Crude oil
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What is crude oil?
Crude oil is a mixture of hydrocarbons, compounds containing carbon and hydrogen only
It is a finite, non-renewable resource
Crude oil itself has no uses and therefore it has to be separated into ___ before it can be used.
How can we do this?
Fractions
We can use fractional distillation to separate crude oil into fractions.
This is carried out in an oil refinery
Each fraction consists of ____
groups of hydrocarbons of similar chain lengths
What determines in which fraction each hydrocarbon molecule will be separated into?
Its size and length
The size of each molecule is directly related to ____
Most of the compound in crude oil are from the same homologous series. Which one?
How many carbon and hydrogen atoms the molecule contains
Alkanes (single bonds only)
Describe how crude oil is separated into fractions by fractional distillation
Crude oil is vapourised/heated
The vapour enters the lower part/bottom of the column
There is a temperature gradient in the column
Accept cooler at the top and hotter at the bottom
The vapours rise up the column until they condense
At a height where the boiling point of the vapour is lower than the temperature in the column
Allow the fractions are separated according to their boiling point

Further explanation:
Key point
What is another name given to the ‘boiling point’?
Condensation point
The
Label the names and uses of the fractions obtained from crude oil
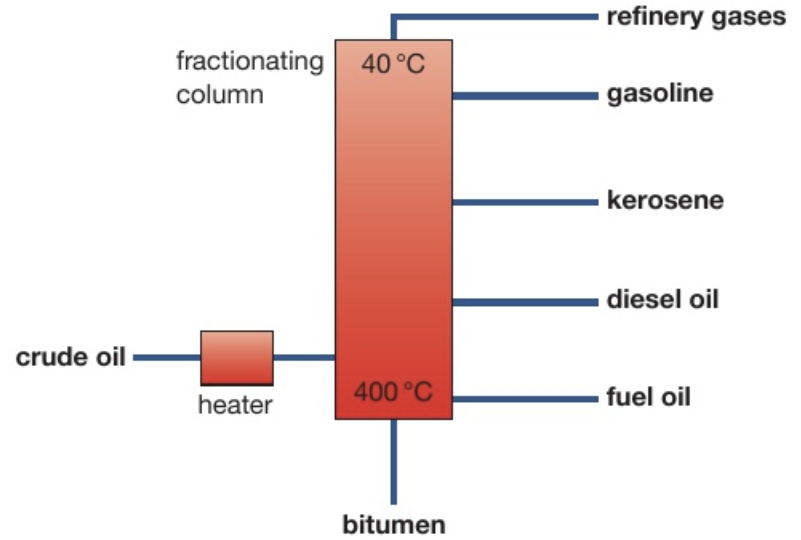
The hydrocarbons in the refinery gas fraction have very small masses and their BP are so low that the temperature of the column never falls low enough for them to condense to liquids
The temp of the column isn’t hot enough to boil some of the very large hydrocarbons found in crude oil so they remain as a liquid. These are removed as a residue from the bottom of the column
Uses of the fractions
All hydrocarbons can ___ in air to form ___ and ____ and release a lot of ___ in the process
Therefore the various fractions can be used as ___
All hydrocarbons can burn in air to form carbon dioxide and water and release a lot of heat in the process
Fuels
What is a fuel?
A fuel is a substance which, when burned, releases heat energy
What is another name given to refinery gases? Uses?
Liquefied petroleum gas
They are used for domestic heating and cooking
What is gasoline (petrol)? Uses?
As with all the other fractions, petrol is a mixture of hydrocarbons with similar boiling points
It is used for fuel for cars
Uses of kerosene?
Jet/aircraft fuel (main thingy) but also for domestic heating oil and as ‘paraffin’ for small heaters and lamps
Uses of diesel?
Fuel for buses, lorries, cars and some railway engines
Some diesel is also used for something else, what? What process does it need to undergo before?
Some is also converted to other more useful organic chemicals, including petrol in a process called cracking
Uses of fuel oil?
Used as a fuel for ships and power stations (savemyexams)/ and for industrial heating (textbook)
Uses of bitumen
It is melted and mixed with small pieces of rock for surfacing roads and roofs
In which fraction are the hydrocarbons containing up to 4 carbons?
Refinery gases are a mixture of methane, ethane, propane and butane
Trends for the main fractions of crude oil
As you go up the fractionating column, the hydrocarbons have:
Lower boiling points
Lower viscosity
Colour lightens
Not in ms but yes in mr wilson’s notes and savemyexams
Higher flammability (mr wilson)
More volatile (savemyexams)
Explain what happens to the boiling point as the molecules get bigger
BP increases
As the molecules get bigger, the intermolecular forces of attraction become stronger — VERY IMPORTANT PHRASE LEARN
More energy is needed to overcome these stronger intermolecular forces of attraction
Explain what happens to the volatility as the molecules get bigger
The liquids become less volatile
The bigger the hydrocarbon, the more slowly it evaporates at room temperature
As the molecules get bigger, the intermolecular forces of attraction become stronger
Explain what happens to the viscosity as the molecules get bigger
The liquids become more viscous and flow less easily
Liquids containing large molecules flow less easily because as the molecules get bigger, the intermolecular forces of attraction become stronger
What happens to the colour as the molecules get bigger?
What happens to the flammability as the molecules get bigger? What does this mean in terms of their utility?
The liquids become darker in colour
Flammability decreases (they do not burn as easily)
This limits the use of the bigger ones as fuels
En teoría combustion (todo lo de incomplete y eso) va en este topic pero no lo he puesto aqui JIJI
Key point
What are fossil fuels?
Fossil fuels include coal, gas and fuels derived from crude oil, which all come from things that were once alive
What are the environmental problems associated with the burning of fossil fuels from crude oil?
Most of the time they refer to ‘impurities’ so you should talk about SULFUR ONLY, however, CO2 is also released
Releases the greenhouse gas CO2. Greenhouse gasses trap the heat radiated from the Earth’s surface (originally from the Sun) which contributes to climate change → leads to sea levels rising, etc
Fuels from crude oil contain sulfur impurities. When the fuels are combusted, the sulfur gets oxidised and forms sulfur dioxide. Sulfur dissolves in water (reacts with water and oxygen) to form sulfuric acid and forms acid rain
Balanced chemical equations for sulfur dioxide and nitrogen dioxide to form acids.

What is acid rain?
Rain with a pH lower than 5.6 and is caused when water and O2 react with sulfur dioxide to produce sulfuric acid or with various oxides of nitrogen to give nitric acid
What is the location where they burn these fuels?
Apart from burning fuels from crude oil and releasing sulfur dioxide, what is another way that acid rain might form that is not from burning fuels? How?
Oxides of nitrogen and sulfur dioxide come mainly from power stations and factories
From car engines. The temperature reached in car engines is enough to allow nitrogen and oxygen from air to react, forming oxides of nitrogen
What is the solution to acid rain?
Removing sulfur from fuels, ‘scrubbing’ the gases from power stations and factories to remove SO2 and NOx and using catalytic converters in cars
Effects of acid rain
Leeches minerals out of the soil
Damages statues/rocks/buildings
Damages leaves preventing photosynthesis
Makes aquatic habitats too acidic for life