IB Biology HL: Enzymes
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Metabolism
All of the chemical reactions that take place in an organism
Metabolic Pathway
A series pf enzymes involved in creating an end product. Respiration and photosynthesis are examples.
Catabolic Reaction
macromolecules broken down into small molecules
ATP released
eg. hydrolysis
Anabolic Reaction
macromolecules created from smaller molecules
ATP used
eg. condensation
Pectinase
Enzyme used to increase volume of juice from fruit
Lactase
Enzyme used to convert lactose into glucose and galactose
pH,Tempatures, and Salinity
Environmental factors that can affect the function of an enzyme
Substrate
Molecule that will be changed by an enzyme
Decribe the steps on enzyme action?
substrate binds to active site due to chemical specificity
bonds in substrates are weakened causing it change
products seperate
How are immbile enzymes used in Lactose free milk?
milkcontaing lactose poured through container with alginate beeds with immobilised lactose
lactose converted into glucose as milks flow through
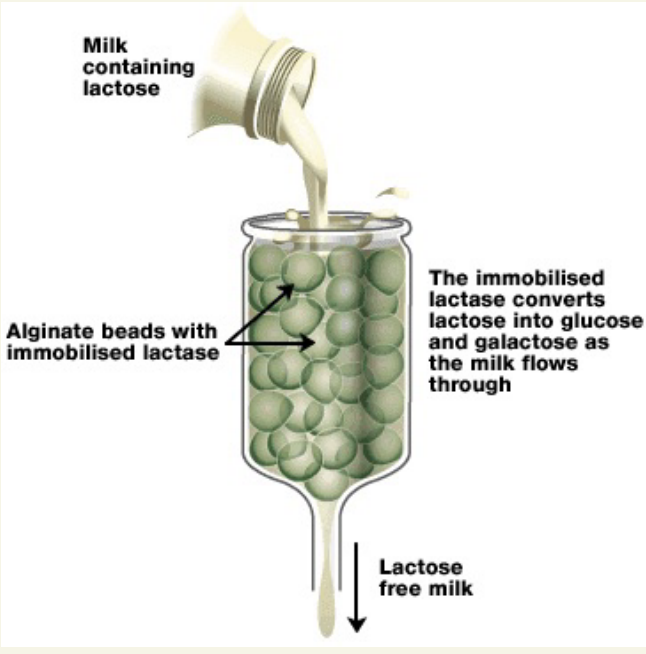
Benefits of immobilised enzymes:
can be reused multiple times
cheaper
time-efficient: no need to separate enzyme from substrate
higher tolerance to pH and Temp
Denaturation
The loss of tertiary structure of an enzyme causing it to no longer function
Temp: vibration break intermolecular bonds - permanent
pH: break hydroden bonds
Activation Energy
the minimum energy it takes for a reaction to occur
enzymes decrease this
Intracellular uses of enzymes:
krebs cycle inside mitochondria
enzymes produced by free ribosomes
Extracellular uses of enzymes:
digestion of food in the small intestine
enzymes from the ER released in the glands
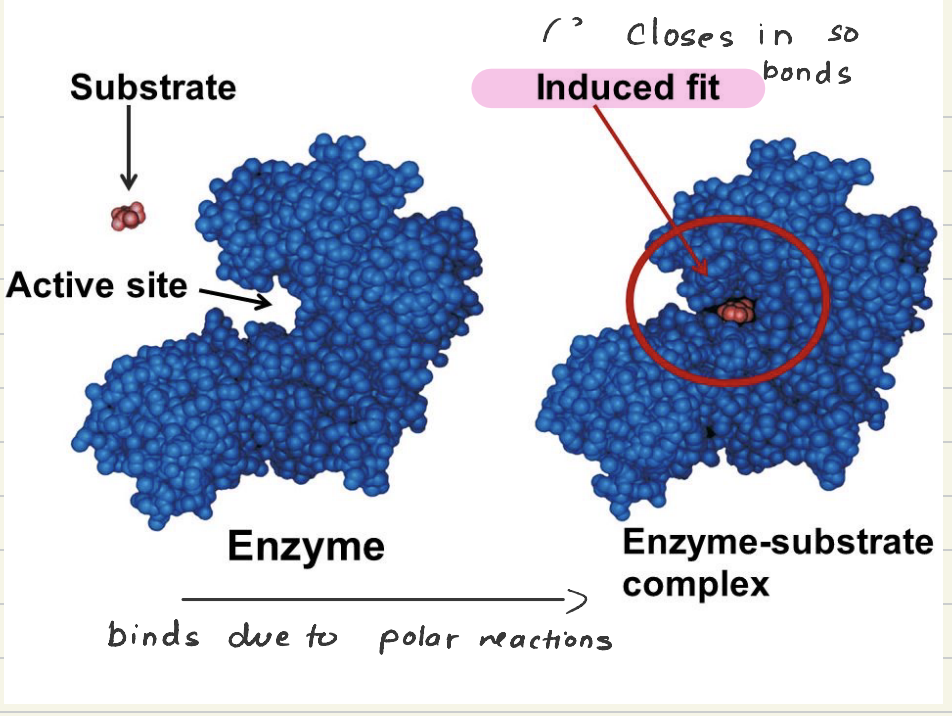
Induced Fit Model
The active site of an enzyme is flexible and can change shape to better hug the substrate
substrates initially bind due to polar reactions
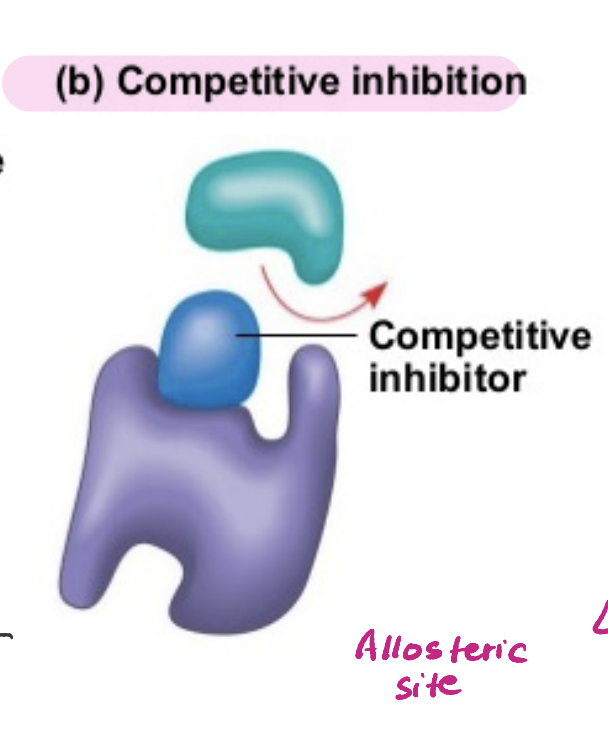
Competitive Inhibition
An inhibitor binds to the active site of an enzyme to reduce or stop its function.
Increasing substrate concentration can overcome the effects of this
Allosteric Activation
An allosteric molecule must bind to the allosteric site in order to change the shape of the active site on an enzyme, so that the substrate can fit and the reaction can occur
Lock & Key Model
The active site of an enzyme is rigid and does not change shape
Allosteric Site
Location AWAY from the active site where a substance may bond in order to effect an enzymatic reaction
Feedback Inhibition
The end product of a reaction acts as an inhibitor to the initial enzyme in the reaction
Coenzyme
Organic substance that facilitates the activity of an enzyme
Cofactor
Inorganic substance that facilitates the activity of an enzyme
Non-Competitive Inhibtion
Substance attaches to an allosteric site in order to slow or stop an enzymes function
Endothermic Reaction
Reaction in which the products have more energy than the reactants
Exothermic Reaction
Reaction in which the reactants have more energy than the product
Globuilar Proteins
Describes the tertiary shape attained by all enzyme proteins
Lock & Key vs. Induced Fit Models
Lock & Key is the idea that only the correct sized substrate (key) fits into the active site (key hole). So only the correct key(substrate) opens a certain lock (enzyme).
Induced Fit Model is the idea that the substrate plays a role in determining the final shape of the enzyme and that the enzyme is partially flexible. Only the proper substrate is capable of inducing the proper alignment of the active site.
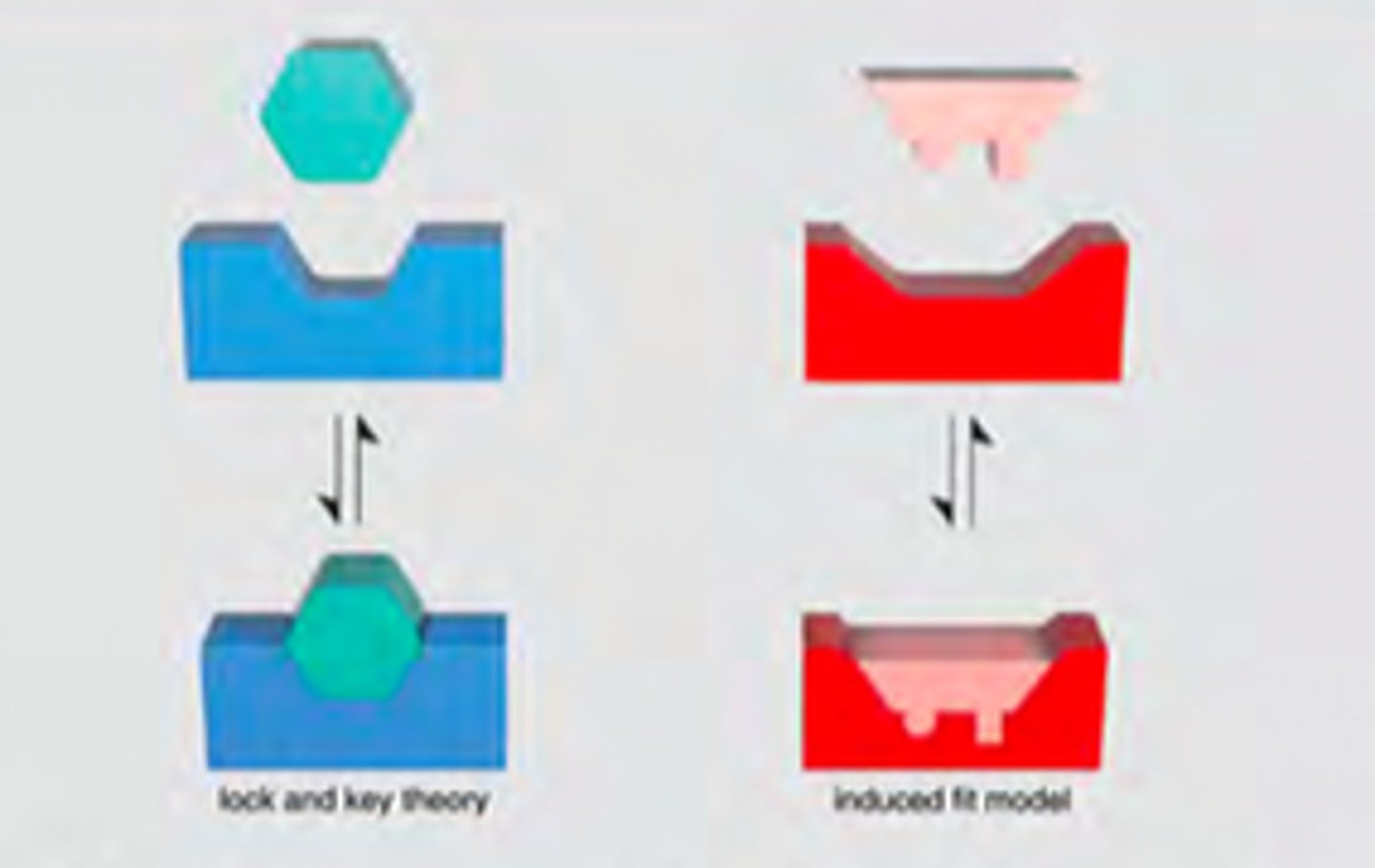
Comparison of Lock & Key and Induced Fit Model
Both agree that only one substrate will work when it meets the active site of the enzyme.
Both require an enzyme and a substrate.
Catabolic vs. Anabolic Pathways
Catabolic reactions break down molecules and release energy by breaking down complex molecules to simpler compounds.
Anabolic reactions build larger molecules from smaller ones, they consume energy to do this.
Similarities: Both are metabolic pathways
Cofactors vs. Coenzymes
A cofactor is a non-protein chemical compound. "helper molecule".
Coenzymes are cofactors that are bound to an enzyme loosely. A coenzyme is a small, organic non-protein molecule. It carries chemical groups between enzymes.
Competitive Inhibitors (active site) vs. Non- Competitive Inhibitors (allosteric site)
Competitive:
substrate and inhibitor chemically similar
inhibitor can bind with the same active site and make it not free for substrate
substrate conc can still increase rate
Non-competitive:
substrate and inhibitor diff
inhibitor binds to an allosteric site
changes shape
substrate onc cna’t speed dup rate
Both:
enzymatic reactions that slow down the rate
both involve reversible + irreversible inhibitors
Substrate analogue:
molecules that are able to form covalent bonds with the active site of an enzyme
How to antibiotics works?
Bacteria cell walls made of peptidoglycans
peptidoglycans held together by crosslinks
when new cells grows, enzymes known as autolysins produced
these create small strechy holes in cell wal so cross links can form
penicillin inhibits DD transpeptidase that catalyse crosslinks formation
no crosslink but lots of holes
bacteria in watery env. so lysis
Activation energy
the minimum amount of energy that is needed to start a chemical reaction/ difference between the energy of the reactants and the maximum energy.
Basic Characteristics of Enzymes
Reusable and specific
Speed up chemical reactions by lowering the activation energy needed to start the reaction
When put with high temperatures it denatures
Enzymes are proteins and each has its own active site.
Enzymes are affected by the acidity or alkalinity of the solutions therefore every enzyme has a different optimum pH. At extreme pH conditions the enzyme denatures
At what temperature is enzyme activity the greatest
37 degrees Celsius
The substance with which an enzyme reacts
Substrate
Enzyme actions can be ____
Reversible
Allosteric Inhibition vs. Activation Process
In warm up
Lactase
Lactase breaks lactose (the sugar in milk) into one glucose and one galactose molecule.
Pectinase
an enzyme that breaks down pectin, a polysaccharide found in plant cell walls. Commonly referred to as pectic enzymes
Amylase
any of several digestive enzymes that break down starches
(saliva glands)
Reversible Inhibiton
inhibition of enzyme activity in which the inhibiting molecular entity can associate and dissociate from the protein's binding site.
Permanent (irreversible)
the action of an inhibitor such that, once bound to a protein, it cannot dissociate.
A new antibiotic has been developed that will use competitive inhibitor enzyme inhibition. This means that the...
Antibiotic will compete for substrate binding sites on the enzyme
Which variable has the least affect on enzyme activity?
Light intensity
Enzymes are very specific in their choices of substrates because each different enzyme has an active site that....
is shaped to fit a certain substrate molecule
The site where molecules other than the substrate bind in an enzyme to alter its activity are called....
Allosteric Site
In an enzyme catalyzed reaction the reactants are called the...
substrate...duh
When the substrate is bound to the enzyme, the shape of the enzyme may change slightly, leading to this
A better induced fit
At the conclusion of an enzyme catalyzed reaction, the enzyme...
frees itself from the product and is ready to be reused.
The ability of an enzyme to catalyze a reaction is not affected by...
excess cofactor.
The temperature is raised slightly above the optimum for a specific enzyme. What would you expect
to observe as a result of the change in temperature?
decreased enzyme activity
Allosteric inhibitor binds here; changes enzyme shape.
non-catalytic binding site