6.1.1 Module 1 Overview: Physical and Chemical Changes
1/15
Earn XP
Description and Tags
This is based on the CHEM 125 Course of Waterloo. Applies to 2025,
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
In a physical change, …
there is no change in the chemical composition of the system
What are examples of physical changes?
temperature
pressure
volume
density
phase change
What is another name for the phase?
state of aggregation
What are some examples of physical transformations?
heating
cooling
expansion
compression
phase changes
What are examples of phase changes?
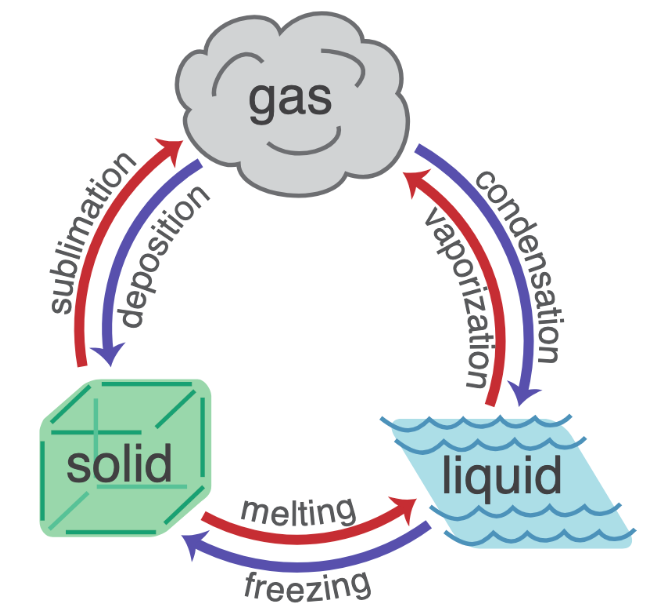
In a chemical change,…
the chemical composition of the system is changed by a chemical reaction
What are the 3 conditions that are true for both physical and chemical changes?
Mass is conserved
Energy is conserved (1st Law of Thermodynamics)
Entropy does not decrease (2nd Law of Thermodynamics)
System
a region of the universe that we are particularly interested in
Surroundings
a region outside that acts as a place to take measurements of the system
What are the types of systems?
Open
Closed
Isolated
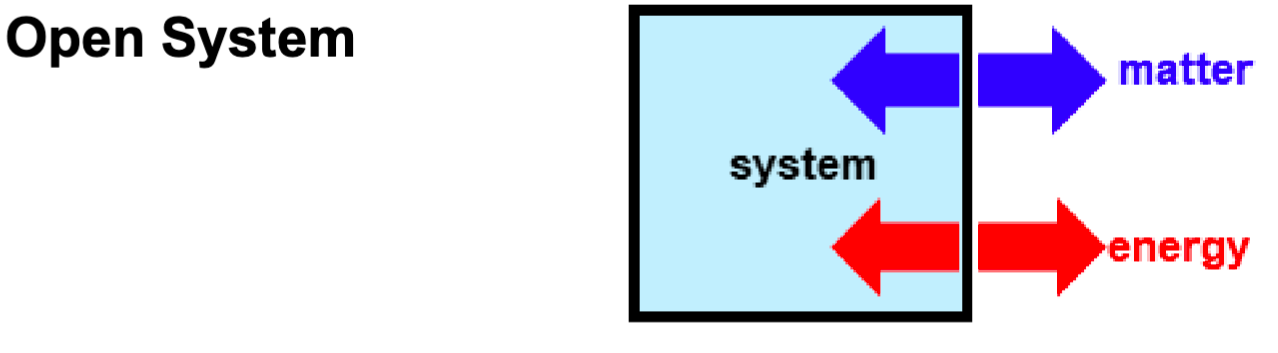
Open System
both matter + energy can be exchanged w/ surroundings
Closed Systems
only energy can be exchanged w/ surroundings
Isolated Systems
neither energy nor matter can be exchanged w/ surroundings
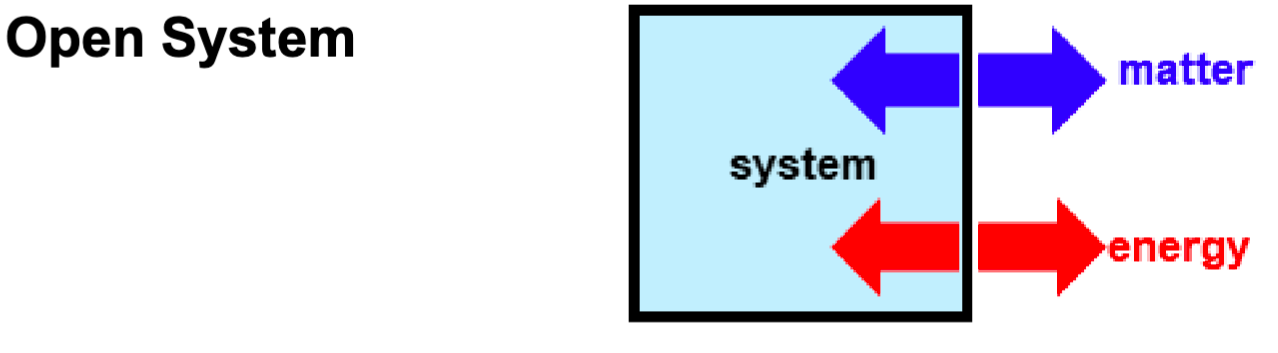
What is this an example of?
open system

What is this an example of?
closed system
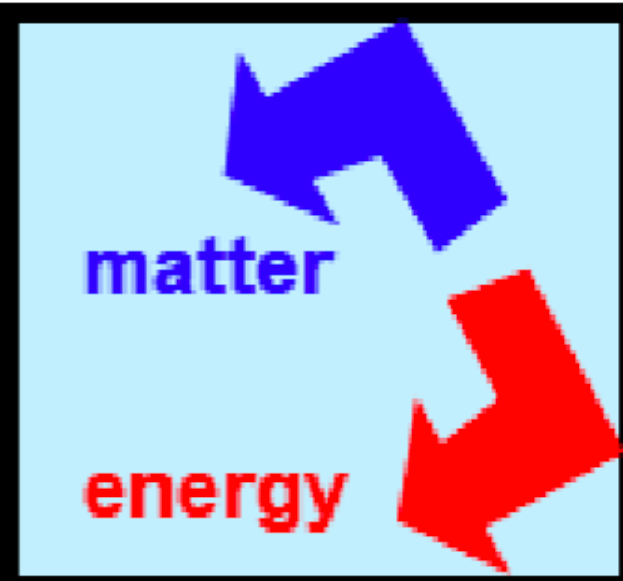
What is this an example of?
isolated system