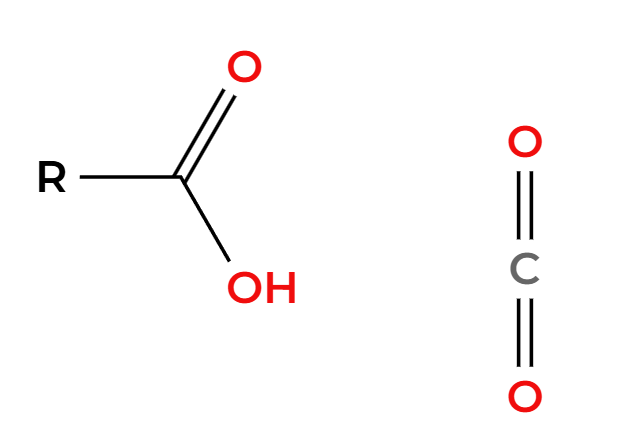
O3
H2O
ozonolysis
O3
H2O
ozonolysis
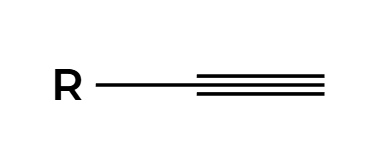
NaNH2
low substituted alpha carbon
coupling reaction
Br2 and light (hv)
Br adds most substituted
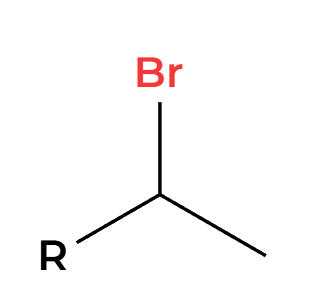
NBS and light (hv)
Br adds more substituted allylic
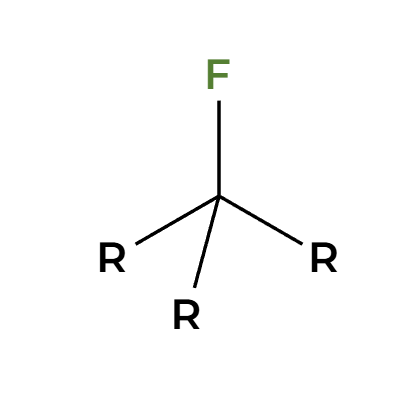
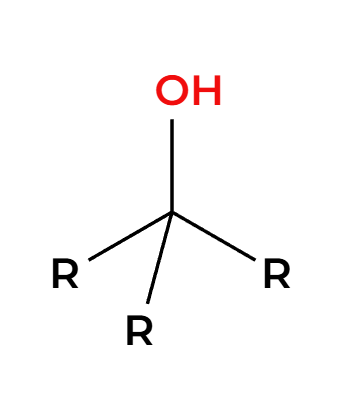
H2O
only works on tertiary alpha C
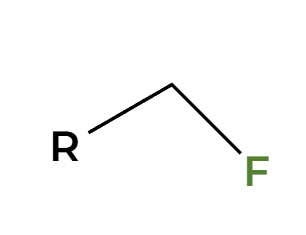
NaOH
only works on primary alpha C
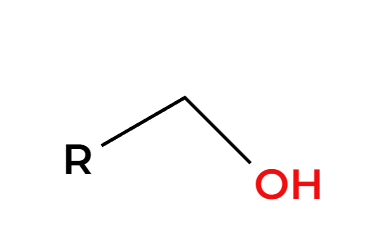
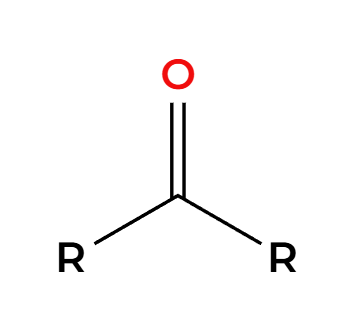
NaBH4 and ROH
mild reduction
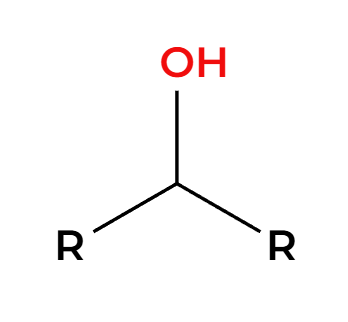
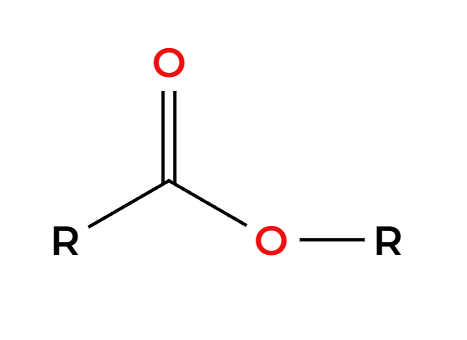
LAH
H2O
strong reduction
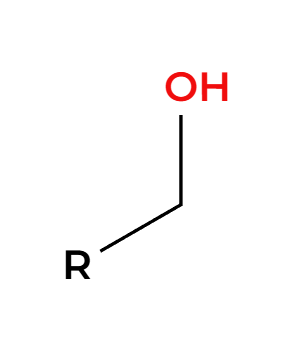
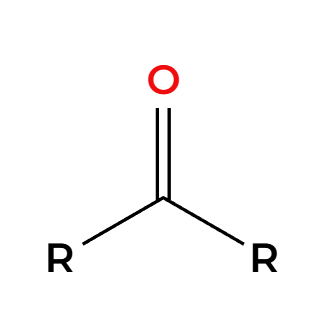
RMgBr
H2O
reduction and coupling
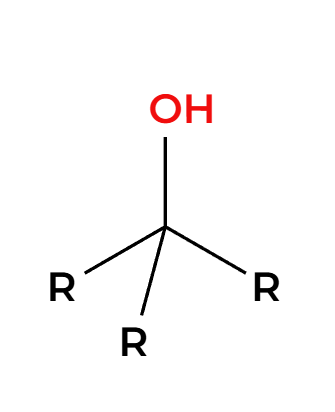
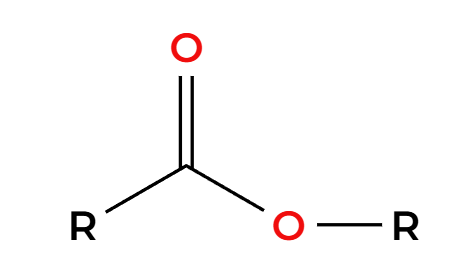
RMgBr
H2O
reduction and coupling
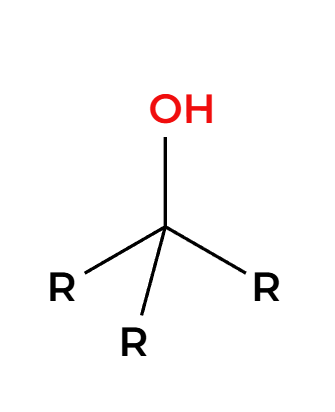
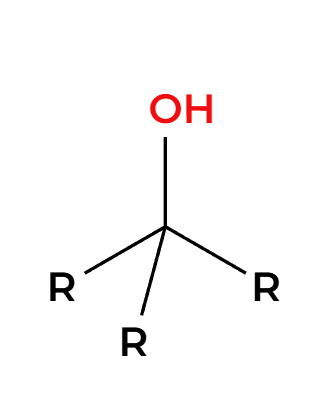
HX
only works on primary or tertiary alpha carbons
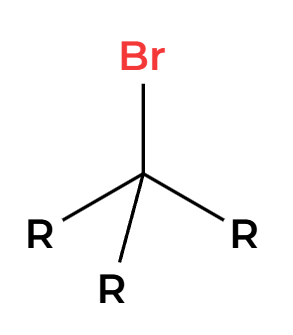
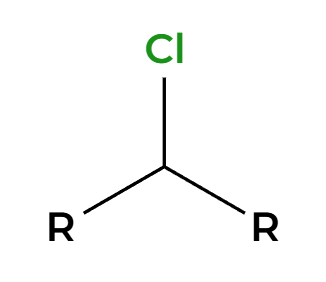
SOCl2
Cl adds 180 opposite
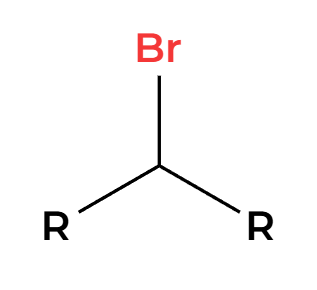
PBr3
Br adds 180 opposite
PCC
mild oxidation
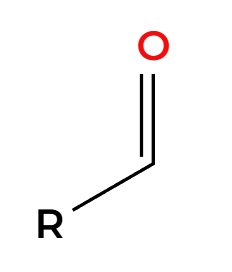
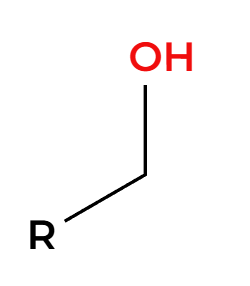
CrO3
strong oxidation
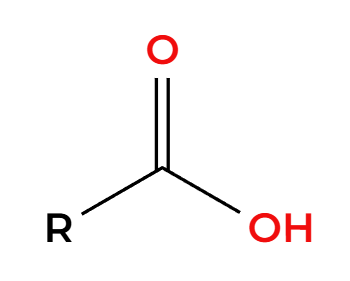
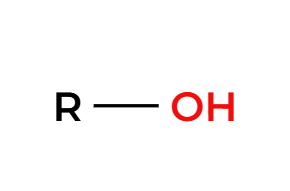
H2SO4 (cat) + heat
makes a symmetrical ether
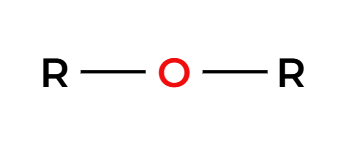
NaH
R’X (primary substituted)
can create uneven ether
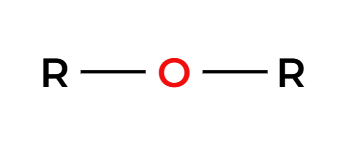
Hg(OAc)2, ROH
NaBH4
OR adds more substituted
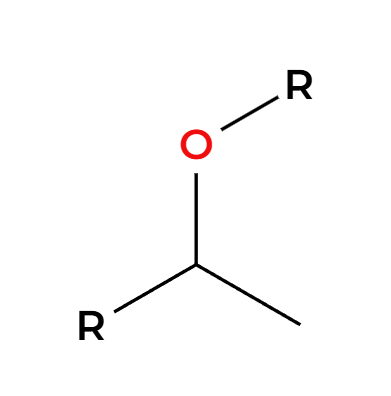
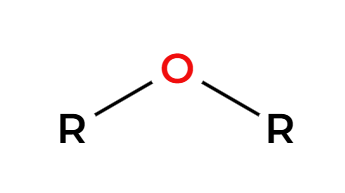
HX(excess) + heat
(+H2O) breaks ethers
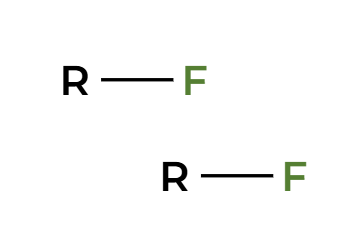
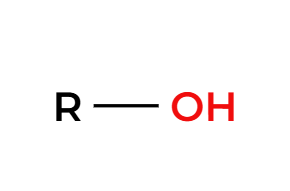
TmsCl
protected alcohol (silyl ether)
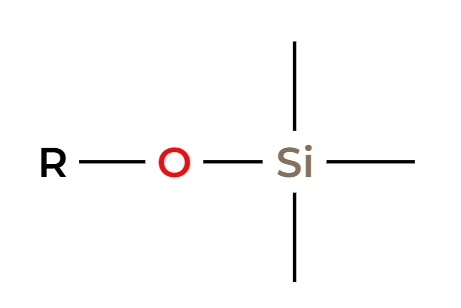
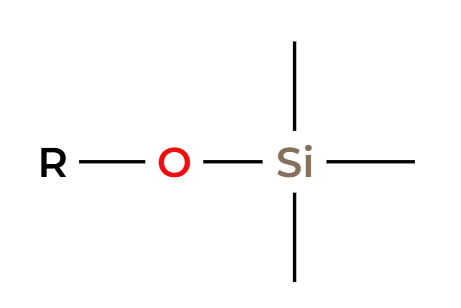
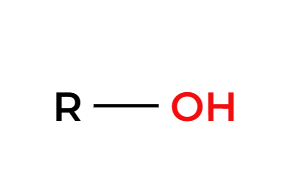
H3O+
deprotected alcohol
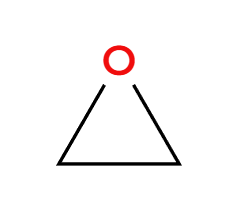
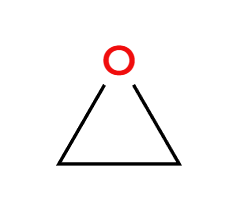
MCPBA
epoxide preparation
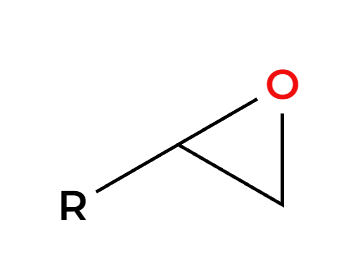
X2, H2O
NaOH
epoxide preparation
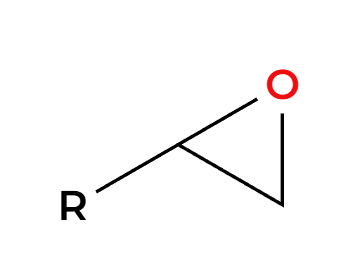
NaOR
H2O
basic condition, OR attaches less substituted
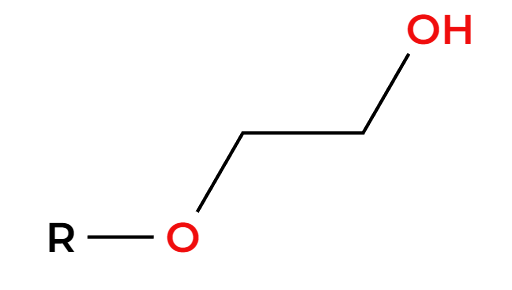
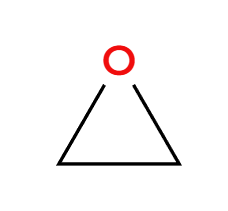
NaCN
H2O
basic condition, CN adds less substituted
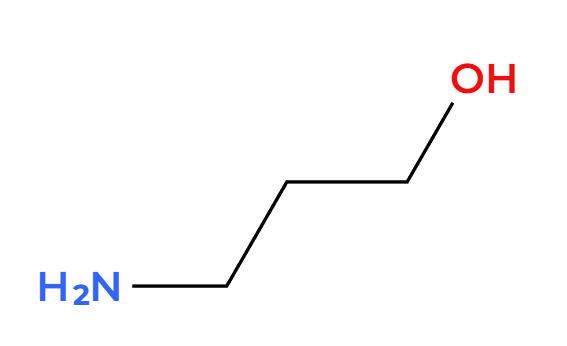
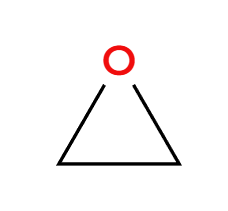
NaSH
H2O
basic condition, SH adds less substituted
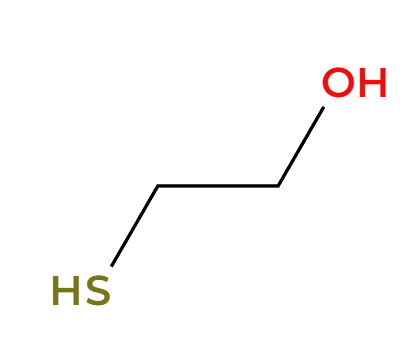
RMgBr
H2O
basic condition, R group adds less substituted
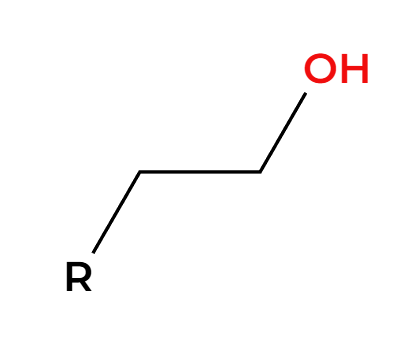
LAH
H2O
basic condition, H adds less substituted
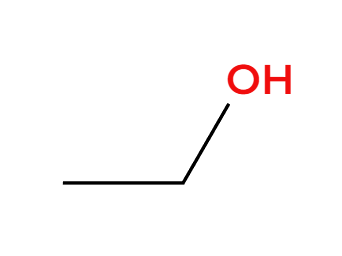
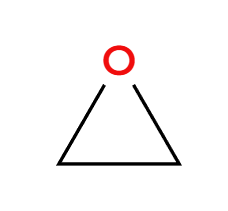
HX
acidic condition, x adds 3>1>2
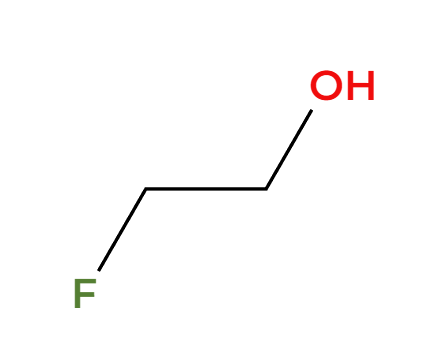
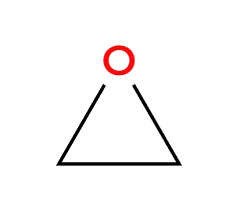
H3O+
acidic condition, OH adds 3>1>2
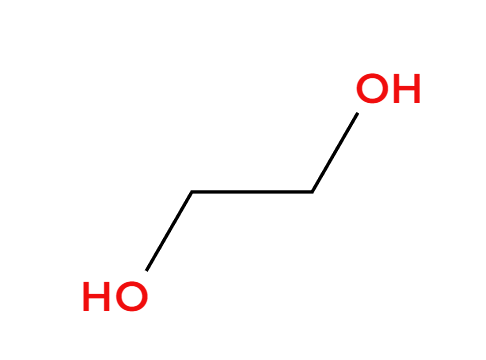
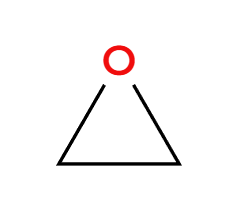
H+ and ROH
acidic condition, OR adds 3>1>2
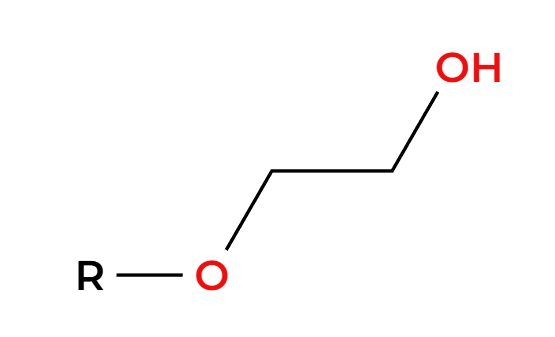
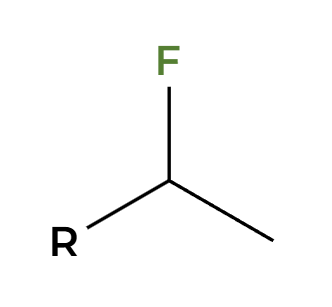
NaSH
backside attack, alkyl halide to thiol
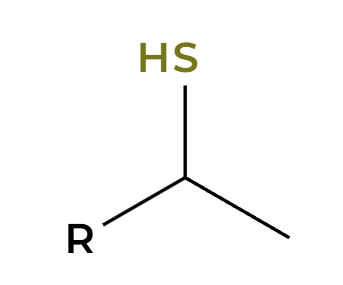
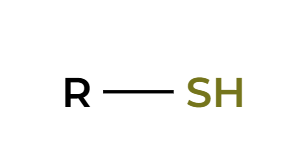
NaH
RX
thiol to sulfide
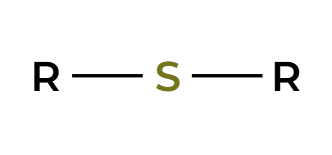
t-bOK
conjugated diene preparation
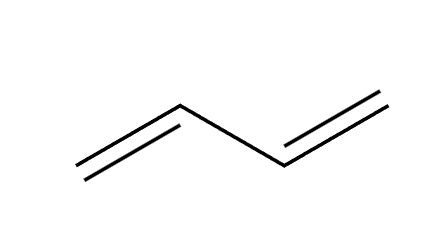
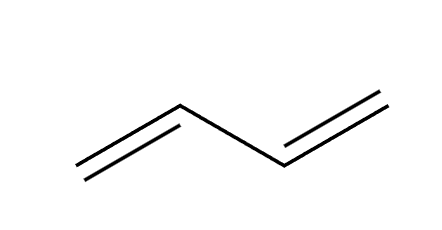
HX
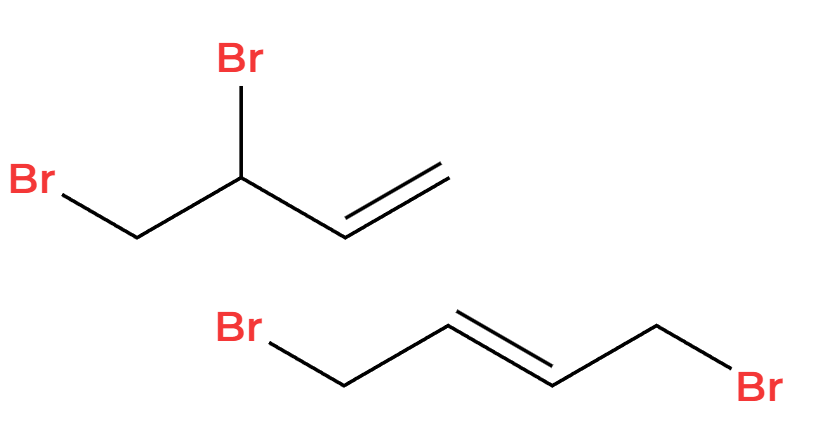
1,2 and 1,4 addition
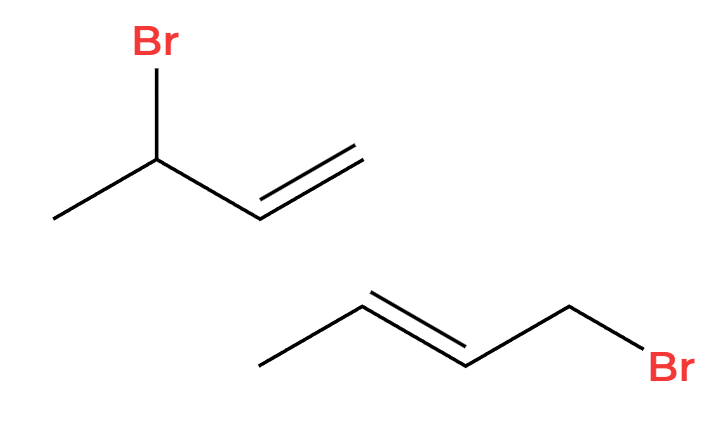
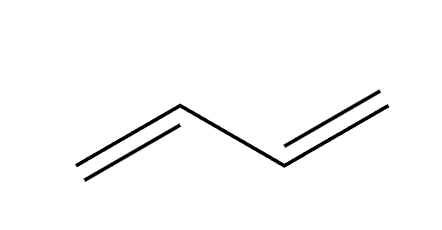
X2
1,2 and 1,4 addition
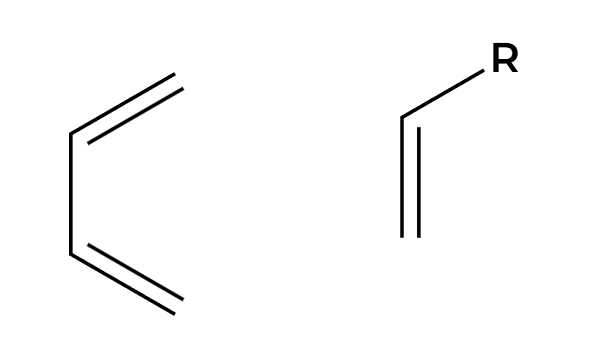
heat
diels alder reaction
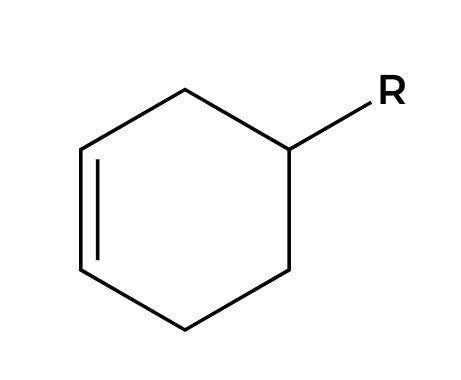
hv
2+2 addition
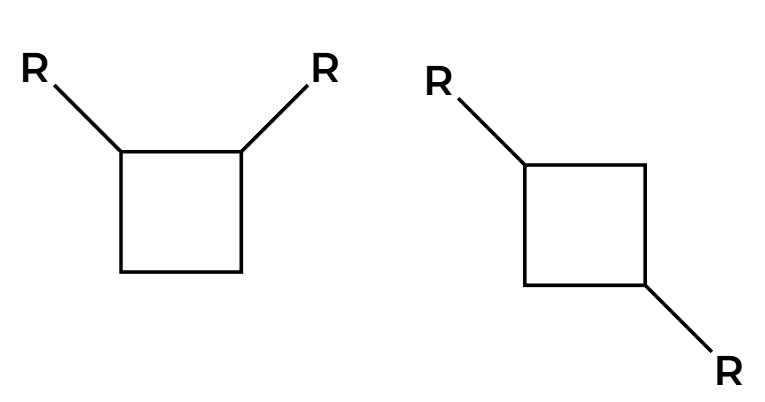
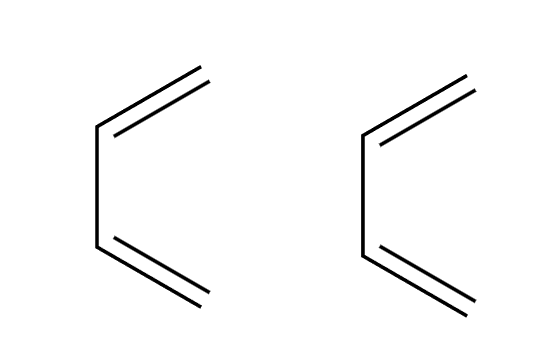
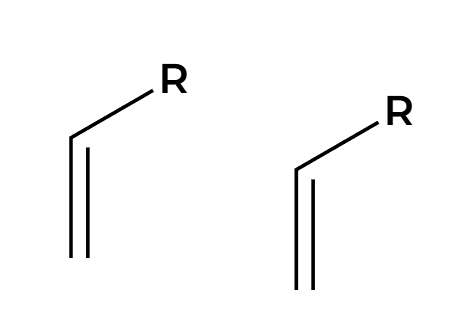
hv
4+4 addition
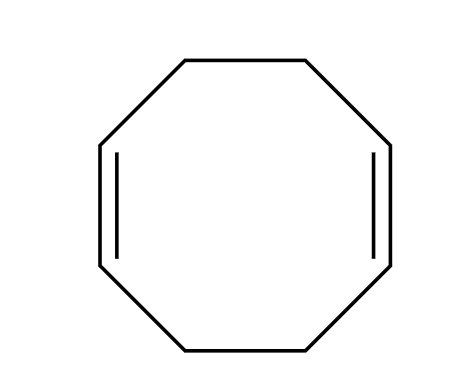
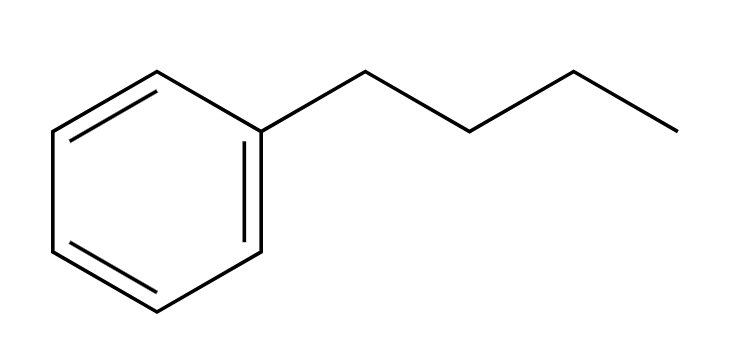
KMnO4
strong oxidation
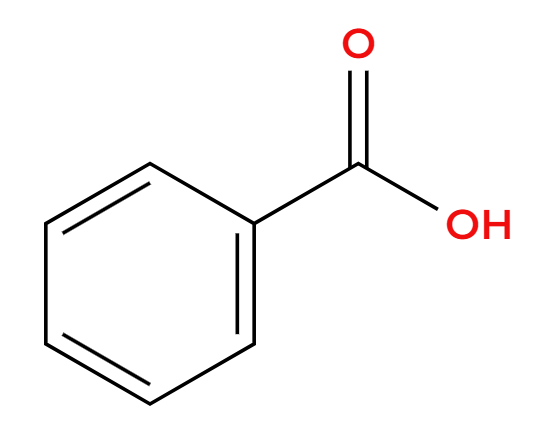
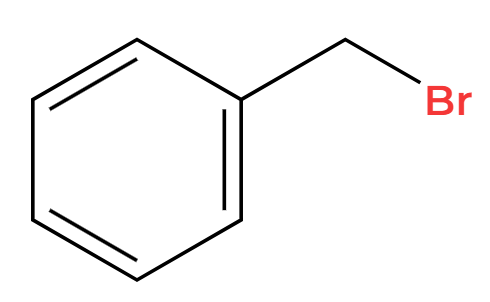
NaCl
Cl replacement
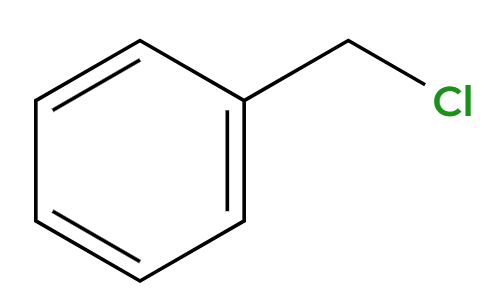
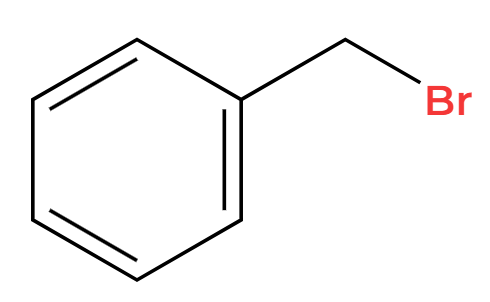
HCl
Cl replacement
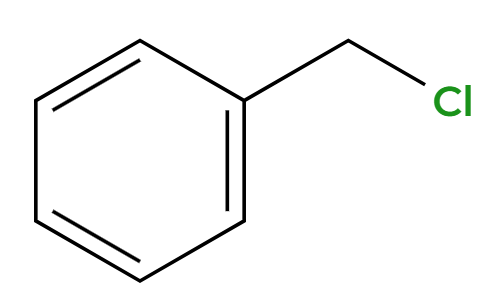
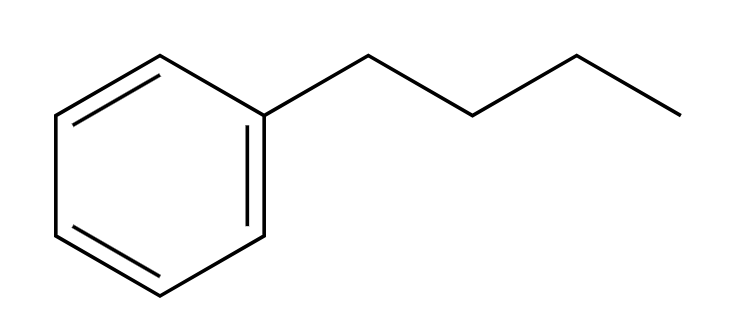
NBS, hv
Br adds at benzylic position
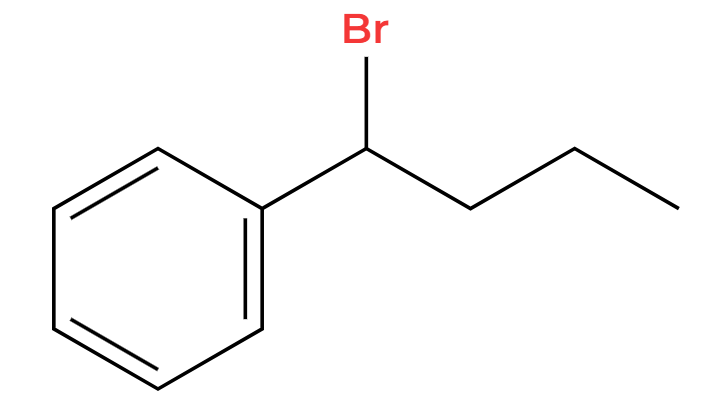
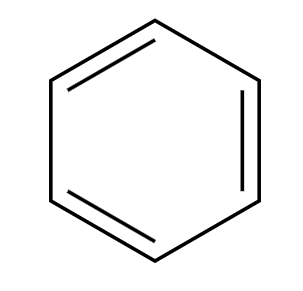
Br2, FeBr3
+HBr
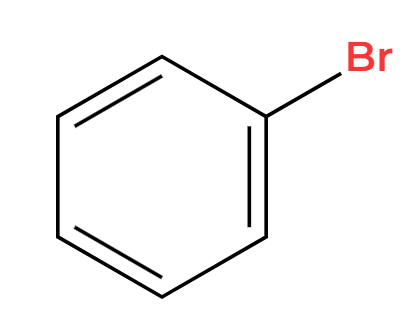
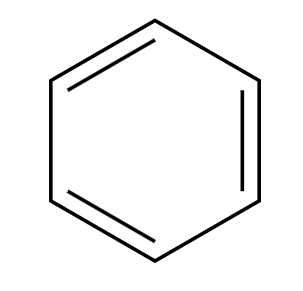
H2SO4 (conc)
+H2O
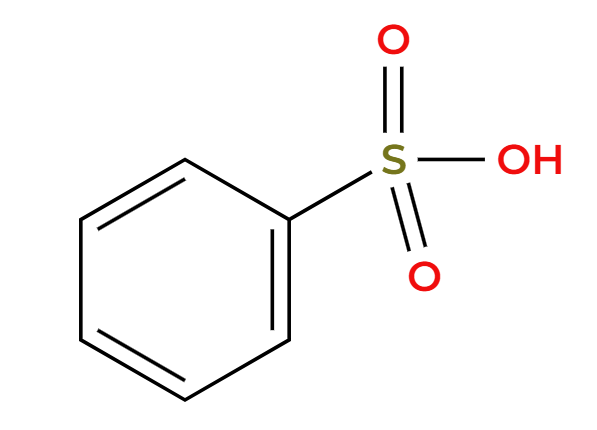
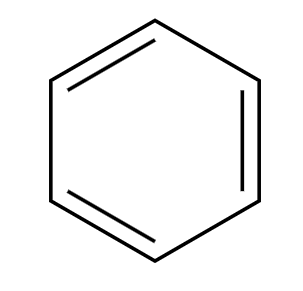
R-Cl, AlCl3
+HCl
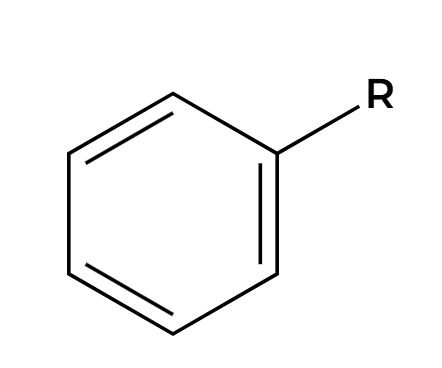
Cl2, AlCl3
+HCl
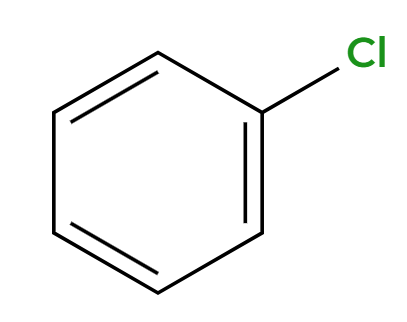
HNO3, H2SO4
+H2O
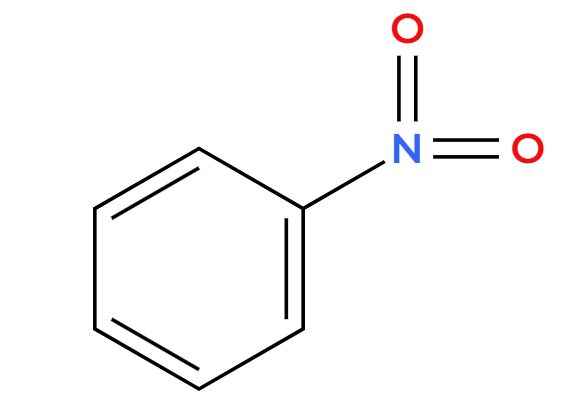
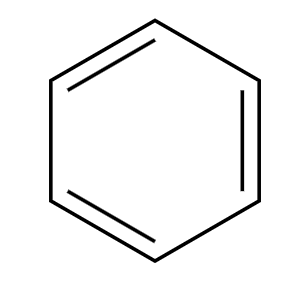
Cl-C=O plus R group, AlCl3
+HCl
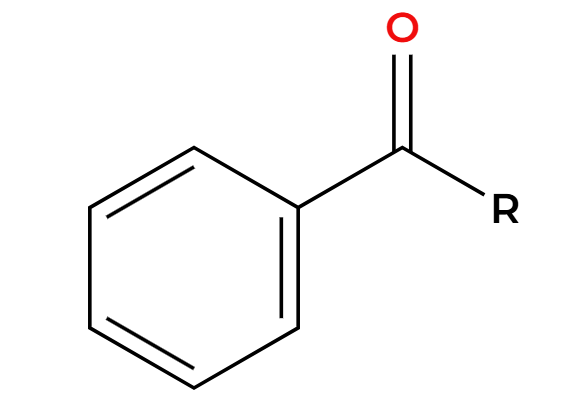
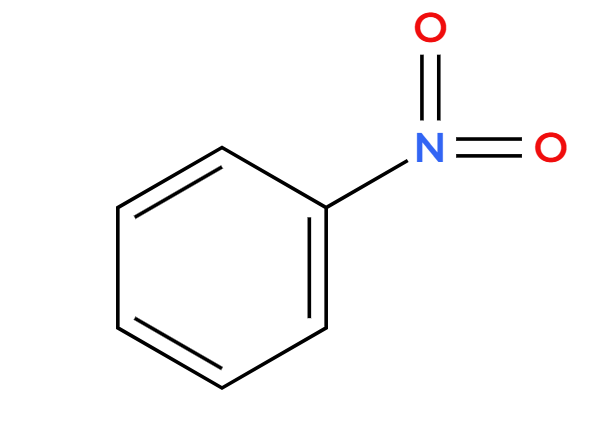
Zn, HCl
.
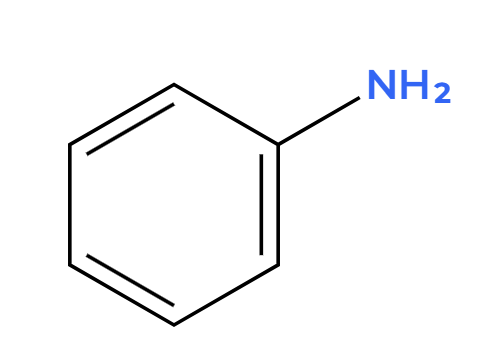
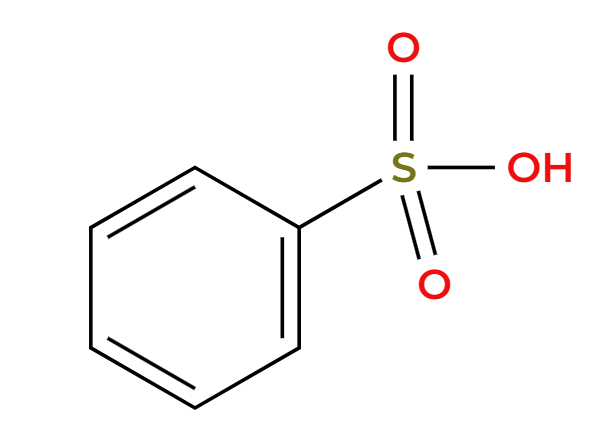
H2SO4 (dilute)
.
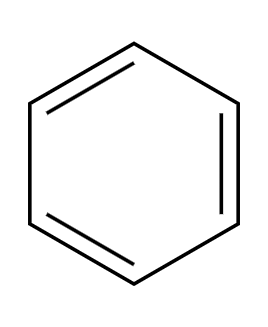
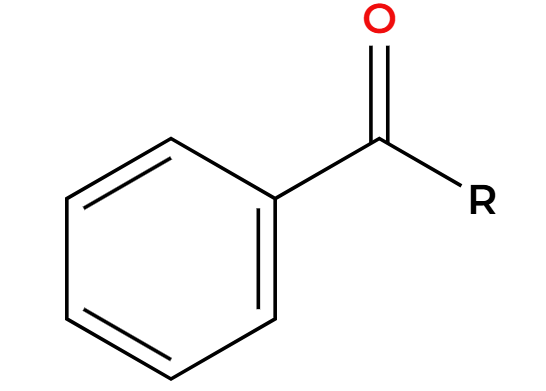
Zn/Hg, HCl
.
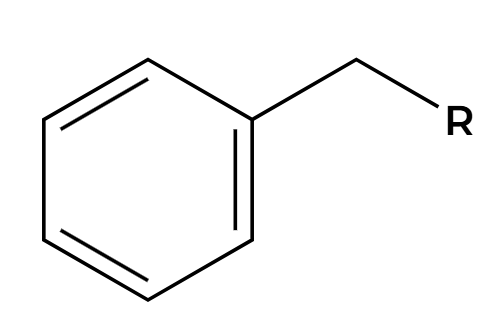
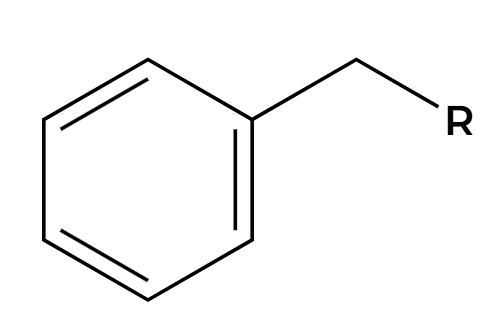
KMnO4, H3O+
.