bio chapter 6
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
Living organisms constantly transform…
energy from one form to another
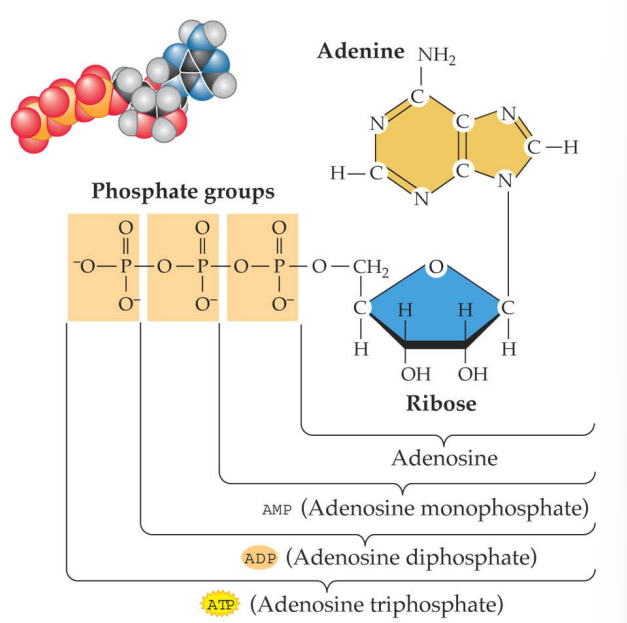
Adenosine triphosphate (ATP)
nucleotide that stores chemical energy in the bonds between its phosphate groups
each PO4- group has a net negative charge
breaking these bonds releases energy
formed by adding a phosphate group to ADP
In eukaryotic cells, mitochondria produce most _
every cell requires _ to power reactions that require energy input
Macromolecules transformation
carbohydrate → simple sugars (energy 4 calories/gram)
protein → amino acids (energy 4 calories/gram)
fat → fatty acids & glycerol (energy 9 calories/gram)
nucleic acid → nucleotides (not a significant source of energy for cells)
How do cells use subunits that are broken down
building blocks to make new macromolecules
energy to fuel cellular work
Whats the most energy dense molecule
fats
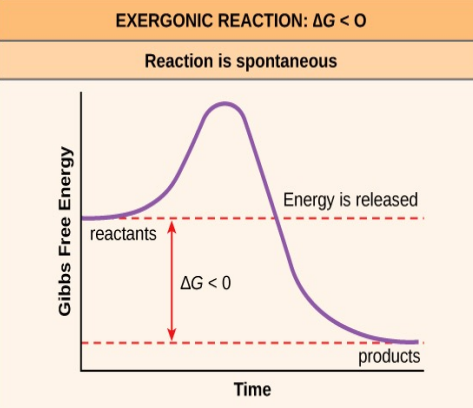
Exergonic reaction examples
cellular respiration
catabolism (breakdown)
electron transport chain (ETC)
breakdown of fuel, increases entropy
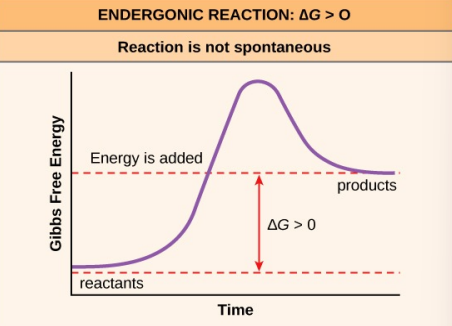
Endergonic reaction examples
active transport
cell movement
anabolism (constructing)
photosynthesis
build macromolecules, lowers entropy
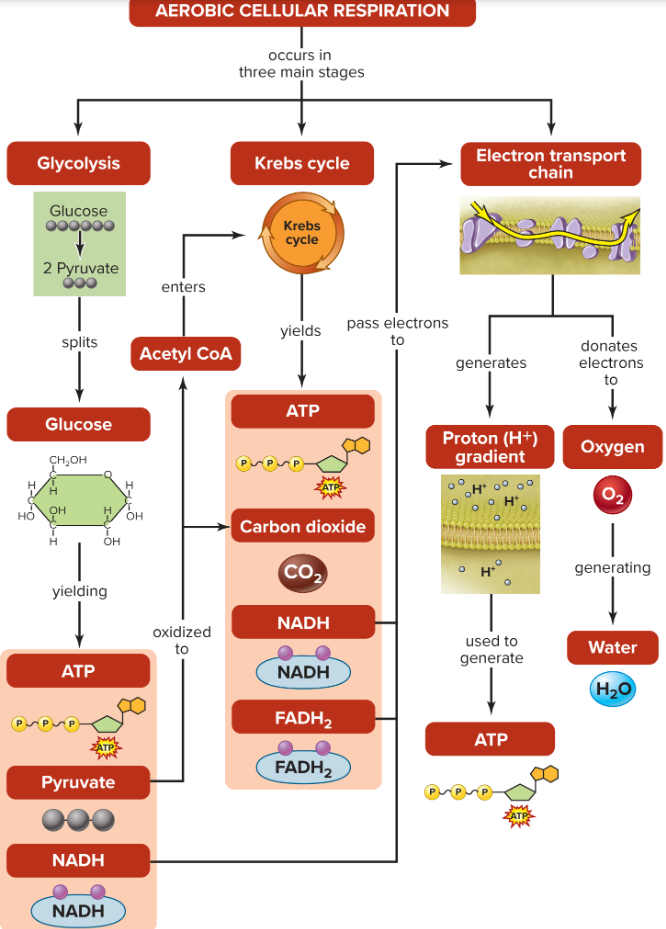
Cellular respiration
can be aerobic or anaerobic
harvests the potential energy stored in food molecules & uses the energy to make ATP
reactants: glucose & O2 consumed
products: CO2, water, and energy (ATP) released
1 molecule of glucose is processed = 36-38 molecules of ATP
formula: C6H12O6 + O2 → CO2 + H2O + ATP
aerobic formula: C6H12O6 + 6O2 + (36 ADP + 36 PO4) → 6CO2 + 6H2O + 36 ATP
necessary reactant: 6O2
waste products: 6CO2 + 6H2O
location: cytoplasm and the mitochondria of eukaryotic cells
Glycolysis begins in the cytoplasm
Subsequent aerobic stages, the Krebs cycle and electron transport chain, occur within the mitochondria.
series of redox reactions that release energy, which the cell uses to synthesize ATP
Glycogen carbohydrates provide
short-term energy storage
(excess simple sugars not immediately used for energy or cell structures are bound together in branching chains called _)
location: muscle and liver tissue
stores approx. 4 calories per gram
Triglyceride fats provide
long-term energy storage
(once glycogen stores have been filled, excess energy from dietary carbohydrates (simple suars), proteins (amino acids), and fats (fatty acids & glycerol) is stored as fat in the form of triglycerides)
location: fat cells
stores approx 9 calories per gram
Where does the energy to power formation of ATP from ADP come from?
breaking down glucose during cellular respiration or from sunlight during photosynthesis
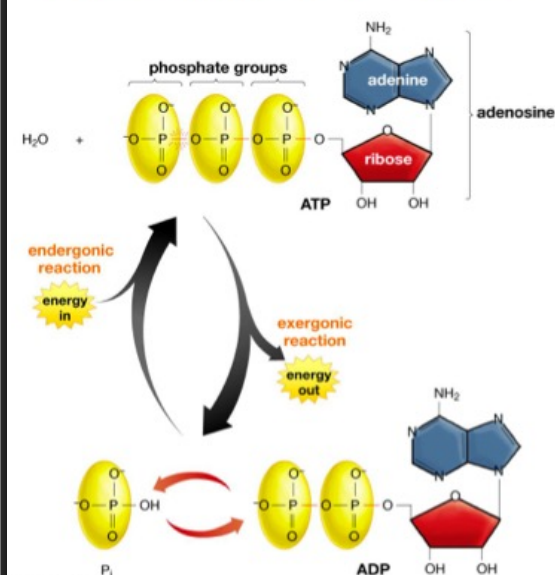
The atp/adp cycle
the fundamental process cells use to store, transport, and release energy
Energy Release (Hydrolysis): When cells need energy, ATP loses a terminal phosphate group through hydrolysis, breaking down into ADP and inorganic phosphate (P), releasing energy for processes like muscle contraction or nerve impulses.
Energy Storage (Phosphorylation): Through cellular respiration or photosynthesis, energy is used to add an inorganic phosphate back onto ADP, reforming high-energy ATP.
Location: This cycle mainly occurs in the cytoplasm and mitochondria of cells.
The Cycle: ATP → ADP + P + Energy (used for work); then, ADP + P + Energy (from food/sun) → ATP
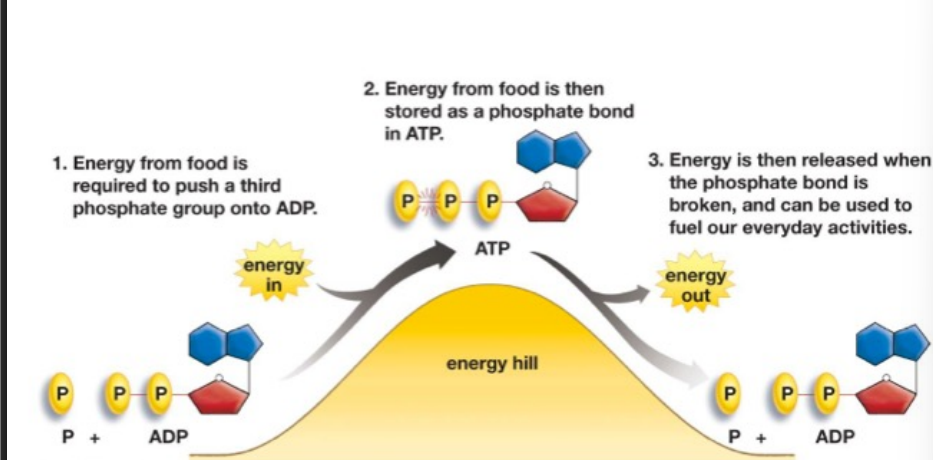
ATP synthesis
energy from food is required to push a 3rd phosphate group onto ADP (P + ADP)
energy from food is then stored as a phosphate bond in ATP
energy is then released when the phosphate bond is broken, and can be used to fuel our everyday activities (P + ADP)
Oxidation (-)
loss of electrons
Reduction (+)
gain of electrons
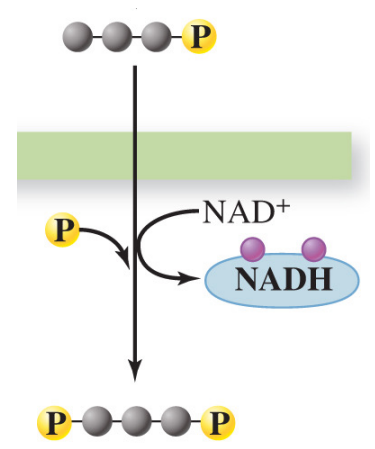
Redox reaction
combined reduction & oxidation
cellular respiration is a series of ___
Electron transfer
electrons (e^-) from high energy molecules are transferred to low energy molecules
Electron carriers (coenzymes)
carry out the electron transfer process
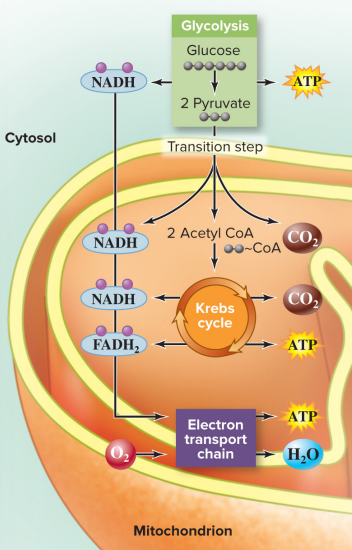
Cellular respiration proceeds in 4 stages
Glycolysis
Pyruvate oxidation
Krebs cycle (aka Citric Acid cycle)
Electron Transport Chain (ETC)
Good People Keep Empathy
Cellular respiration overview
glucose enters glycolysis: broken into glucose derivatives, 2 ATP produced
glucose derivatives are broken down in pyruvate oxidation / citric acid cycle (CAC): CO2 and 2 ATP produced
electrons enter the electron transport chain (ETC) (O2 is required): H2O and ~32 ATP produced
Energy efficiency
__ of cellular respiration is 37% - 37% of the energy stored in the bonds of a glucose molecule is stored in ATP, the rest goes to waste (as heat)
Glycolysis alone is 2% efficient
A typical car engine is 25% efficient (25% of the energy stored in the gasoline molecules is converted to mechanical energy)
Glycolysis “sugar-splitting” occurs in
cytosol
First step of cellular respiration
least efficient (only 2 ATP/glucose)
Oldest evolutionarily of the three steps
takes place in all living organisms
• Does NOT require oxygen
Can occur in the presence or absence of oxygen
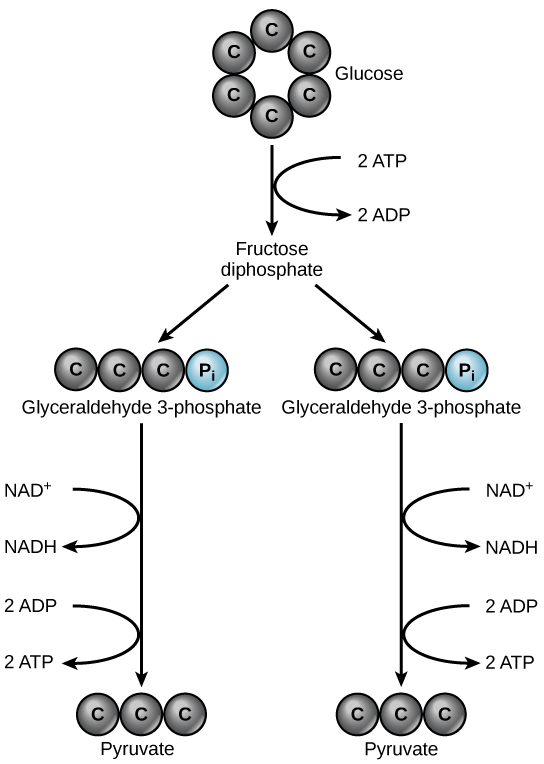
Glycolysis “sugar splitting”
Summary
Location: cytosol
Oxidized: the 6-carbon sugar glucose
Reduced: NAD^+ → NADH
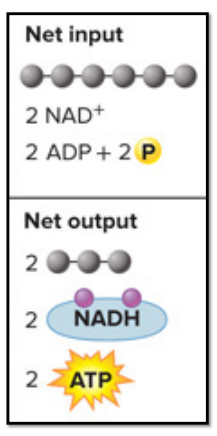
Net Input: glucose (6 carbon molecule), 2 NAD^+, 2 ADP + 2P (so 2 ATP)
Net Output: 2 pyruvates (two 3 carbon molecule), 2 NADH, 2 net ATP
“spend’ 2 ATP, synthesis 4 ATP: net gain of 2 ATP
Steps
2 ATP donates 2 phosphates (one each) to a glucose, which then becomes 2 ADP
Glucose (6 carbon molecule) splits into two 3 carbon molecules each containing 1 phosphate group
Each phosphate group is removed from the 3 carbon molecules, which produces 2 ATP for each so 4 total
During the process of removing the phosphate group, an electron is stored in a molecule called NADH (2 of them), which was orginally NAD^+
when NAD^+ takes on an electron (reduced), it becomes NADH
Extra notes
initially “spend”/invest ATP to begin this (hydrolyzed to 2 ADPs)
oxidation/reduction reactions
3 carbon molecules are oxidized
2 NAD^+ are reduced to 2 NADH
energy harvest
4 ADP are converted to 4 ATP
doesnt require oxygen (can occur in aerobic + anaerobic conditions)
glycolysis stores energy in the form of electrons inside NADH
How are evolutionary conservation and glycolysis connected
Glycolysis is highly conserved across all domains of life because it is an ancient, fundamental metabolic pathway for anaerobic energy production (ATP) that evolved before oxygen was present in Earth's atmosphere
Glycolysis is used by nearly all organisms (bacteria, archaea, and eukaryotes), indicating it was present in a common ancestor
Aerobic (O2) respiration
glucose + O2 = ATP
extracts energy/complete oxidation of glucose to CO2 in the presence of O2, producing ATP / captures energy from the oxidation of glucose and reduction of oxygen & stores the captured energy in the bonds of ATP
electrons stripped from glucose are used to reduce O2
formula: C6H12O6 + 6O2 → 6 CO2 + 6H2O + 36 ATP
products contain less potential energy than the reactants (exerogonic)
autotrophs like plants use this to generate ATP as well
Pyruvate oxidation
Summary
Pyruvate is oxidized → to acetyl-CoA (CO2 is waste)
NAD^+ reduced → NADH
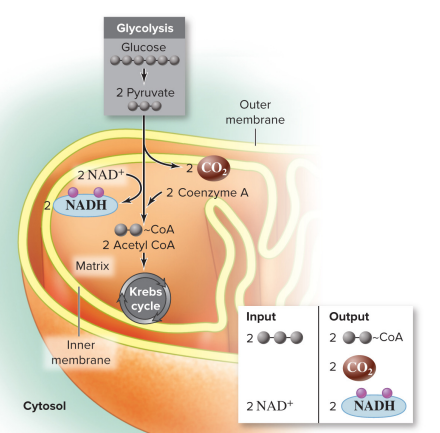
location: mitochondrial matrix in eukaryotic cells & cytosol in prokaryotes
Input: 2 pyruvates, 2 NAD^+, 2 Coenzyme A (CoA)
Output: 2 Acetyl CoA, 2 CO2, 2 NADH
Steps
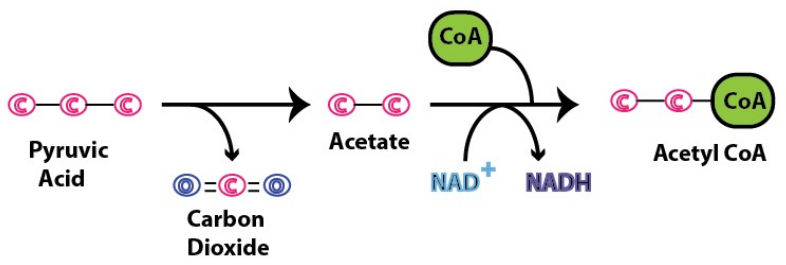
Decarboxylation (Remove CO2): A carboxyl group is removed from pyruvate, releasing CO2
Oxidation (Create NADH): The remaining two-carbon fragment is oxidized (acetate), and the electrons are transferred to NAD^+ forming NADH (reduced)
Formation of Acetyl CoA: The oxidized two-carbon acetyl group attaches to Coenzyme A, forming Acetyl CoA.
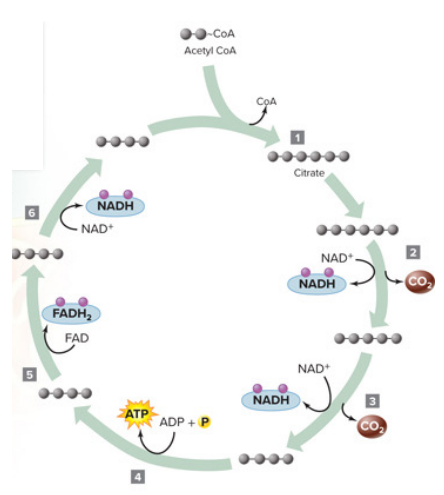
Krebs cycle (Citric Acid Cycle)
Captures/extracts the high energy electrons of acetyl CoA by oxidizing it / citrate → 2 CO2
Electron carriers NAD+ and FAD and reduced → NADH and FADH2
2 ATP
location: mitochondrial matrix in eukaryotic cells and cytosol in prokarytoic cells
does not directly use oxygen but it is an aerobic process that stops without it
Steps
Oxaloacetate (OAA) is a vital 4-carbon molecule (C4H4O5) reacts with acetyl-CoA to initiate energy production and creates citrate (6 carbon)
Coenzyme A is released and recycled to deliver more acetate
the acetyl group's two carbon atoms is oxidized to two molecules of carbon dioxide (CO2)
the electron carriers 3 NAD^+ and 1 FAD are reduced (gain electron) to 3 NADH and 1 FADH2 (per turn of the cycle)
Each molecule in the CAC is less energetic than its predecessor
Key Byproducts Per Cycle (One Turn/One Acetyl-CoA):
CO2: 2 molecules (waste product)
NADH: 3 molecules
FADH2: 1 molecule
ATP: 1 molecule
Key inputs per Glucose (Two Turns):
Acetyl-CoA: 2 molecules
NAD^+: 6 molecules
FAD: 2 mollecules
ADP: 2 molecules
+ Phosphate: 2
Key Byproducts Per Glucose (Two Turns):
CO2: 4 molecules (waste product, contains remaining carbon atoms from the og 6 carbon glucose molecule)
NADH: 6 molecules
FADH2: 2 molecules
ATP: 2 molecules
since 1 molecule of glucose produces 2 Acetyl-CoA molecules, the cycle turns twice per glucose, resulting in a total of 2 ATP molecules
For every glucose molecule that enters glycolysis, 2 citric acid cycles must take place bc…
glycolysis produces two molecules of pyruvate, which are then converted into two molecules of acetyl-CoA.
Each acetyl-CoA molecule enters the cycle separately, requiring two turns to process the full energy potential of one glucose molecule
Purpose of NADH and FADH2
carrying electrons to the electron transport chain
Proton gradient (H^+ gradient)
established by the ETC
an electrochemical difference in hydrogen ion concentration across a membrane, creating potential energy (proton-motive force) essential for powering cellular processes
primarily formed by proton pumps (ETC) in mitochondria, chloroplasts, and bacteria, driving ATP synthesis via ATP synthase
ATP synthase
enzyme that uses the potential energy in a proton (H^+) gradient to produce ATP
enzyme complex that admits protons through a membrane, triggering the production of ATP
uses the energy of a proton gradient to add a phosphate to ADP
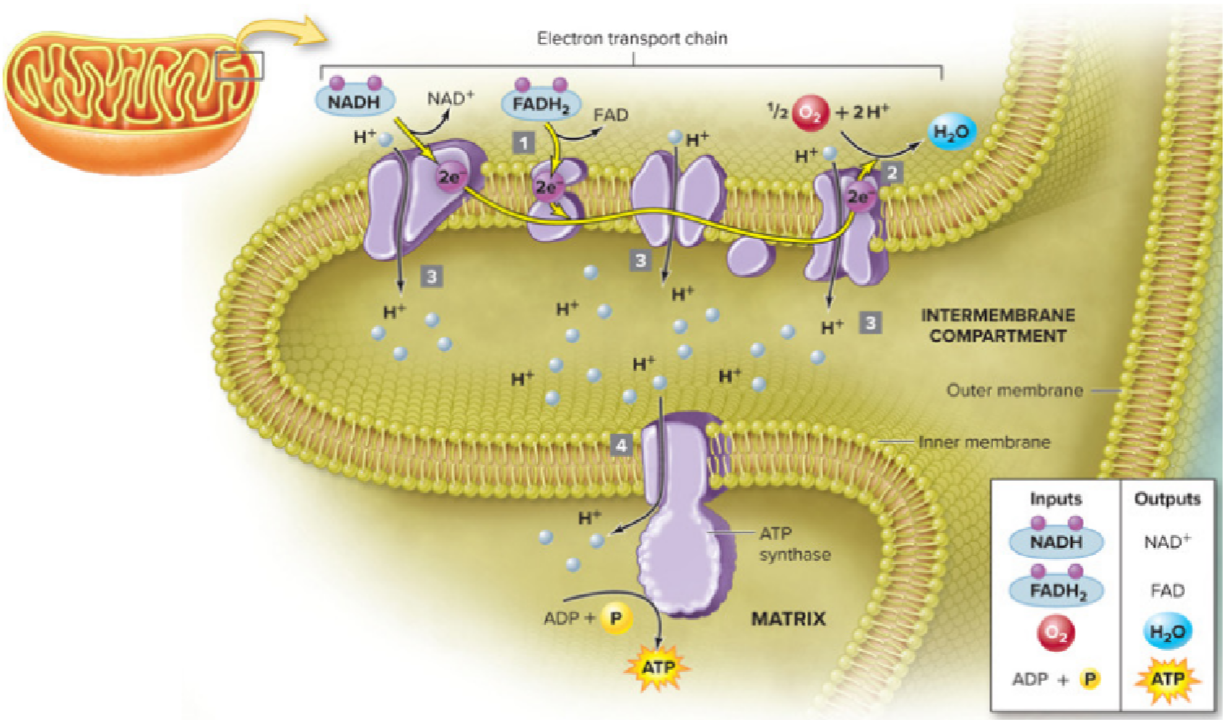
Electron transport chain (ETC)
Summary
Active transport of H^+
Proton gradient drives ATP synthesis via ATP synthase
Oxidized: NADH → NAD^+ and FAD → FADH2
Reduced: O2 → H2O
Location: inner mitochondrial membrane (cristae) in eukaryotes and the plasma membrane in prokaryotes
Inputs: NADH, FADH2, O2, ADP + P
Outputs: NAD+, FAD, H2O (waste), ATP (~32 to 34)
Extra
Membrane bound molecular complex that shuttles electrons to slowly extract their energy
harnesses the potential energy of NADH & FADH2, which donate electrons to proteins in this chain
The final step in cellular respiration, a series of redox reactions
Steps
NADH and FADH2 bring electrons harvested glycolysis, pyruvate oxidation and the CAC are oxidized: give their electrons to Enzyme Complexes. By accepting the electrons, Enzyme Complexes are reduced.
Enzyme Complex I gives up its electrons (becomes oxidized) to the next molecule in the ETC (Mobile protein I)
Each molecule in the ETC is lower in energy than its predecessor
As electrons move along a series of proteins, energy is released at each step (exergonic)
This energy is used to maintain proton gradient (that holds potential energy)
Pumps hydrogen ions from the matrix across the inner membrane of mitochondria against their concentration gradient (active transport)
Hydrogen ions are pumped from the inside to the outside, even though there are more hydrogen ions on the outside (going against the concentration gradient) (matrix → intermembrane compartment)
ATP synthesis using ATP synthase
the H^+ ions then spontaneously flow back across the membrane but only though a protein channel called ATP synthase (facilitated diffusion)
the passage of hydrogen ions through ATP synthase causes it to spin very rapidly
the kinetic energy from the spinning ATP synthase is used to attach a phosphate group to ADP, forming ATP
Oxygen is the final electron receptor in the ETC
Electrons from the ETC combine with H^+ and oxygen to form water, a byproduct of the ETC
if there is no oxygen, electrons have nowhere to go, ETC gets backed up and the whole process grinds to a halt. Long term results would result in no ATP molecules produced, and cells die
Once 1 molecule of glucose is processed, how many molecules of ATP are produced?
theoretically yields ~36 molecules
actual yield is abt 30 ATPs per glucose
Importance of respiration/breathing/oxygen
the O2 we breath is needed for the ETC to function
when we breathe, the oxygen enters the blood, and the blood takes it to all cells in the body
cells use the oxygen to synthesize ATP, producing CO2, and water as waste products, which are picked up by the blood
blood enters the lungs again, CO2 is expelled
What if oxygen is not around?
glycolysis is folllowed by fermentation
Fermentation
glycolysis occurs before this
produces significantly less ATP
2 ATP molecules per molecule of glucose
NADH transfers electrons to pyruvate and is oxidized to NAD^+, which can then be used in glycolysis
* remember, we normally recycle NADH to NAD^+ using the ETC
no oxygen = we dont use ETC
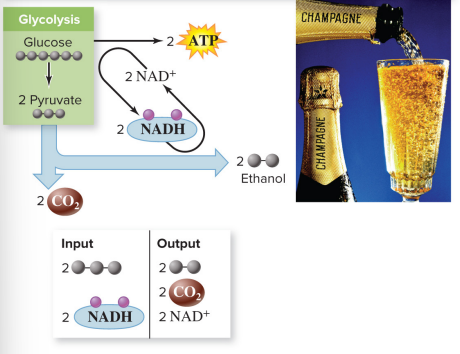
Alcoholic fermentation
metabolic pathway in which NADH reduces the pyruvate from glycolysis, producing ethanol and CO2
Input: 2 pyruvates, 2 NADH
Output: 2 ethanol, 2 CO2, 2 NAD^+
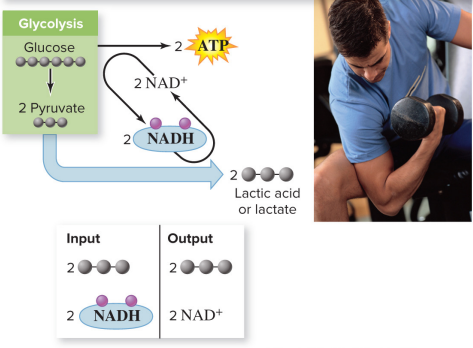
Lactic acid fermentation
metabolic pathway in which NADH from glycolysis reduces pyruvate, producing lactic acid or lactate
Input: 2 pyruvates, 2 NADH
Output: 2 lactic acid/lactate (waste), 2 NAD^+
Photosynthesis comparison
Food: produced
Energy: stored as glucose & other sugars
Light: required
H2O: consumed
CO2: consumed
O2: released
Equation: 6CO2 + 6H2O + Light Energy → C6H12O6 + 6O2
Respiration comparison
Food: consumed
Energy: released from glucose & other food molecules
Light: not required
H2O: released
CO2: released
O2: consumed
Equation: C6H12O6 + 6O2 → 6CO2 + 6H2O + ATP
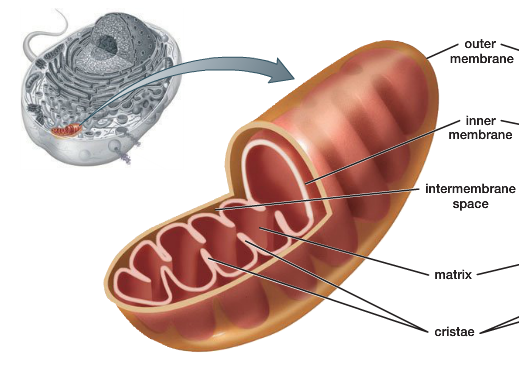
Mitochondria
powerhouse of the cell
converts the energy in food into a form that the cell can use to power its normal activites
contain own DNA
generate most chemical energy (ATP) for eukaryotic cells
each _ has 2 membranes enclosing a central matrix (the inner compartment of a _)
Intermembrane compartment
the space between a mitochondrion’s two membranes (between the outer & inner membrane)
Cristae
folds of the inner membrane & they greatly increase the surface area of the inner membrane
house electron transport chain proteins
these proteins pump protons into the intermembrane compartment, ATP synthase also spans the inner membrane
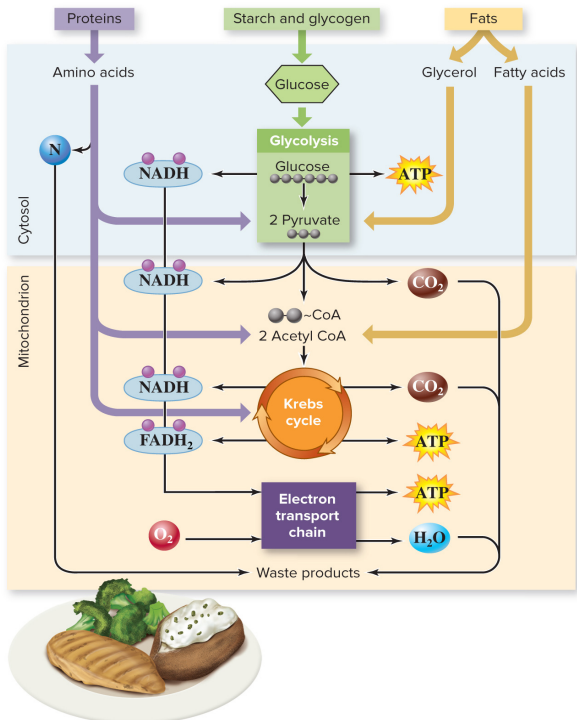
How does other food molecules (proteins, fats, carbohydrates) enter the energy extracting pathways?
Polysaccharides (starch/glycogen)→ glucose
Amino acids → pyruvate, acetyl CoA, or an intermediate of the Krebs cycle
Fatty acids → acetyl CoA
Glycerol → pyruvate
Anaerobic respiration
the process of breaking down sugars to generate energy (2 ATP per glucose) without oxygen
occurring in the cytoplasm
essential for organisms in low-oxygen environments and provides quick energy during intense exercise, yielding products like lactic acid in humans or ethanol/CO₂ in yeast
it does not use oxygen as the final electron acceptor, relying instead on inorganic molecules (like sulfate or nitrate) or fermentation to produce energy
final electron acceptors: NO3-, SO4²-, CO2
Mitochondrial matrix
the fluid enclosed by the inner membrane of a mitochondrion
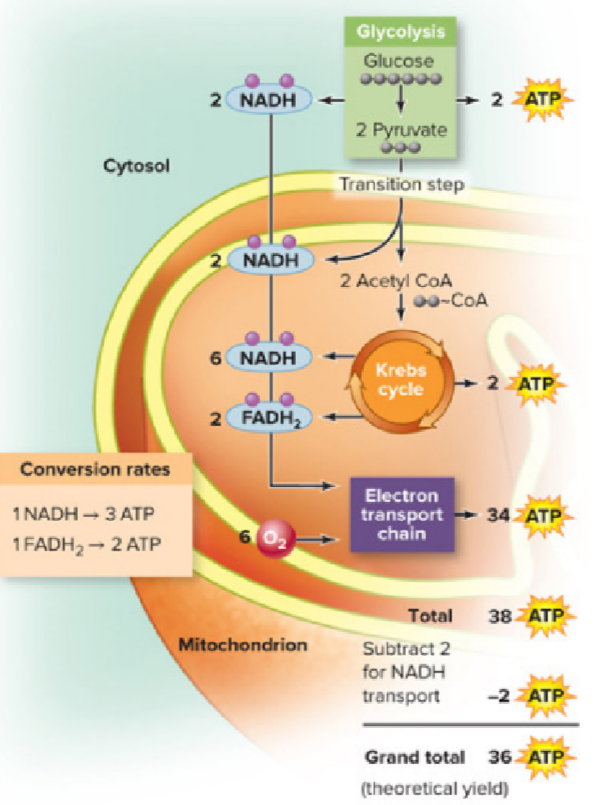
Before the NADH and FADH2 produced during glycolysis & the krebs cycle enter the ETC, the net number of ATP molecules produced per glucose molecule is…
4 molecules
Energy pathways that do not require O2 to generate ATP
fermentation (with glycolysis)
anaerobic respiration
What has been produced from the og gluclose molecule after glycolysis, oxidation of pyruvate, & the krebs cycle, but before molecules enter the electron transport chain
CO2
NADH
FADH2
ATP
Aerobic cellular respiration requires that organisms need to acquire _ that can diffuse into their cells & need to eliminate _ that diffuses out of their cells
O2; CO2
The rearrangement and oxidation of intermediates in the Krebs cycle transfer electrons to form __ and eventually recreate a 4-carbon molecule that allows the cycle to repeat
NADH and FADH2
How much ATP is generated/used during the passage of 1 molecule of glucose (net theoretical total production)?
36 ATP
How much ATP is generated/used during the passage of 1 molecule of glucose (glycolysis)?
produces 4 ATP but also uses 2 ATP; so net 2 ATP are produced
How much ATP is generated/used during the passage of 1 molecule of glucose (Krebs cycle)?
2 ATP generated
How much ATP is generated/used during the passage of 1 molecule of glucose (electron transport)?
34 ATP generated
How much ATP is generated/used during the passage of 1 molecule of glucose (moving NADH from glycolysis into mitochondrion)?
2 ATP used
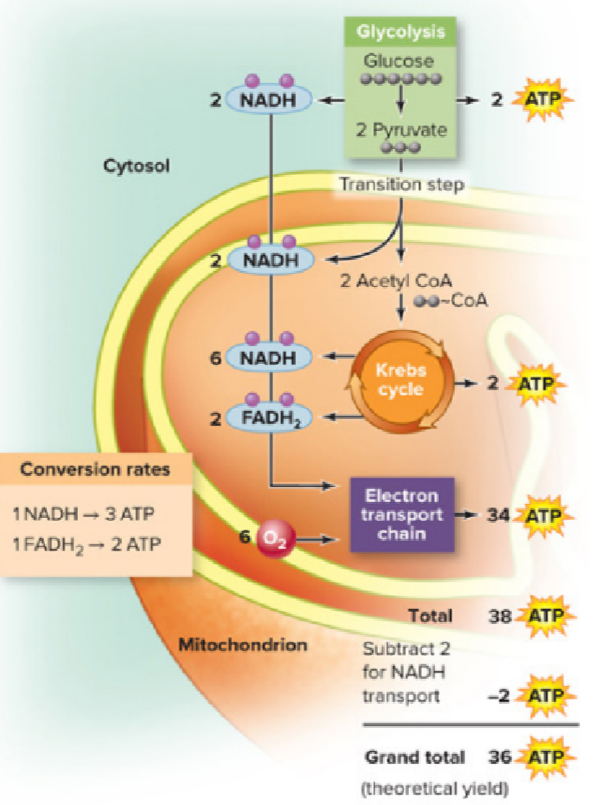
What causes the difference between the theoretical & actual yields of ATP in aerobic respiration?
protons leak across the inner mitochondrial membrane without using ATP synthase
ATP is spent transporting ADP & pyruvate into the mitochondrial matrix
2 molecules ATP must be used to transport NADH produced in glycolysis into the mitochondrion, the net theoretical yield of aerobic respiration is 36 ATP molecules
When carbohydrate supplies are depleted in cells, amino acids from proteins can enter aerobic respiration after _ is removed from the amino acids and excreted
nitrogen
What can be a final electron acceptor in anaerobic respiration
NO3-
SO4²-
CO2
What are the products of glycolysis?
2 NADH
2 ATP
2 pyruvates (two 3 carbon molecule)
What are the products of pyruvate oxidation?
2 NADH
2 Acetyl CoA
2 CO2 (waste)
releasing one CO2 per pyruvate
What are the products of the Krebs cycle?
6 NADH
2 FADH2
2 ATP
4 CO2
waste product, contains remaining carbon atoms from the og 6 carbon glucose molecule
What are the products of the Electron Transport Chain?
ATP (~34)
NAD+
FAD
H2O (waste),
Amount that energy yield is lowered due to leaking protons & shuttling pyruvate
6 ATP molecules
What is NADH oxidized?
NAD^+
looses electrons
What is NAD^+ reduced?
NADH
gains electrons
What is FADH2 oxidized?
FAD
looses electrons
What is FAD reduced?
FADH2
gains electrons
What is oxidized/reduced in glycolysis?
Oxidized: the 6-carbon sugar glucose
Reduced: NAD^+ → NADH
Location: Cytosol
What is oxidized/reduced in pyruvate oxidation?
Oxidized: 3-carbon molecule pyruvate
Reduced: NAD^+ → NADH
Location: Mitochondrial matrix
What is oxidized/reduced in Krebs cycle?
Oxidized: Acetyl-CoA (into 2 molecules of CO2)
Reduced: NAD^+ → NADH and FAD → FADH2
Location: Mitochondrial matrix
What is oxidized/reduced in Electron Transport Chain (ETC)?
Oxidized: NADH → NAD^+ and FADH2 → FAD
Reduced: O2 → H2O
Location: Cristae (inner mitochondrial membrane)