CE 320 Exam 3
1/63
Earn XP
Description and Tags
The University of Alabama Environmental Engineering - Dr. Leigh Terry
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
How does organic pollution lead to fish kills?
The resultant oxygen depletion
What is a stream’s natural recovery process when dealing with oxygen-demanding pollution?
Natural Reaeration
Assimilation Capacity - As long as the pollutant load stays within the stream’s capacity, it can neutralize the waste and recover naturally
Oxygen Balance
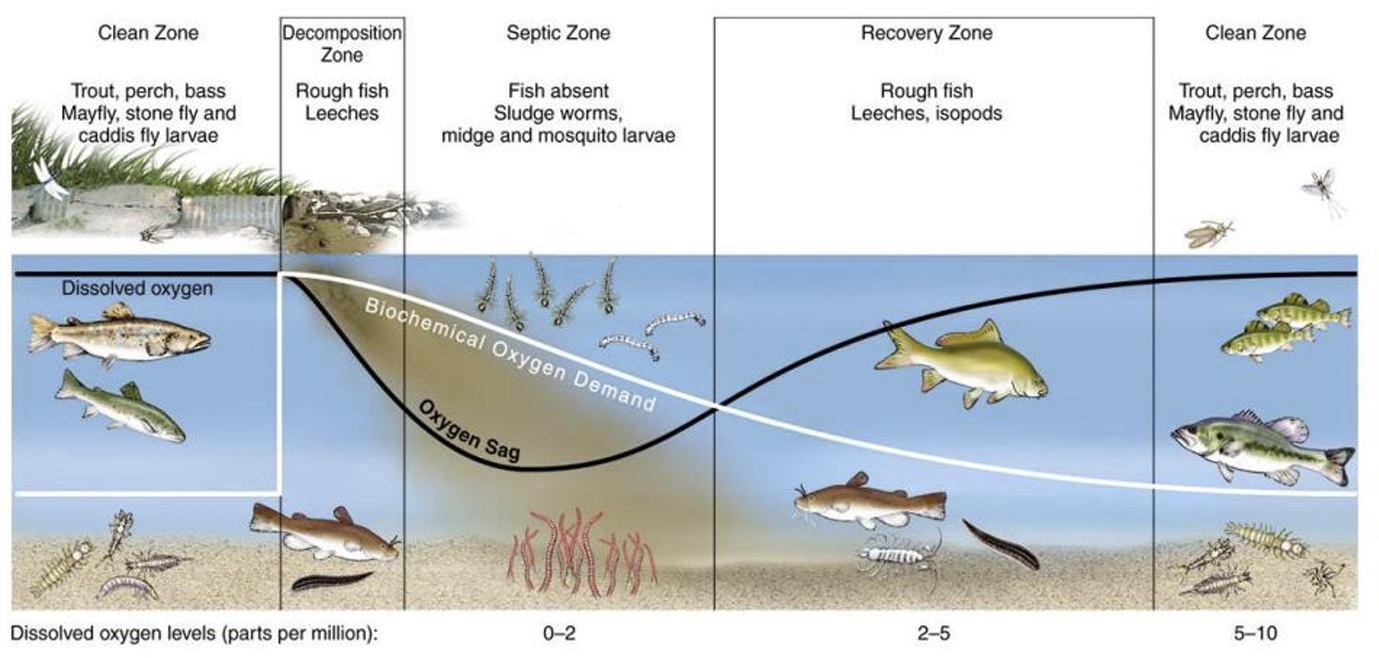
DO Sag
Dissolved oxygen concentrations decline as oxygen demanding pollutants are oxidized, then recover and the stream returns to a healthy condition
Effects of Temperature on Dissolved Oxygen
Impacts the solubility of oxygen in water - lower temp water can hold more oxygen
Impacts the rate at which microbes degrade oxygen demanding compounds
What are the worst-case conditions downstream?
The critical point (C)
Streeter-Phelps Model
A tool to assess ability of a stream to absorb an oxygen demanding pollutant model
When does deoxygenation occur?
Occurs as microbes degrade the waste introduced to the river and consume DO (aqueous O2)
When does reaeration occur?
Occurs as oxygen is resupplied to the river from the atmosphere
How does kd compare to k in shallow and fast-moving streams?
kd>>k
How does kd compare to k in deep and slow-moving rivers?
kd is similar to k
How do depth and turbulence affect the reaeration coefficient (kr) in a river?
kr is inversely proportional to depth and directly proportional to velocity
High kr = shallow/fast
Low kr = deep/slow
What is the DO Standard?
The minimum DO to protect wildlife
DO Standard vs DO at Critical Point
DO @ critical point > DO Standard = OK
DO @ critical point < DO Standard = remediation needed
Main factors that influence critical DO level
Ultimate BOD (Lw)
Qw
DOw
kd
kr
Possible solutions for low DO
Manage the waste stream
Lower wastewater volume
More effective wastewater treatment processes
Aerate wastewater effluent before discharging
Extreme: aerate the river at the critical point
What are indicators of stream health?
DO and BOD
What does BOD model?
A 1st order decay reaction
Municipal Water Supply and Sanitation Process
Watershed Protection → Pumped → Drinking Water Treatment Plant → Piped Distribution System → Household → Sewers → Wastewater Treatment Plant → Discharged
Safe Drinking Water Act (SDWA)
1974
Covers “public water systems”
Gives the US EPA authority to set minimum drinking water quality standards
National Primary Drinking Water Regulations (NPDWR)
Protects human health
Legally enforceable standards
National Secondary Drinking Water Regulations (NSDWR)
Provide guidelines for aesthetic quality
Not enforceable federally, just guidelines that states can opt to enforce
What is MCLs?
Maximum Contaminant Levels
Primary MCLs
To protect health
Most common is an upper limit
Some use a “Treatment Technique” (TT) requirement
What did the EPA pass in 2024?
First-ever national drinking water standard for PFAS
Designated PFAS-PFOA and PFOS as hazardous substances under the Superfund
Secondary MCLs
To protect aesthetic quality
Can have a maximum
Can have a range
Public Water System (PWS)
Served by the SDWA
13-15% of Americans use private household wells for drinking water, which SDWA doesn’t apply to
>150,000 PWS in the US
Community Water System (CWS)
Same customers year round, >250 million people served in the US
Non-Community Water System (NCWS)
Non-Transient (NTNCWS) >/= 25 same people >/= 6mo/yr but not year round
7 million people served
Transient (TNCWS) camps, resorts, campgrounds
13 million people served
Size Classification of PWS
Classified by number of people served
Very small: 25-500
Small: 501-3,000
Medium: 3,001-10,000
Large: 10,001-100,000
Very Large: >100,000
What are the major technical goals of drinking water treatment?
Remove particles (including sediment, microorganisms, etc.)
Kill any remaining microorganisms that aren’t removed
Conventional Surface Water Treatment for Drinking Water
Screening (removes sticks, leaves, etc.) → Rapid Mix → Flocculation → Sedimentation (sludge exits) → Filtration → Disinfection → To distribution
What is the purpose of each unit process in drinking water treatment?
The first 4 units work together to remove particles/dissolved contaminants from water
Disinfection is a final step for killing any remaining pathogens
What does rapid mix do?
A chemical coagulant is added to the water and a high-energy mixers disperses the chemicals to neutralize the charges on the particles so they are ready to stick together. (Alum, Al2(SO4)3)
What does flocculation?
The water is gently mixed and forms “floc”, large, heavy clumps, of the particles. This allows the water to move onto the next step, sedimentation as the heavy floc can now sink to the bottom.
How does sedimentation work?
Allows time for large floc to settle
Large alum complexes “sweep” through water capturing smaller molecules and particles, pulling them to the bottom
Inlet Zone
Purpose is to evenly distribute flow across sedimentation basin
Settling Zone
Enable design flow, slow enough to allow floc to settle out
Outlet Zone
Weirs provide large area for water to flow out: prevent high velocity from stirring up floc
What happens when the floc settles?
The solids are removed as wet “sludge”, dewatered settling pond or belt press), then shipped to land fill.
Filtration
Passage of water through a bed of porous media
Goals: To remove remaining floc, particles, and chlorine-resistant pathogens
How does filtration work with disinfection?
It removes the most chlorine-resistant pathogens
How does disinfection work?
It kills the remaining pathogens, usually using chlorine
Why is chlorine allowed a “residual” in piped distribution systems?
In case there’s subsequent infiltration of treated water
To prevent overgrowth of biofilms in pipes
Redundancy
At least two of each unit process operated in parallel
ex. Need to backwash
What’s the most common coagulant?
Aluminum Sulfate (Alum)
Traditional coagulants are salts of +3 cations, Al3+ or Fe3+
How do coagulants work? (chemical process)
Natural water is full of ions and natural organic matter (NOM) carrying charge, and these ions become concentrated around charged particles. So adding cations compresses the repulsive layers
What is “Jar Testing”?
Lab simulation of coagulation/flocculation
What are the two major design configurations of the sedimentation tank?
Horizontal flow (rectangular)
Up flow (circular)
What are the 4 zones of the sedimentation tank?
Inlet Zone
Settling Zone
Outlet Zone
Sludge Zone
Sludge Zone
Where solids are removed
Weir Loading Rate (WLR)
A standard design code parameter that allows the design of the outlet Weir Length (WL)
Assumptions of horizontal sedimentation basins
Particles and velocity vectors are evenly distributed across the width and depth of the tank (what the “inlet zone” is built to accomplish)
Liquid moves as in ideal slug down the length of the tank (plug flow)
Any particle hitting the bottom of the tank is removed
What are filters in conventional DWT called?
Rapid sand filters
How do rapid sand filters work?
Gravity driven based on elevation head
Water → Sand → Support Gravel → Underdrain
How does backwashing work?
Pressure is used to rapidly drive water back up through filter → suspends sand and cleans filter → graded media bed
Why is it advantageous to use two types of filter media with different densities?
It prevents the top of the filter from clogging instantly, allowing the entire depth of the bed to pull its weight, leading to longer filter runs
What is commonly used with sand in the filters?
Anthracite coal