Radiation Detectors
1/195
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
196 Terms
Are all chemical dosimeters active or passive?
passive
What are the two most well-known chemical dosimeters? Are they relative or absolute?
Fricke ferrous sulfate (absolute) and alanine (relative)
Radiolytic Yield
amount of quantity, x, produced from the incoming radiation
Fricke Ferrous Sulfate chemical dosimeter
One starts with an aqueous solution of ferrous (Fe2+) ions. Some of the radicals and molecular products resulting from exposure to ionizing radiation oxidize the Fe2+ ions into Fe3+ (ferric) ions.
What is the number of Fe3+ ions proportional to?
dose
Is the fricke solution water equivalent?
The Fricke solution is at least 96% water, making it a very water equivalent dosimeter.
What is signal yielded from the alanine free radical independent and dependent of?
The alanine free radical yields a signal that is dose dependent, yet is independent of the dose rate, energy, and is relatively insensitive to temperature and humidity changes.
Is the detector or the method of readout of an alanine chemical dosimeter sensitive to temperature and humidity?
Note that the method to read out alanine is sensitive to temperature and humidity, but the detector itself is not.
How are the fixed free radicals in alanine read out post irradiation?
electron spin resonance (ESR) or electron paramagnetic resonance (EPR) reader
How does an EPR reader work?
An EPR reader works by measuring the energy level transitions of unpaired (free) electrons at a specific resonant frequency within a variable magnetic field. The number of free radicals is proportional to the amplitude of the peak in an EPR spectrum.
One is the biggest advantage for using an alanine chemical dosimeter?
Its an organic compound. This suggests that the radiation-induced reactions in biological molecules are even closer to the radiation effects in living tissues than in water.
What is a set back for alanine chemical dosimters?
Maintaining an EPR system is costly and time consuming, which renders it difficult for other labs or clinics to maintain.
What does radiochromic film consist of?
Radiochromic film (RCF) consists of thin, microcrystalline monomeric di-acetylene emulsion coated on a flexible polyester film base.
How does radiochromic film work?
RCF is mostly translucent before irradiation, except for a slight yellow-ish tint due to a dye being added to prevent polymerization from visible or UV light. After exposure to ionizing radiation, the di-acetylene monomers undergo “progressive chain growth photopoly- merization”. This results in the formation of colored polymer chains that grow in length and the film turns blue.
How long does an RCF take to develop?
RCF continues to self develop for about 24 hours post irradiation and then stabilizes. This reduces its usefulness as an instant-gratification dosimeter.
How is the signal from a RCF obtained?
The signal from RCF is obtained from the color change, or darkening, of the film, which is proportional to the dose.
The amount of darkening of a RCF is determined through…
the transmission of light through the film
The amount of attenuation of a RCF due to ionizing radiation depends on…
the wavelength of the incident light
What are advantages of a RCF?
relatively low energy dependence
effective atomic number is ≈ 6.5
What are disadvantages of a RCF?
RCF tends to be sensitive to temperature and light.
A polarized densitometer affects the OD.
RCF tends to be nonlinear at higher doses.
Is calorimetry a form of absolute or relative dosimetry?
absolute
How do calorimeters work?
Calorimeters measure the heat deposited in a medium as a results of the ionizing radiation interactions in that medium
Why is graphite used instead of water for calorimeters?
A single Gy of absorbed dose to water yields a raise in temperature of only 0.239 mK. This is an extremely small temperature difference to measure
For graphite calorimeters, the dose to graphite must be converted into what?
dose to water
Are thermistors or thermocouples more sensitive?
Thermistors provide nearly an order of magnitude higher sensitivity than thermocouples
Do neutrons generate bremsstrahlung x-rays?
No
Are the ranges of secondary charged particles resulting from neutrons short or long?
Short
Why is the absorbed dose of neutrons effectively equal to kerma and why is the CPE well approximated?
neutrons do not generate bremsstrahlung x rays and the ranges of the heavy secondary charged particles are relatively short
What is the most probably energy for thermal neutrons?
0.025 eV
Epithermal neutrons
1 eV to 1 keV. Thermal neutron capture in hydrogen dominates in this range (resulting in γ rays)
Fast Neutrons
Neutron energy > 100 keV. Recoil protons resulting from elastic scattering of hydrogen nuclei dominates.
What are the five main processes by which neutrons interact with a material (the nuclei)?
• Elastic scattering
• Inelastic scattering
• Neutron capture
• Nuclear spallation
• Nuclear fission
Benefit of LiF TLD for neutron dosimetry?
LiF has an advantage of having some 6Li in it, which is highly favorable for a 6Li(n,alpha) reaction. The Hydrogen in a person’s body converts the higher energy neutrons to thermal neutrons, increasing the sensitivity even more.
How is are TLD materials made sensitive to fast neutrons?
neutrons by using a plastic or other hydrogen-containing layer in contact with the material.
What is one of the the most photosensitive devices used today?
PMTs
What is converted in a PMT? What is it converted to?
PMT converts light signals of no more than a hundred photons into a useable current pulse without adding a large amount of noise.
How is the sensitivity of a PMT described?
Quantum efficiency (# of emitted photoelectrons/ # incident photons)
What is the maximum range of quantum efficiency for common PMT photocathodes?
20-30%
For PMTs, what is the overall multiplication factor (δ) for a single dynode?
(# of secondary electrons emitted)/(# primary incident electrons)
For maximum amplification for a dynode, should the overall multiplication factor be large or small?
large
What is the electron transit time of a PMT?
The electron transit time of a PM tube is defined as the average time difference between the arrival of a photon at the photocathode and the collection at the anode. Transit times range from 20 - 80 ns.
Does transit time or spread in transit time determine the time width of the pulse of electrons arriving at the anode of the PMT?
spread in transit time
Compared with the interdynode distances, is the distance between the photocathode and the first dynode kept relatively large or small in order to allow for uniform collection over large photocathodes?
large
Why is magnetic shielding required for PMTs?
It prevent gain shifts of the PM tube.
Overall Luminous Sensitivity
Defined as the ratio of the measured anode current (mA) to the luminous flux from a tungsten light source of specified temperature. Units are (A / lm).
Cathode Luminous Sensitivity
Similar to Overall Luminous Sensitivity. But, but just the current due to the electrons leaving the cathode (not the multiplier).
Overall Radiant Sensitivity
Ratio of the anode current to radiant power at a given wavelength incident on the photocathode. Units are (A / W).
Dark Current
Normally specified in terms of anode current measured without illumination on the photocathode.
Anode Pulse Rise Time
The time it takes for the output pulse to rise from 10 to 90% of the peak.
Anode Pulse Width
The time width of the output pulse measured at half maximum amplitude.
SiPMT
• Array of tiny (tens of microns) Geiger-mode APDs
• Each APD has its own quench resistor, forming a microcell
• A SiPM device may include may thousand microcells
• SiPM optically coupled to scintillator
Should each microcell of a SiPMT be dependent of independent?
independent
SiPMT Advantages
higher photon detection efficiency
more compact
more immune to magnetic influence
operates at far lower voltages
easily integrated with readout electronics
What aspects of a PMT is better than for a SiPMT?
much lower dark current, and wider dynamic range
What do FACs measure?
FACs do not rely upon cavity theory. They directly measure exposure without a surrounding wall or phantom material.
What is the primary kerma measurement device for photon energies less than a few hundred keV?
FACs
For FACs, what does the radiation beam pass through and what helps define the volume?
radiation beam passes midway between two electrodes and the guards help define the volume
Why must the lateral dimensions of the FAC be large?
One of the key concepts of a free air chamber is to ensure CPE in the air. So it needs to accommodate electrons produced inside the volume of interest
Since FAC must be large enough to accommodate electrons produced inside the volume of interest, FACs are typically used below what energy?
400 keV
Does ΦA remain constant and equal to Φ0A0 throughout the length of the FAC?
Yes, the beam area A will conversely increase in proportion to the square of the distance from the source
How are scatter corrections for FACs commonly calculated?
Monte Carlo simulations
Why are large-volume, large-aperture FACs used?
Strength of sources for brachytherapy are quantified by air-kerma strength instead of activity. The air-kerma strength is a metric closely related to air kerma rate
As relative humidity increases, does (W/e)air increase or decrease?
decrease
As relative humidity increases, does ρair increase or decrease?
decrease

What is this?
FAC

Who is this bad boy?
Pancake G-M Tube
What region do most dosimetry ion chambers operate?
near-saturation (“ionization chamber”) region
As the voltage increases, why are there nonlinear effects that are introduced?
build-up of ions in the gas (i.e. space charge)
What region do nonlinear effects get introduced?
limited proportional region
Describe the Geiger-Meueller Region.
At very high voltages, the buildup of ions will reduce the electric field so that no additional gas multiplication can occur. Each pulse is the same magnitude.
What is a Townsend avalanche?
electron leads to an avalanche where drifting electrons can induce additional ionization in the gas
In a proportional counter, is the Townsend avalanche independent of all other avalanches?
Yes
In a G-M Tube, is the Townsend avalanche independent of all other avalanches? Why?
No. In the G-M tube, substantially higher electric fields are introduced, which increases the intensity of the avalanche. A single avalanche can itself trigger more avalanches at different positions within the tube.
Why are the pulses from a Geiger tube all of the same amplitude, regardless of the number of original ion pairs created?
Once a Geiger discharge occurs, the collective effect of all the avalanches come into play and terminates the chain reaction. This limiting point is always reached after about the same number of avalanches has been created.
What are the two main disadvantages of a G-M tube?
All energy information is lost and there is a large amount of dead time
What is the space charge effect?
When the concentration of ions is sufficiently high, and their presence reduces the magnitude of the E field in the vicinity of the anode wire.
What type of gases are mostly used for G-M tubes?
noble gases (non-electronegative)
Why is the counting efficiency for any charged particle that enters the volume of a G-M tube essentially 100%?
a single ion pair can form a pulse in a G-M tube
What is a common type of survey meter?
G-M Tube
Why are halogens mostly used as a quenching gas?
they can be replenished by spontaneous recombination unlike organic molecules
Why is a quenching gas used?
Slow, interactive positive ions formed mostly from the primary gas collide with neutral gas molecules and also some from the quench gas. When colliding with the quench gas with lower ionization potential, the original positive ion is neutralized and positive ion of quench gas begins to drift in its place. If there is enough quench gas, all ions that arrive at the cathode will be those of the quench gas.
What type of detector is this?
ionization chamber
What is the practical quantity of interest for ion chambers
the total number of ion pairs created along the track of the radiation
Is the average energy lost per ion pair formed is always less than or greater than the ionization energy?
Greater
Explain how an ion chamber works
Incoming radiation passes through a fill gas and ionizes atoms directly or indirectly, creating electrons and positively charged atoms. The resulting charges are collected by applying an electric field between electrodes
Are ion chambers operated in pulsed or current mode?
Both
What is an ion pair?
the resulting positive ion and free electron after a neutral molecule is ionized
What are the basic constituents of the electrical signal developed by the ion chamber? What is the practical quantity of interest?
Ion pairs; total number of ion pairs created along the track of the radiation
In order to permit ionization, what is the a minimum energy a particle must transfer?
energy greater than the ionization energy of the gas molecule (between 10 - 25 eV)
What is W?
The average energy lost by the incident particle per ion pair formed
What is W for air with incoming fast electrons?
33.97 eV
What is W for air with incoming alpha particles?
32.2 eV
What is the most important aspect for a relative dosimeter?
Its precision
Why is exposure significant?
The energy fluence Ψ is proportional to the exposure X for any given photon energy.
The mixture of elements in air is similar to soft biological tissue (in atomic number).
The value of collision kerma Kc in muscle, per unit of exposure X, is nearly independent of photon energy.
How is charge, exposure, collisional karma, and air karma related?
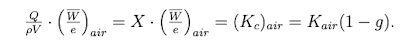
Whenever an external electric field is applied to the gas-filled region of an ion chamber, the electrons and ions will move…
away from their point of origin
For ions in a gas, at 1atm, and at a typical E field of 104 V/m, the drift velocity will be on the order of …
1 m/s
How much greater is the mobility of free electrons compared to ions?
1,000 times greater
Charge transfer collision
occurs when a positive ion encounters another neutral gas molecule. Electron is transferred, reversing the roles of each
Diffusion
the result of ions tending to diffuse away from regions of high charge density. More likely for electrons (than ions).