Nucleophilic addition to carbonyl groups
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
12 Terms
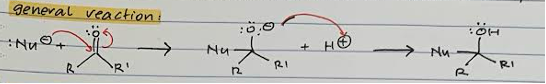
General reaction
Nucleophile attack on carbonyl C and transfer of proton from double bond to O
Protonation of -O with acid
Nucleophilic addition product
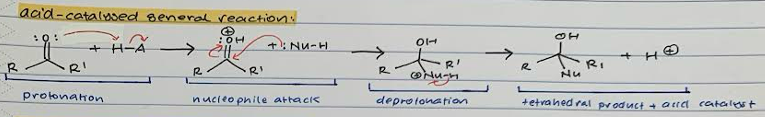
Acid-catalysed general reaction
Protonation of =O with acid.
Nucleophile attack on carbonyl C and transfer of proton from double bond to OH-
Deprotonation of nucleophile on carbonyl C
Tetrahedral product and regenerated acid catalyst
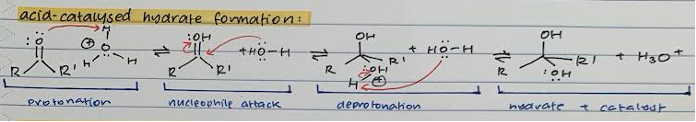
Acid-catalysed HYDRATE formation
Protonation of =O with acid (H3O+, H+ etc)
Nucleophile attack of H2O on carbonyl C and transfer of proton from double bond to OH-
Deprotonation of H2O with H2O
Hydrate product and regenerated acid catalyst
Base-catalysed HYDRATE formation
Nucleophile attack of OH- on carbonyl C and transfer of proton from double bond to O
Protonation of O- with H2O to give second OH group
Hydrate product with regenerated base catalyst

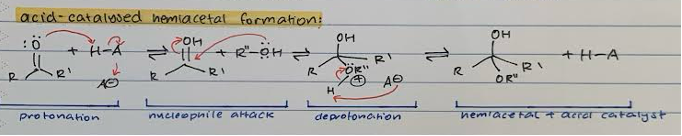
Acid-catalysed HEMIACETAL formation
Protonation of double bond O with acid catalyst to give OH
Nucleophile attack on carbonyl C with R’’-OH and transfer of proton from double bond to OH
Deprotonation of H from R’’-OH with A- from catalyst to give OR’’
Hemiacetal and regener
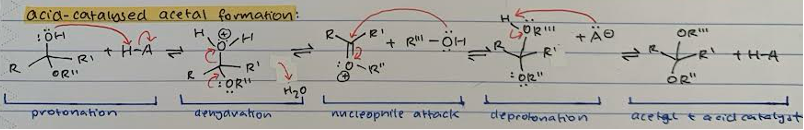
Acid-catalysed ACETAL formation
Protonation of hemiacetal OH group with acid catalyst
Dehydration, removal of H2O and transfer of proton from OR’’ to give double bond
Nucleophile attack with R’’’-OH on carbonyl C and transfer of proton from double bond to OR’’
Deprotonation of R’’’-OH with A- from initial protonation
Acetal product and regenerated acid catalyst
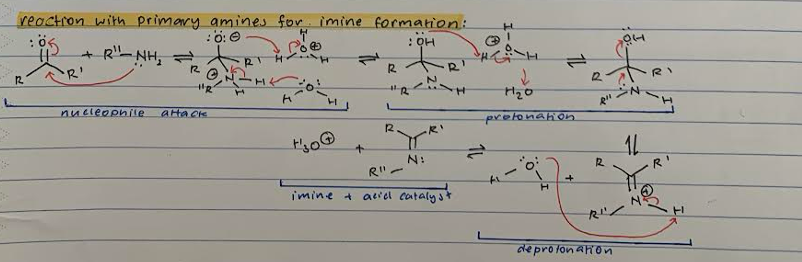
Reaction of PRIMARY AMINE for IMINE formation
Nucleophile attack of Carbonyl C with R’’-NH2 and transfer of proton from double bond to O
Protonation of O- with acid (H3O+) and deprotonation of R’’-NH2 with H2O
Protonation of OH with acid (H3O+) to release H2O. Transfer of proton from R’’-NH to make double bond and proton transfer to separate OH-
Deprotonation of R’’-NH with H2O
Imine product and regenerated acid catalyst
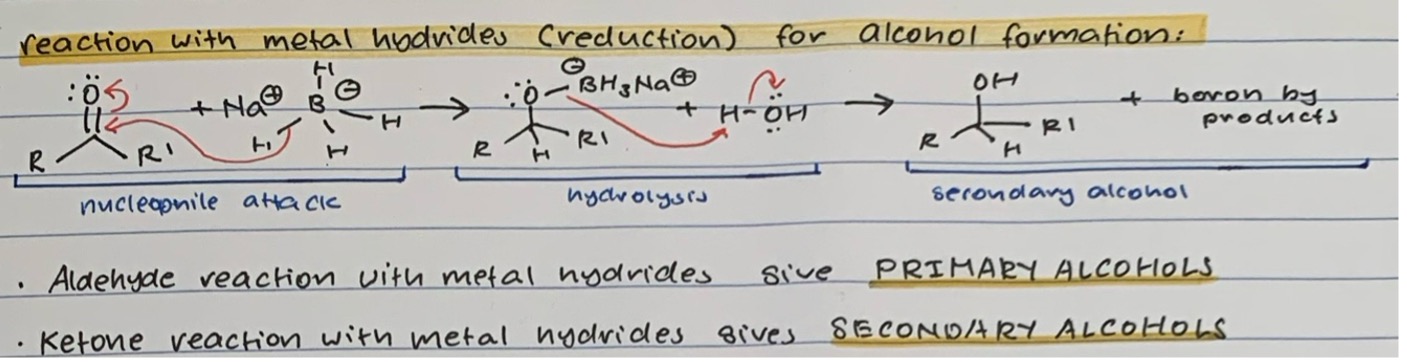
Reduction with METAL HYDRIDES for ALCOHOL formation
Nucleophile attack of carbonyl C with NaBH4 or LiAlH4 and transfer of proton from double bond to O
Hydrolysis of O-BH3Na+ with H2O to give OH
Secondary alcohol product
ALDEHYDE + metal hydride —> PRIMARY ALCOHOL
KETONE + metal hydride —> SECONDARY ALCOHOL
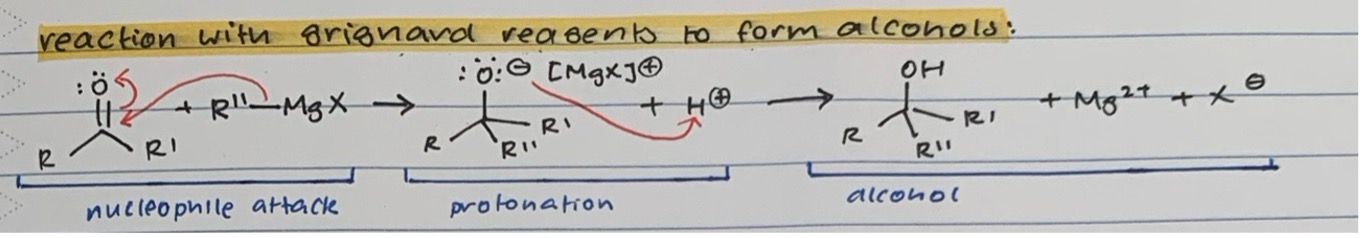
Reaction with GRIGNARD REAGENTS for ALCOHOL formation
Nucleophile attack of R’’-MgX on carbonyl C and transfer of proton from double bond to O
Protonation of O- with acid (H+)
Alcohol product
Aldehyde + Grignard reagent
Give primary alcohol. Other aldehydes from secondary alcohol.
Ketones + Grignard reagent
Gives tertiary alcohol
Carbon dioxide + Grignard reagent
Gives carboxylic acid (R-COOH)