Chapter 6.1
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
What is energy?
The ability to perform work
What are forms of energy?
Thermal
Light
Sound
Potential
Mechanical
Chemical
Electrical
Nuclear
What is thermal energy?
Heat energy
What is light energy?
Energy from electromagnetic waves
What is sound energy?
Energy from vibrations
What is potential energy?
Stored energy
What is mechanical energy?
Energy of motion
What is chemical energy?
Energy stored in chemical bonds
What is electrical energy?
Energy from moving charges
What is nuclear energy?
Energy store in the nucleus
What is energy transfer?
Movement of energy from one object to another
What is energy conversion?
Change of energy from one form to another
What happens during imperfect energy transfer?
Some energy is lost, usually as heat
What is enthalpy (H)?
The sum of internal energy and PV (pressure x volume)
What does enthalpy approximate?
Thermal energy of the system and surroundings
What is enthalpy of reaction (ΔH)?
Heat gained or lost during a reaction
What does a positive ΔH mean?
Energy is gained (endothermic)
What does a negative ΔH mean?
Energy is released (exothermic)
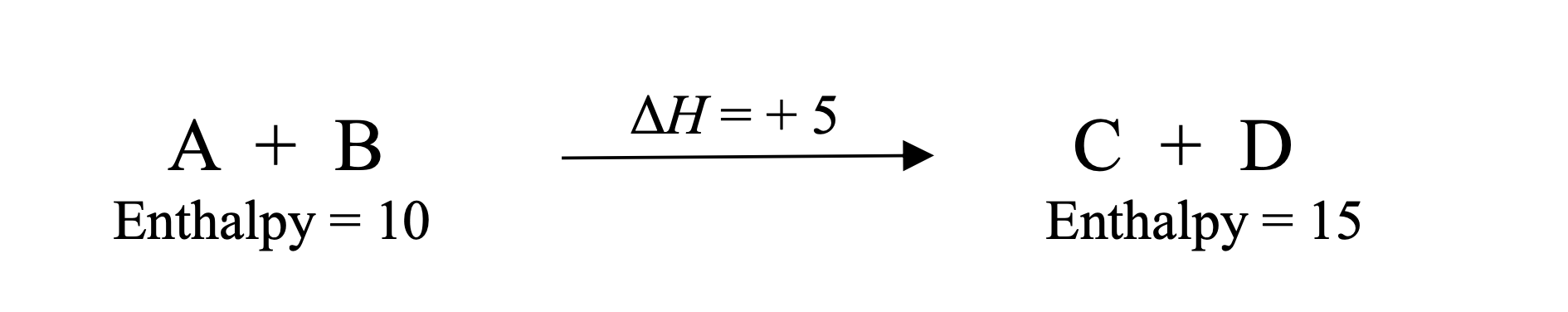
In A + B → C + D, why is ΔH = +5?
Products have higher enthalpy (15 − 10 = +5)
What does ΔH = +5 mean physically?
The reaction absorbs energy (endothermic)
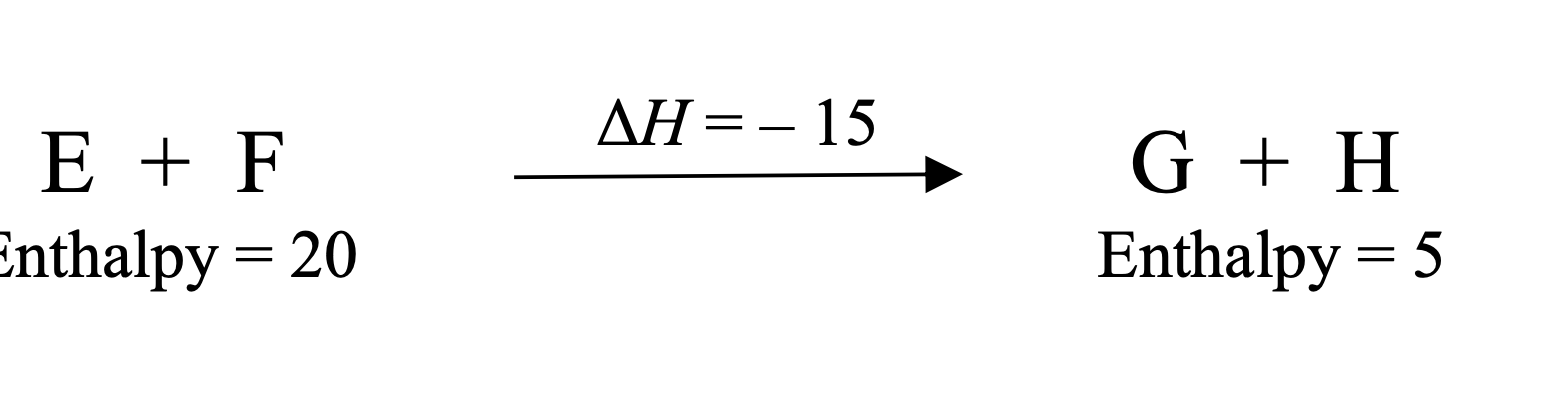
In E + F → G + H, why is ΔH = −15?
Products have lower enthalpy (5 − 20 = −15)
What does ΔH = −15 mean physically?
The reaction releases energy (exothermic)
Key pattern for exams (ΔH sign)?
Positive = energy in, Negative = energy out
What does exothermic mean?
A reaction that releases heat (heat exits the system)
In an exothermic reaction, are products higher or lower in energy than reactants?
Lower in energy
What is the sign of ΔH in an exothermic reaction?
Negative
What happens to the temperature of the surroundings in an exothermic reaction?
It increases
What does “endothermic” mean?
A reaction that absorbs heat (heat enters the system)
In an endothermic reaction, are products higher or lower in energy than reactants?
Higher in energy
What is the sign of ΔH in an endothermic reaction?
Positive
What happens to the temperature of the surroundings in an endothermic reaction?
It decreases
What must happen for bonds to break during a reaction?
Energy must be absorbed
Do all reactions require energy to start?
Yes, activation energy is required
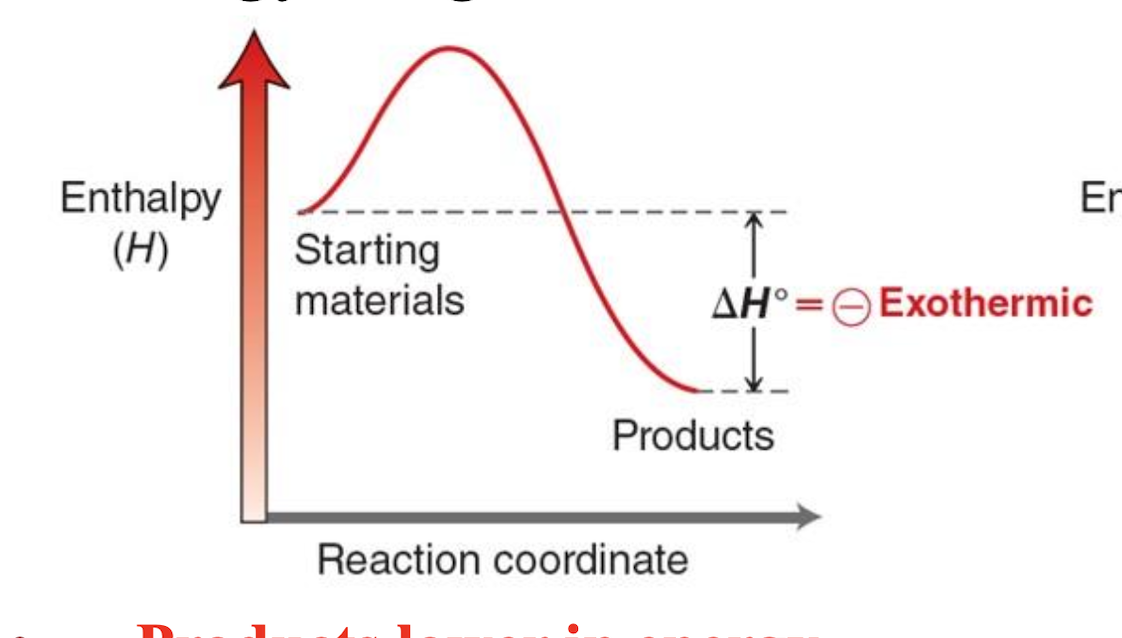
Explain what is happening in an exothermic energy diagram
The reactants start at a higher energy, require activation energy to reach the peak, and then release energy as they form lower-energy products; ΔH is negative because energy is released to the surroundings, increasing temperature.
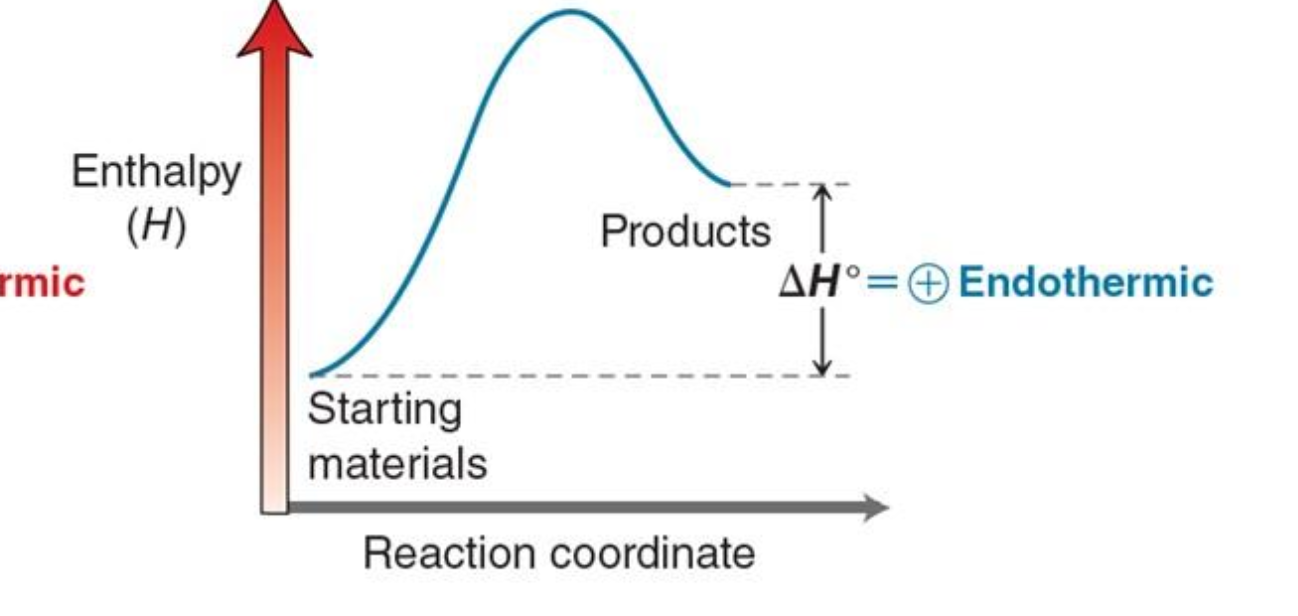
Explain what is happening in an endothermic energy diagram
The reactants start at a lower energy, absorb energy to reach the activation energy peak, and form higher-energy products; ΔH is positive because energy is absorbed from the surroundings, decreasing temperature.
What is bond dissociation energy (BDE)?
The energy required to break a bond
Are electrons in a bond high or low in energy?
Low in energy (bonding orbital)
What must happen to electrons to break a bond?
They must move to a higher-energy state
Does breaking a bond release or require energy?
It requires energy
Why do bonds require energy to break?
Because bonded electrons are in a stable, low-energy state
What are the two ways bonds can break?
Homolytically and heterolytically
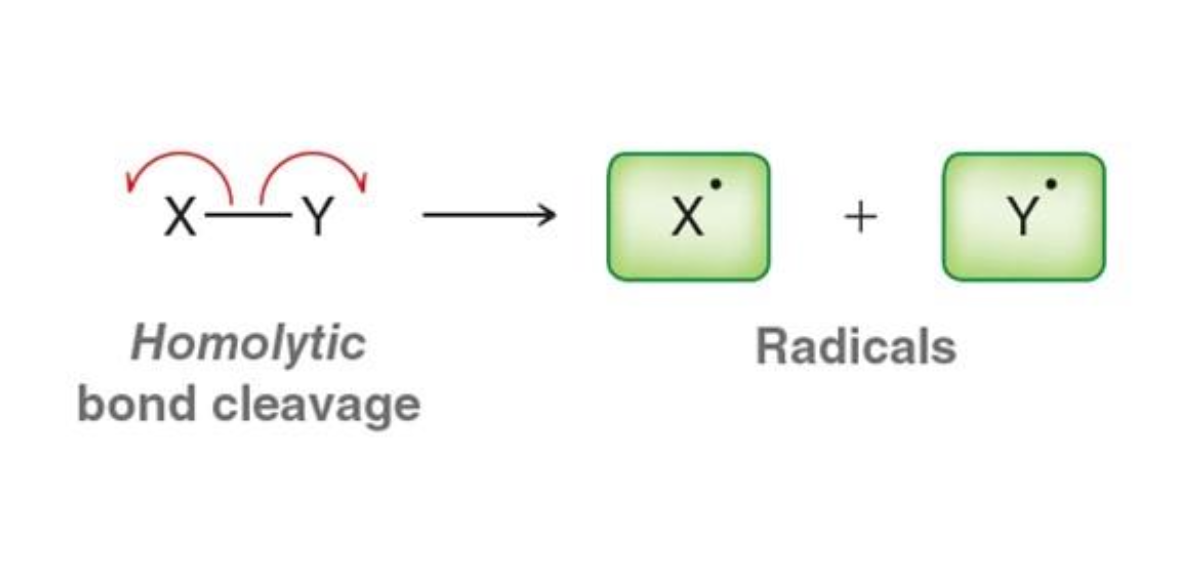
What happens in homolytic bond cleavage?
Electrons are split evenly between atoms
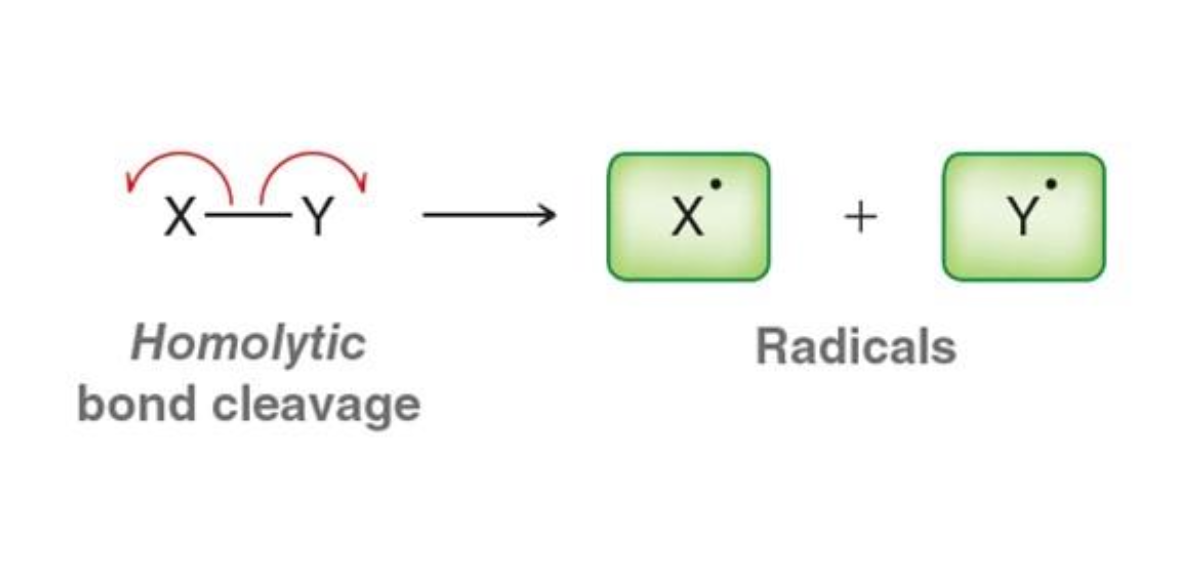
What is formed from homolytic cleavage?
Radicals
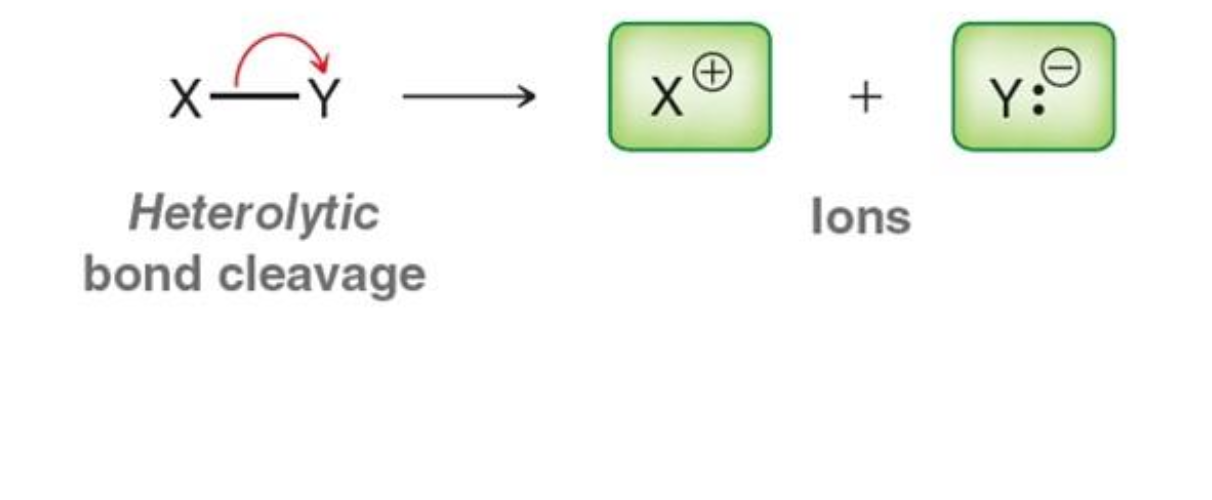
What happens in heterolytic bond cleavage?
One atom takes both electrons
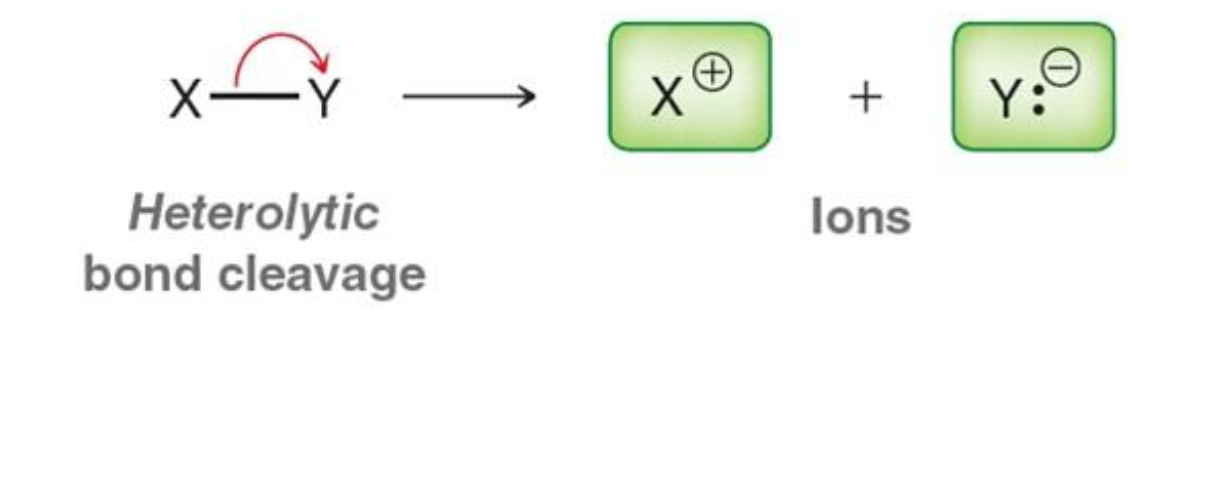
What is formed from heterolytic cleavage?
Ions (cation and anion)
Which type of bond breaking does bond dissociation energy (BDE) refer to?
Homolytic cleavage
Do stronger bonds have higher or lower bond dissociation energy (BDE)?
Higher BDE
As bond strength increases, what happens to BDE?
It increases
Which bond is strongest: H–F, H–Cl, H–Br, or H–I?
H–F
Which bond is weakest: H–F, H–Cl, H–Br, or H–I?
H–I
How does bond strength change down the halogen group (F → I)?
It decreases
How does BDE change down the halogen group (F → I)?
It decreases
What equation is used to calculate ΔH using bond energies?
ΔH = BDE (bonds broken) − BDE (bonds formed)
Do bonds broken require or release energy?
Require energy
Do bonds formed require or release energy?
Release energy
What does a negative ΔH indicate?
Exothermic reaction
What does a positive ΔH indicate?
Endothermic reaction
In this example, why is the reaction exothermic?
More energy is released from forming bonds than required to break bonds
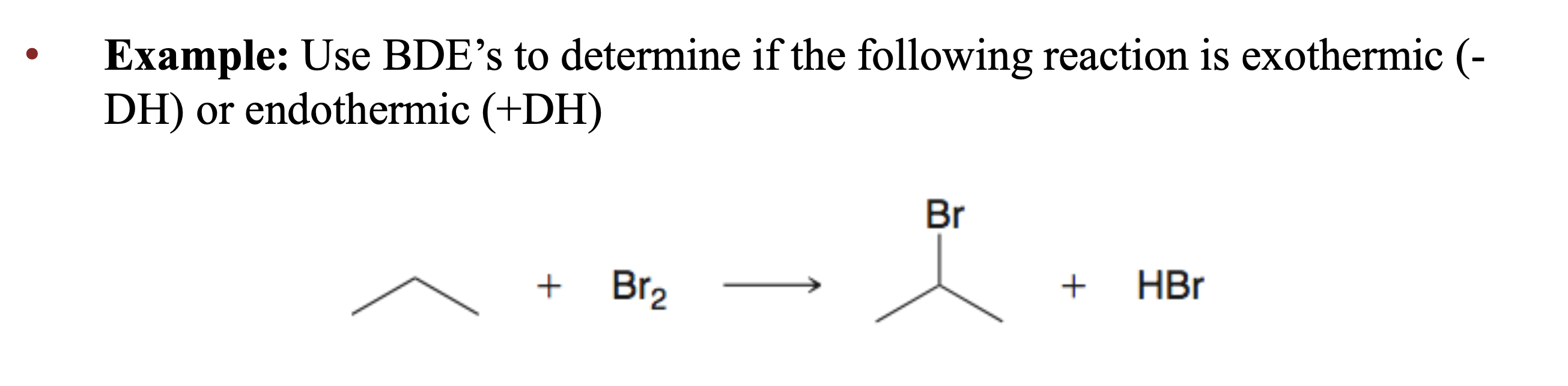

Are exothermic reactions always spontaneous?
No
Are endothermic reactions always nonspontaneous?
No
Does ΔH alone determine if a reaction is spontaneous?
No
No
Lower in energy
In an endothermic reaction, are products higher or lower in energy?
Higher in energy
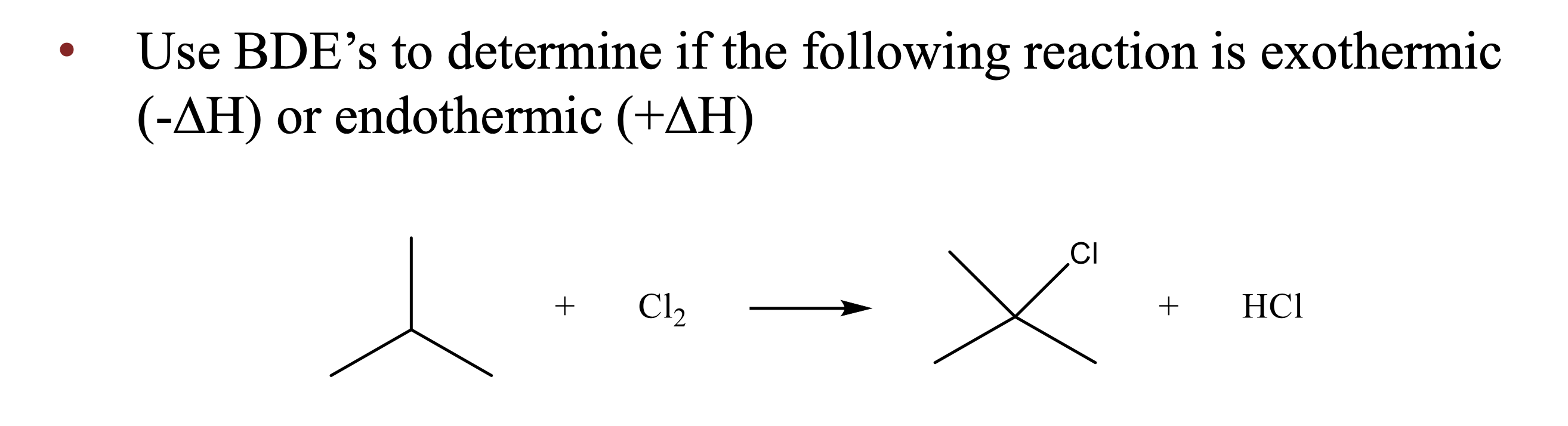
Bonds broken (C–H and Cl–Cl) require less energy than bonds formed (C–Cl and H–Cl) release, so ΔH is negative and the reaction is exothermic.
What is entropy (ΔS)?
The disorder or randomness of a system
What does entropy relate to at the molecular level?
The number of possible arrangements (microstates)
What happens to entropy when volume increases?
It increases
Why does entropy increase when gas expands?
There are more possible positions for molecules
More volume for a gas leads to what change in entropy?
Greater entropy
What equation determines spontaneity using entropy?
ΔSₜₒₜ = ΔS_sys + ΔS_surr
What two components make up ΔSₜₒₜ?
System and surroundings
What does ΔS_sys represent?
Entropy change of the system (reaction)
What does ΔS_surr represent?
Entropy change of the surroundings
What does it mean if ΔSₜₒₜ is positive?
The process is spontaneous
What does the 2nd law of thermodynamics state about entropy?
Spontaneous processes increase the entropy of the universe
When is ΔS_sys positive based on number of moles?
When there are more moles of products than reactants
Why does more moles increase entropy?
More particles means more possible arrangements
What happens to entropy when a cyclic compound becomes acyclic?
It increases
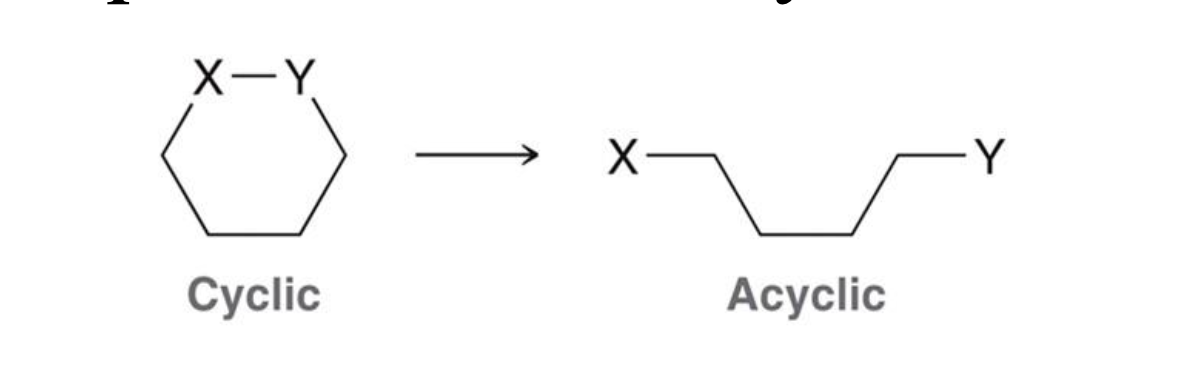
Why does going from cyclic to acyclic increase entropy?
The molecule has more freedom of movement
What is a simple rule for predicting ΔS_sys?
More particles or more freedom = higher entropy
What equation relates total entropy to system and surroundings?
ΔSₜₒₜ = ΔS_sys + ΔS_surr
What is the equation for ΔS_surr?
ΔS_surr = −ΔH_sys / T
What equation combines ΔH and ΔS into one value?
ΔG = ΔH − TΔS
What does ΔG < 0 indicate?
Spontaneous
What does ΔG > 0 indicate?
Nonspontaneous
What determines spontaneity more directly: ΔH or ΔG?
ΔG
What is the equation for Gibbs Free Energy?
ΔG = ΔH − TΔS
What does ΔG determine?
Spontaneity of a reaction
What does ΔH represent?
Change in enthalpy (heat)
What does ΔS represent?
Change in entropy (disorder)
What does a negative ΔG indicate?
Spontaneous reaction
What does a positive ΔG indicate?
Nonspontaneous reaction
Why is ΔG useful?
It combines enthalpy and entropy to determine spontaneity
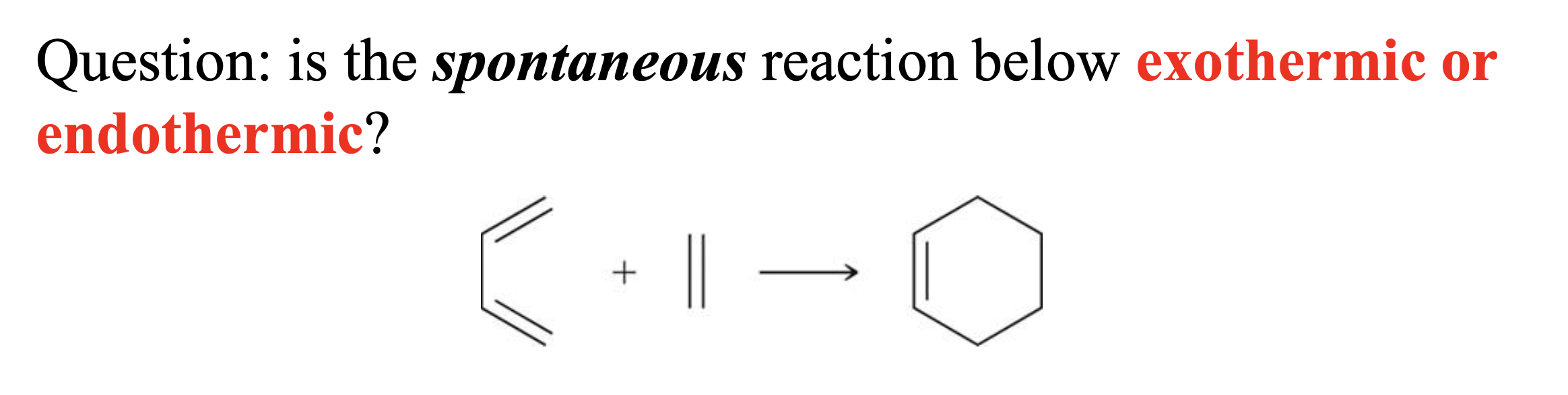
Exothermic
What does a negative ΔG indicate?
Spontaneous reaction
What is an exergonic reaction?
A reaction that releases free energy
Are spontaneous reactions exergonic or endergonic?
Exergonic
In an exergonic reaction, are products higher or lower in free energy?
Lower