BIMD 301 Unit C
1/200
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
201 Terms
Cell Membrane
the semipermeable membrane surrounding the cytoplasm of a cell
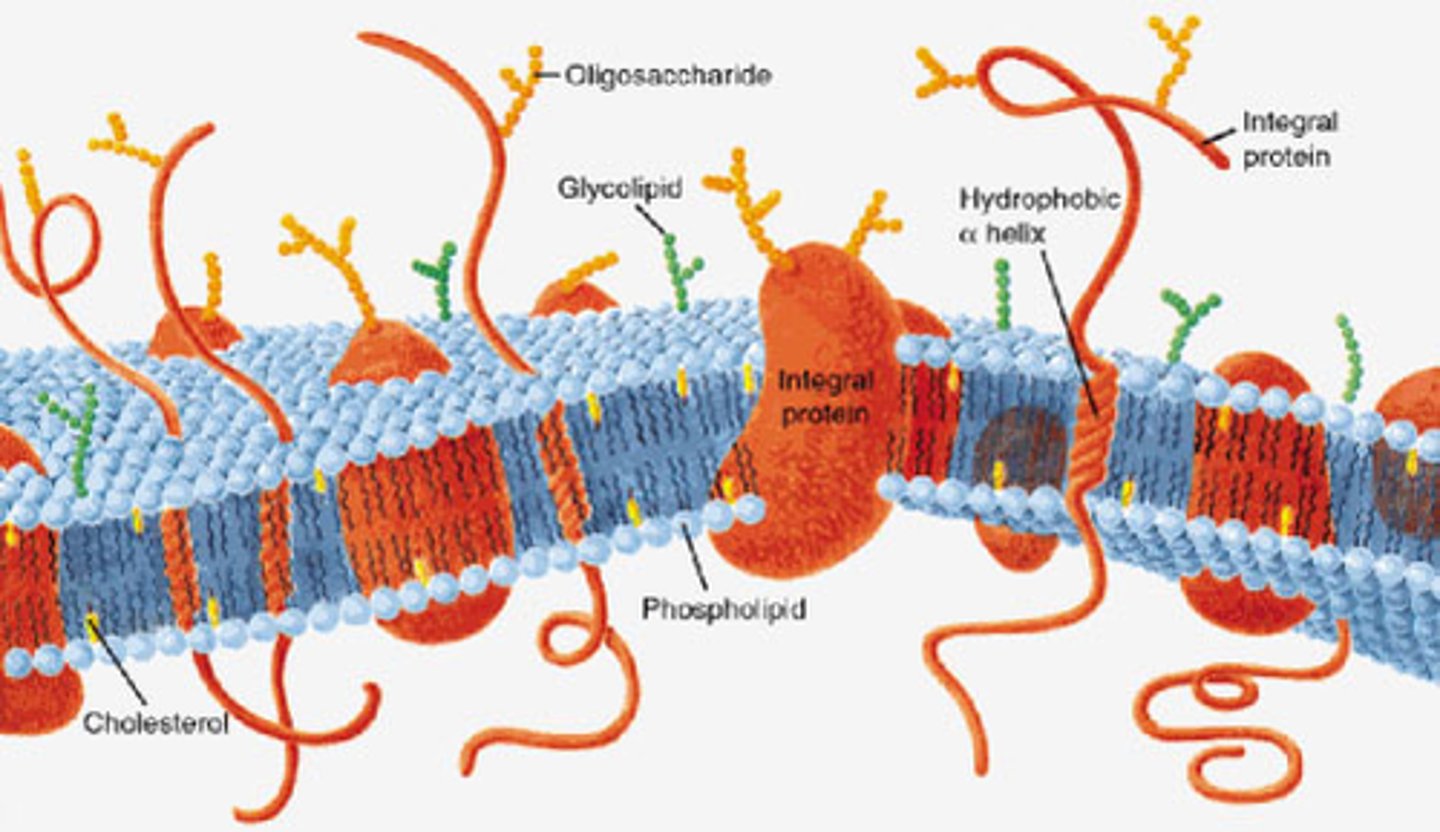
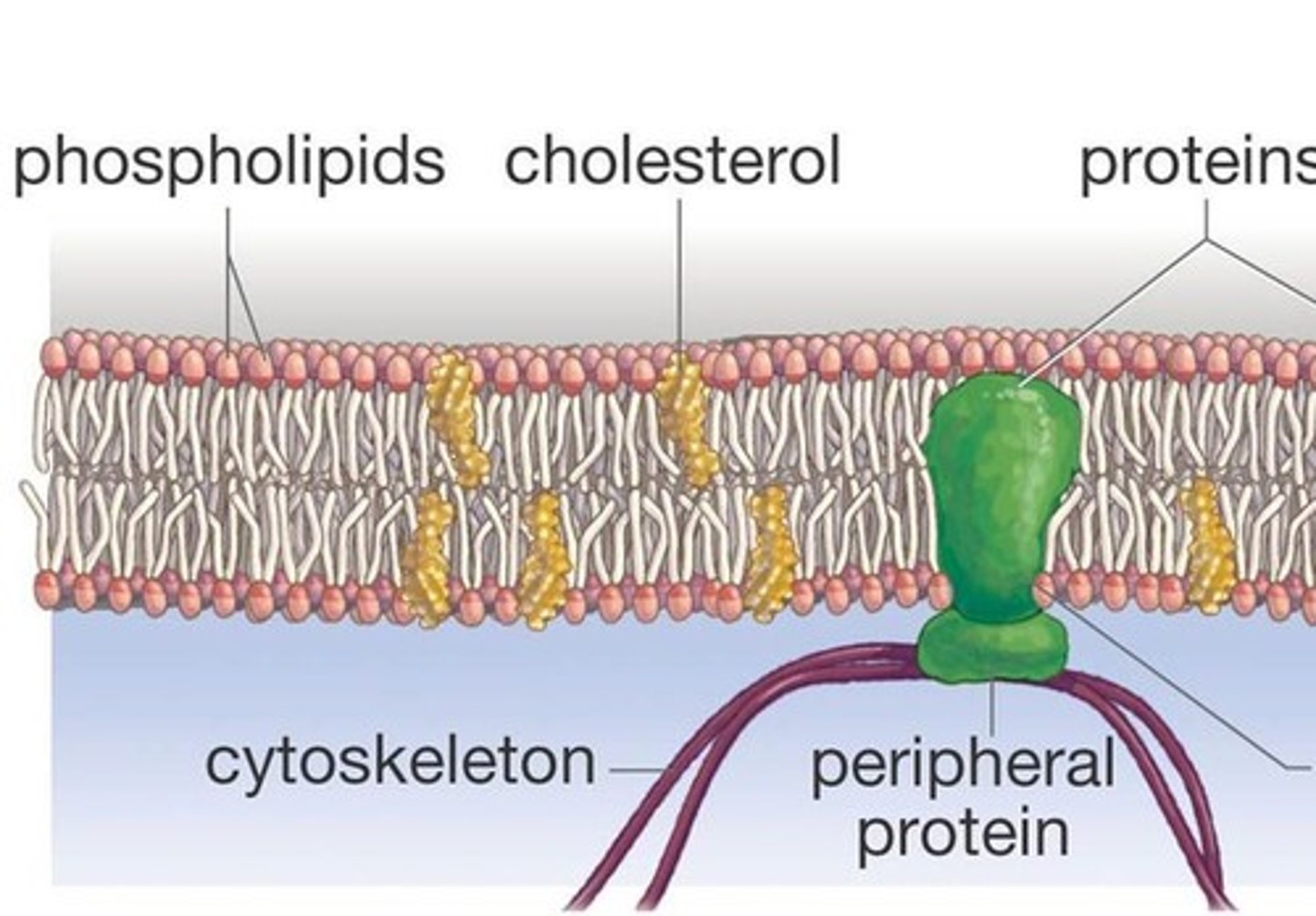
Cell Membrane Composition
phospholipids, cholesterol, proteins, and carbohydrates (glycolipids)
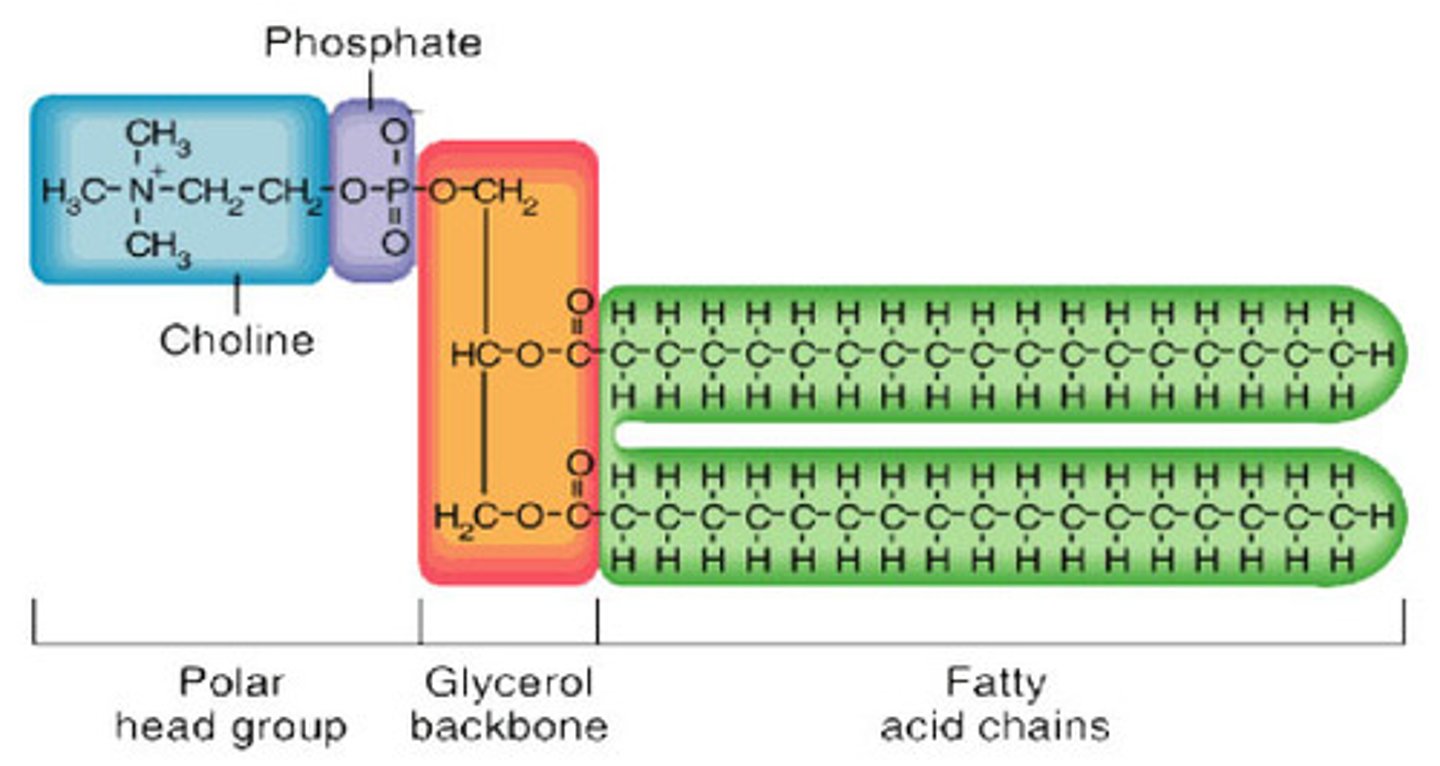
Phospholipid Structure
glycerol (or sphingosine), 1 or more fatty acid tails, alcohol and a phosphate group
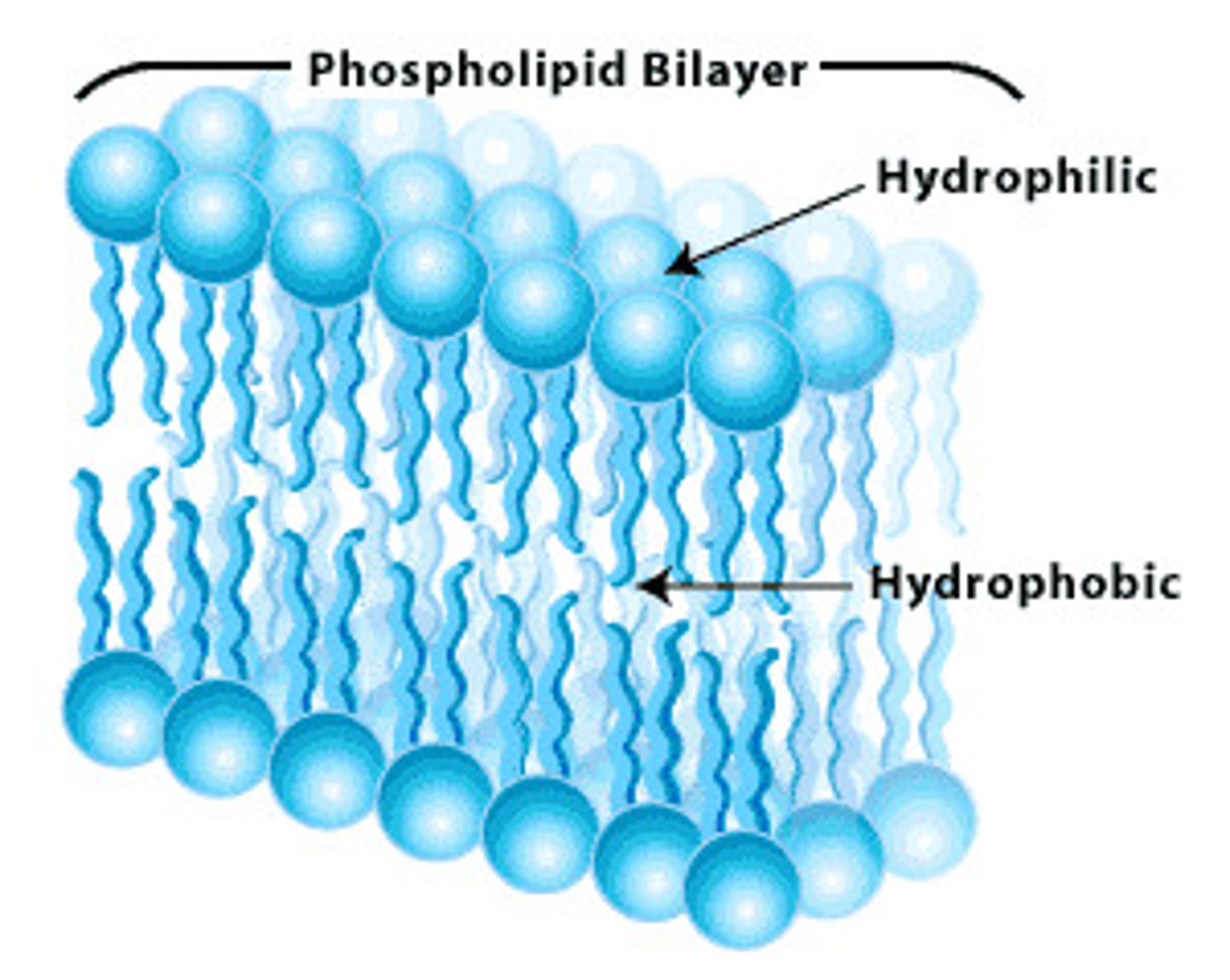
Phospholipid Bilayer Structure
composed of phospholipid molecules arranged with polar heads facing the outside and nonpolar tails facing the inside
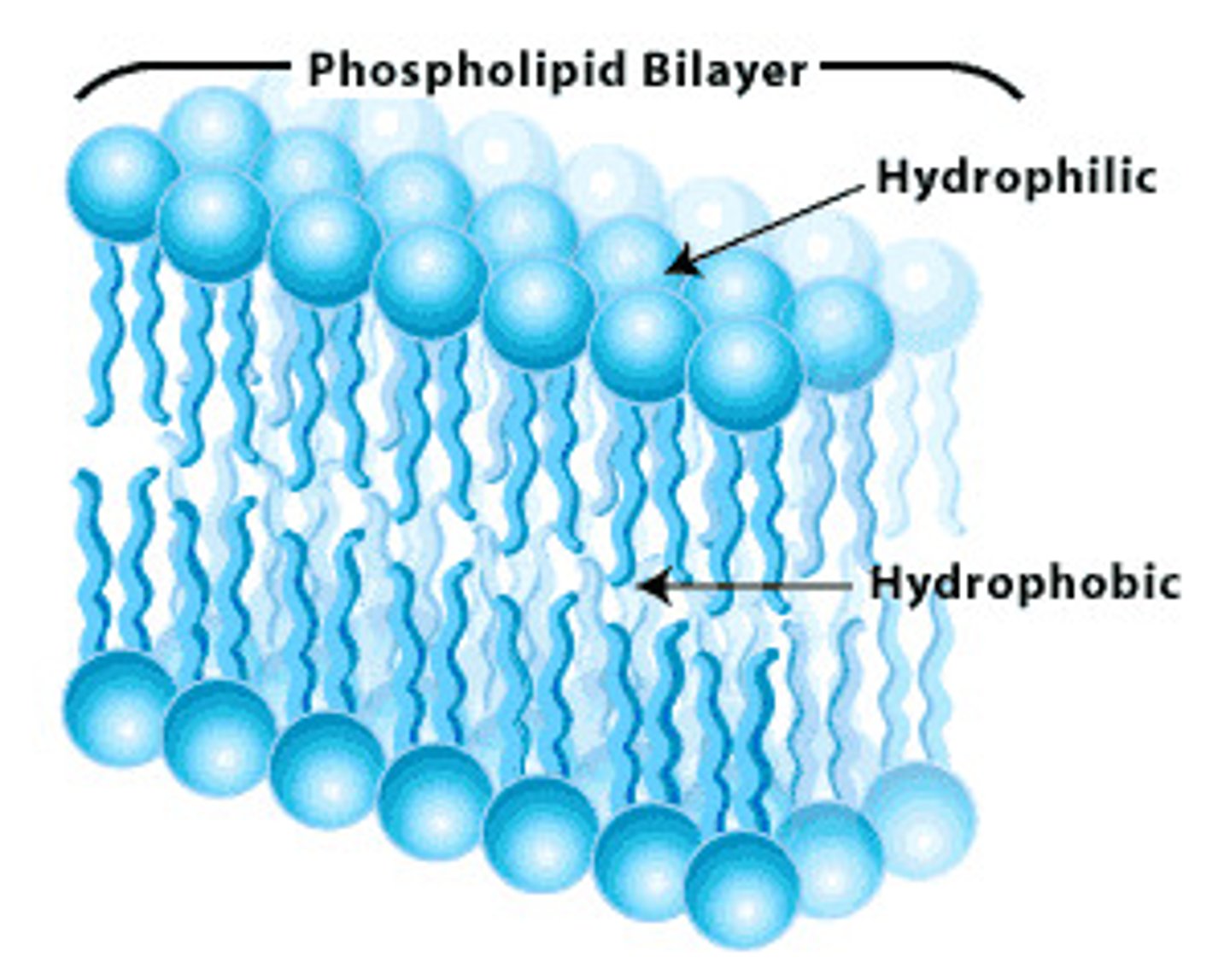
Amphipathic Structure of Phospholipids
contains both hydrophobic and hydrophilic regions
Hydrophobic Tails
-consits of saturated and unsaturated fatty acids
-repel water
-face the inside of the bilayer
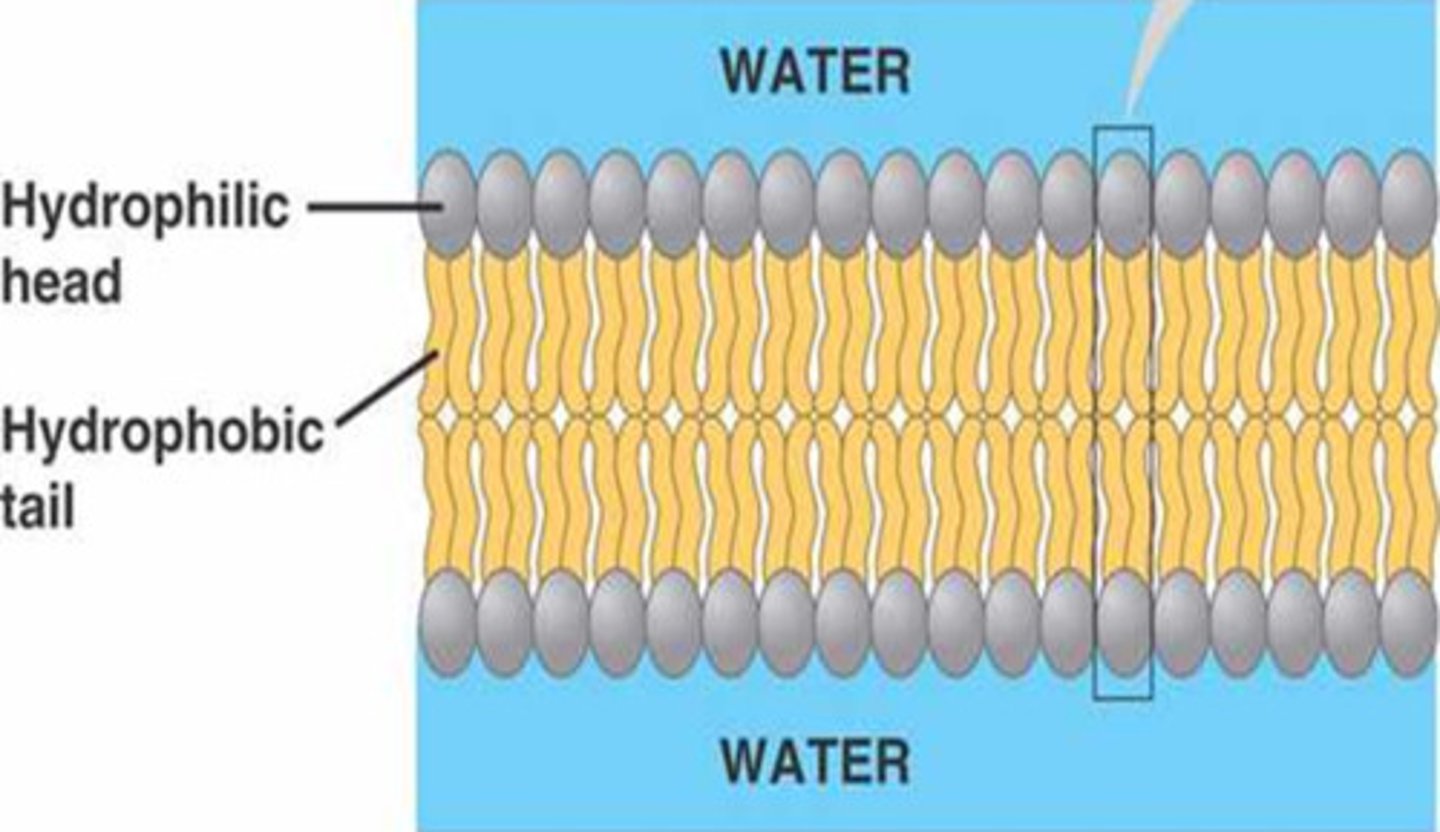
Hydrophillic Heads
-consists of a negatively charged (polar) phosphate group
-attracted to water
-face the outside of the bilayer
Lipid Bilayer Formation Mechanism
powered by the hydrophobic effect (increased entropy of H2O)
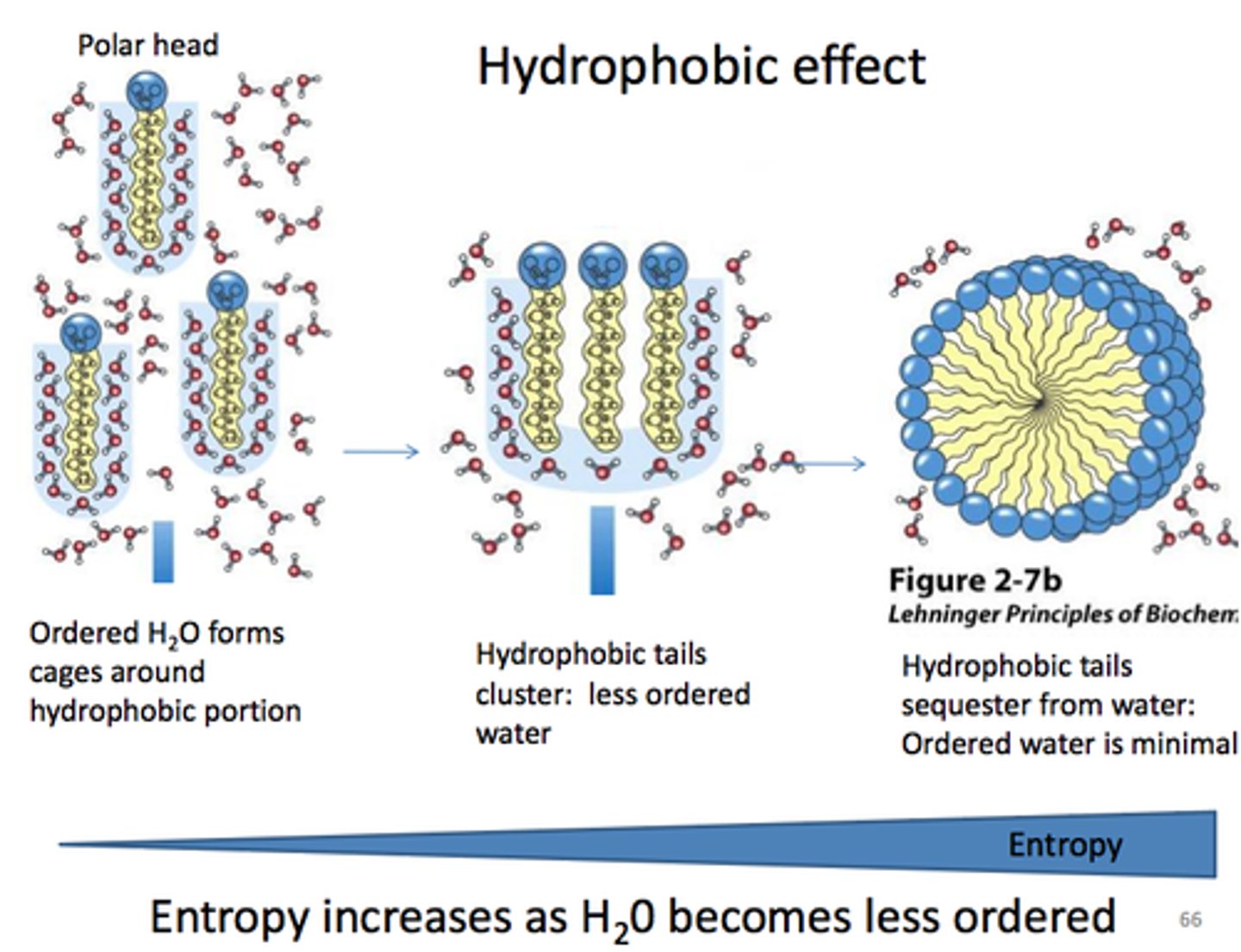
Lipid Bilayer Formation
occurs spontaneously in aqueous solution
Lipid Bilayer Charge
inside of the membrane is negatively charge
Types of Phospholipids
phosphoglycerides and sphingolipids
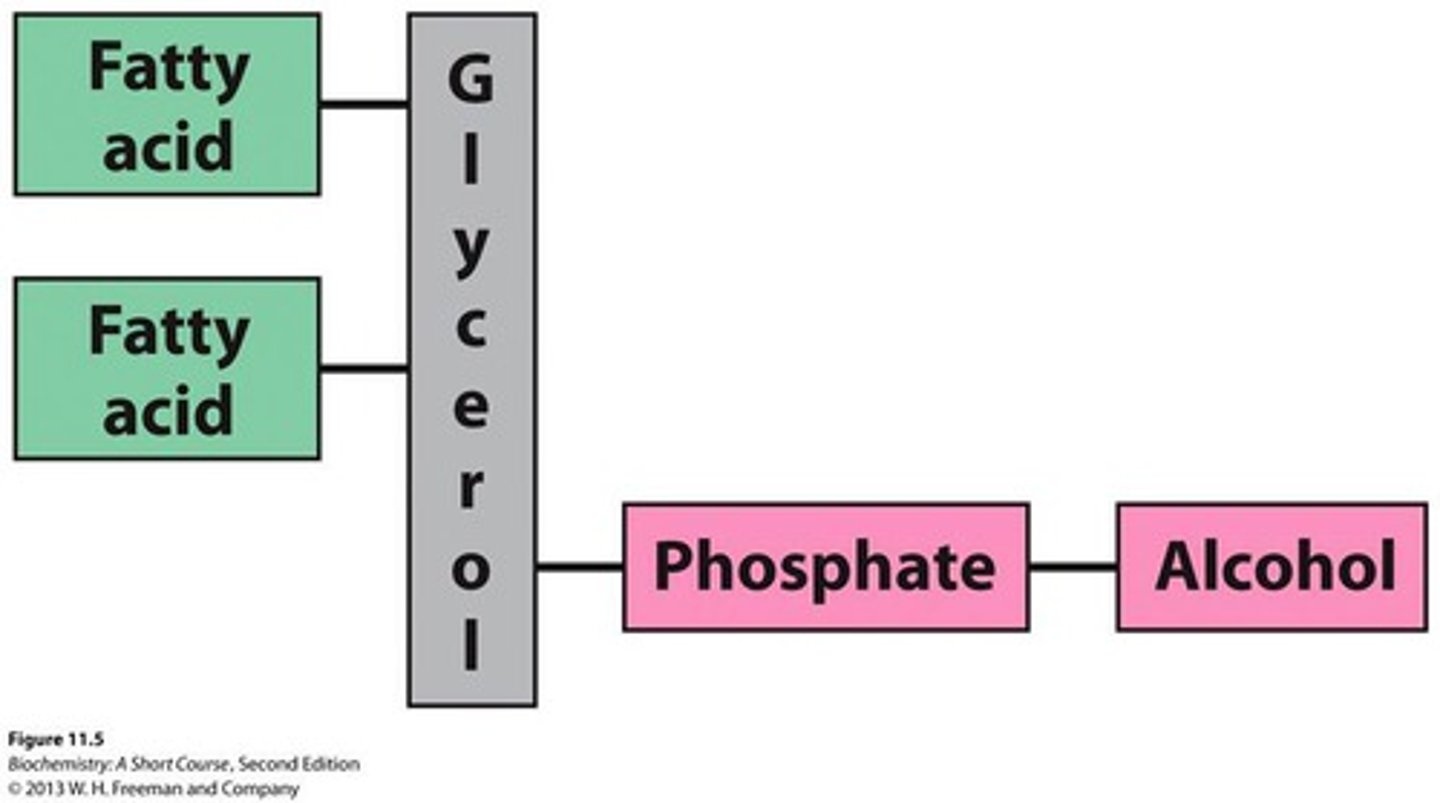
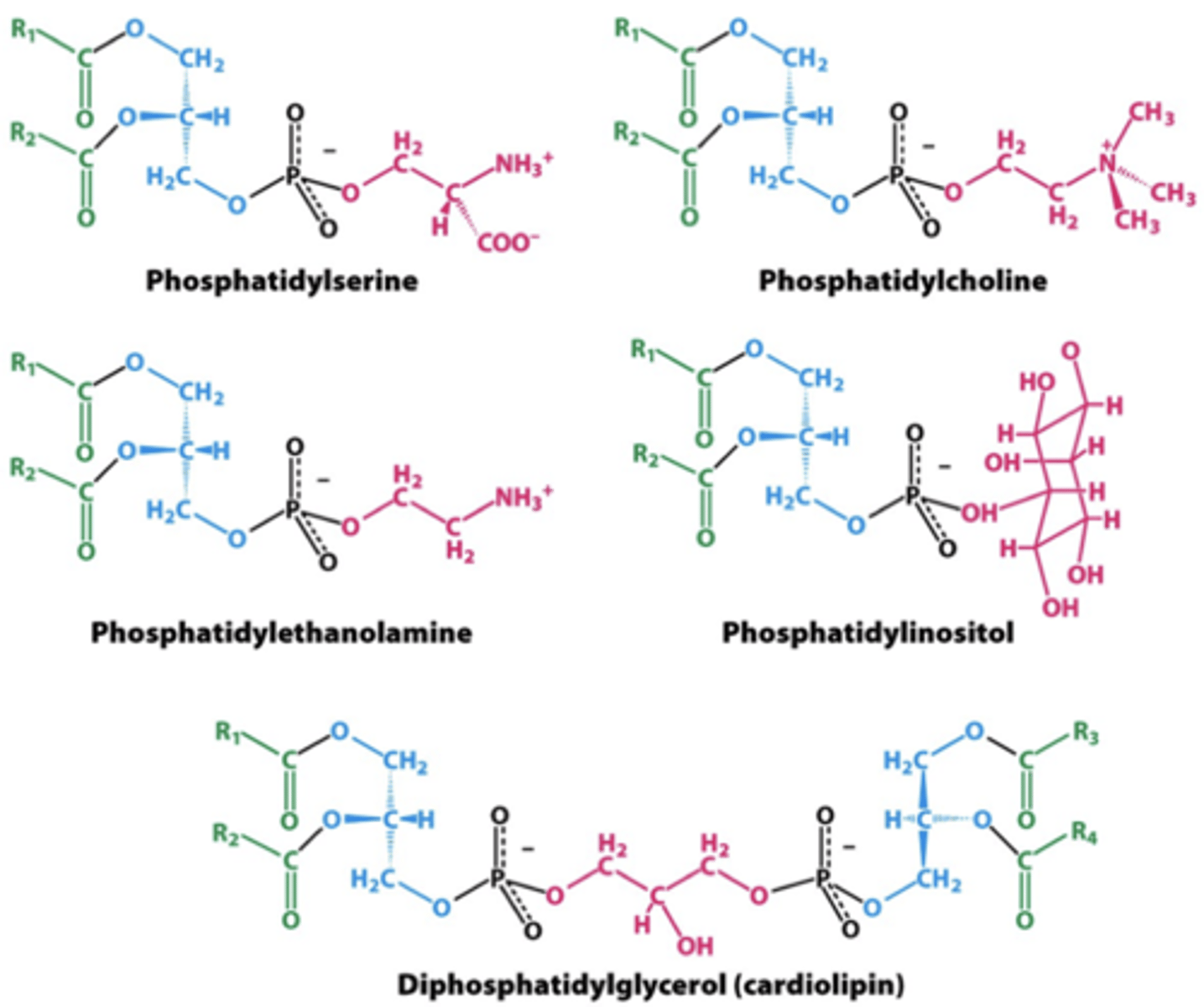
Phosphoglyceride
a type of phospholipid consisting of a glycerol molecule, two fatty acids, an alcohol and a phosphate group
Common Membrane Phosphoglycerides
phophatidylcholine (PC) = outer
phosphatidylethanolamine (PE) = inner
phophatidylinostiol 4-phosphate (PIP) = inner
Phosphatidylserine (PS) = inner
Inner Leaflet
The layer of the cell membrane phospholipid bilayer that faces TOWARD the cytoplasm.
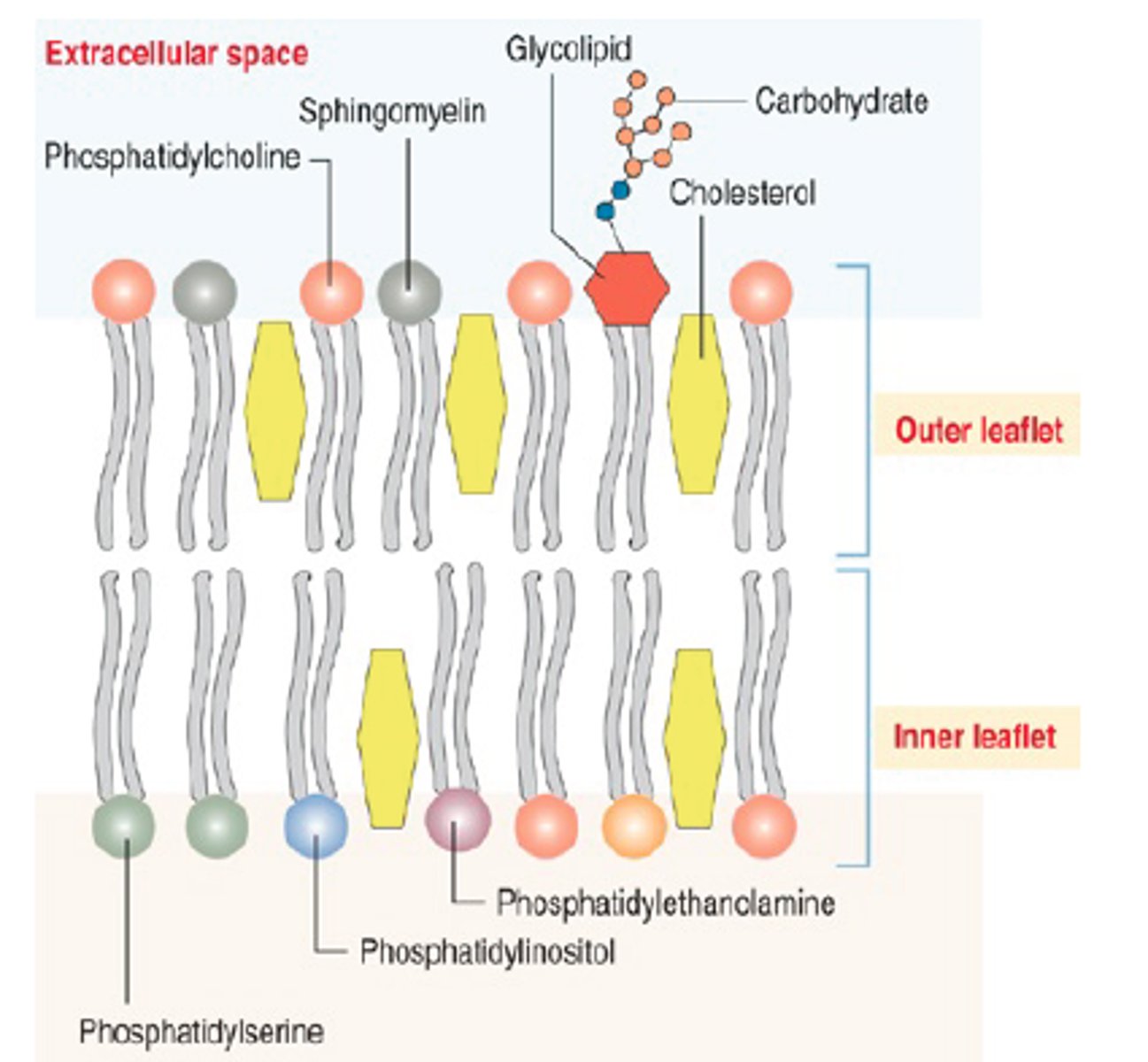
Outer Leaflet
The layer of the cell membrane phospholipid bilayer that faces AWAY from the cytoplasm.
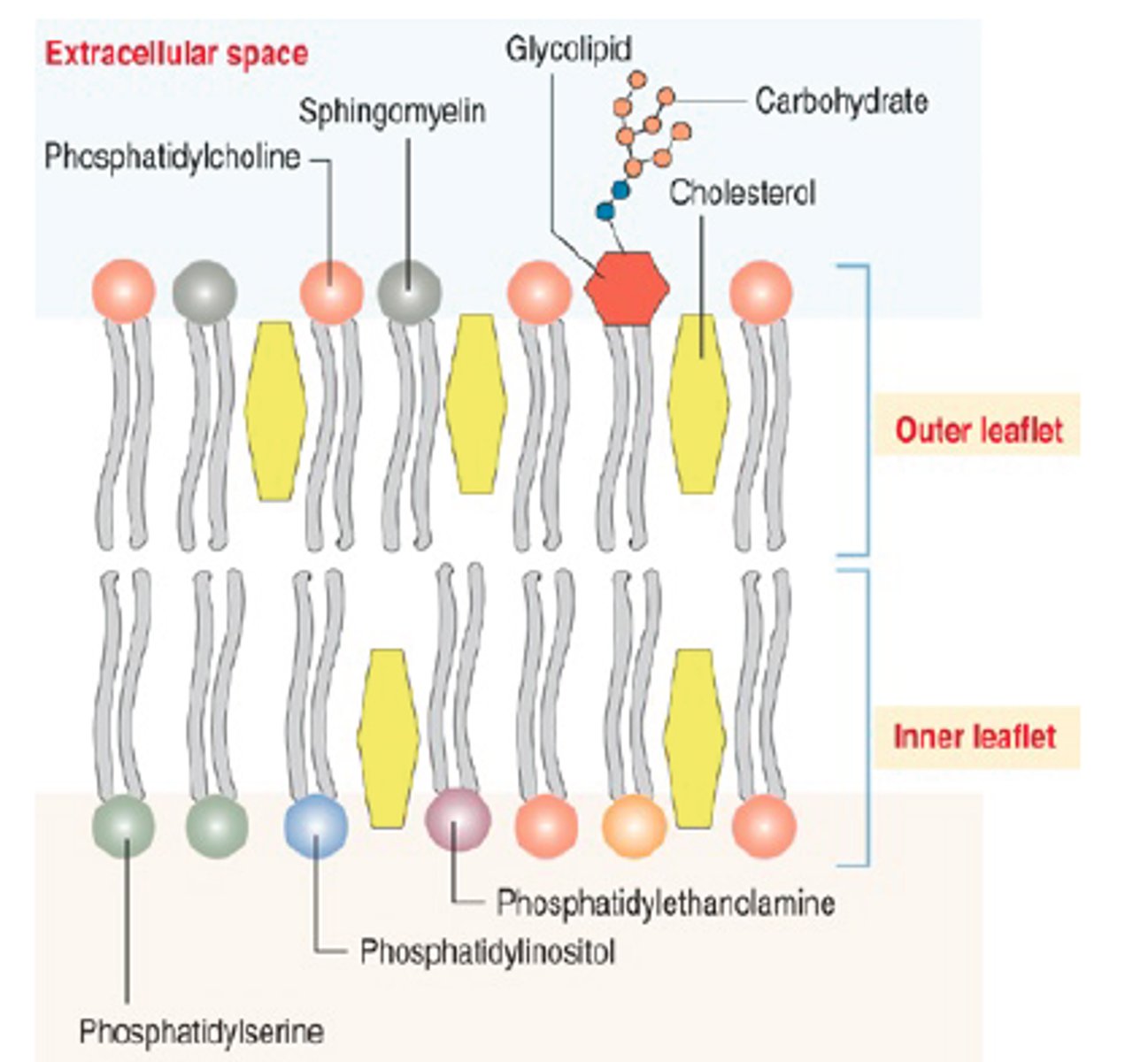
Sphingolipids
long fatty acid chain
polar head group
backbone= spingosine not glycerol
Glycolipids
-carbohydrates covalently bonded to lipids
-sugar residues on extracellular matrix
Membrane Fluidity
-extent to which lipid molecules can move in the plane of the membrane
-determine dby properties of fatty acid chains
-affected by melting temperature (rigid -> fluid) and degree of saturation (# double bonds)
-longer chain = decreased fluidity (rigid favored)
-double bonds = decreased fluidity (less packed)
-cholesterol = inhibits fluidity at low temp
Cholesterol
-type of lipd = steroid
-inserts into membrane with polar hydroxyl group close ot phosplipid head groups
-maintains membrane fludity at a variety of temperatures
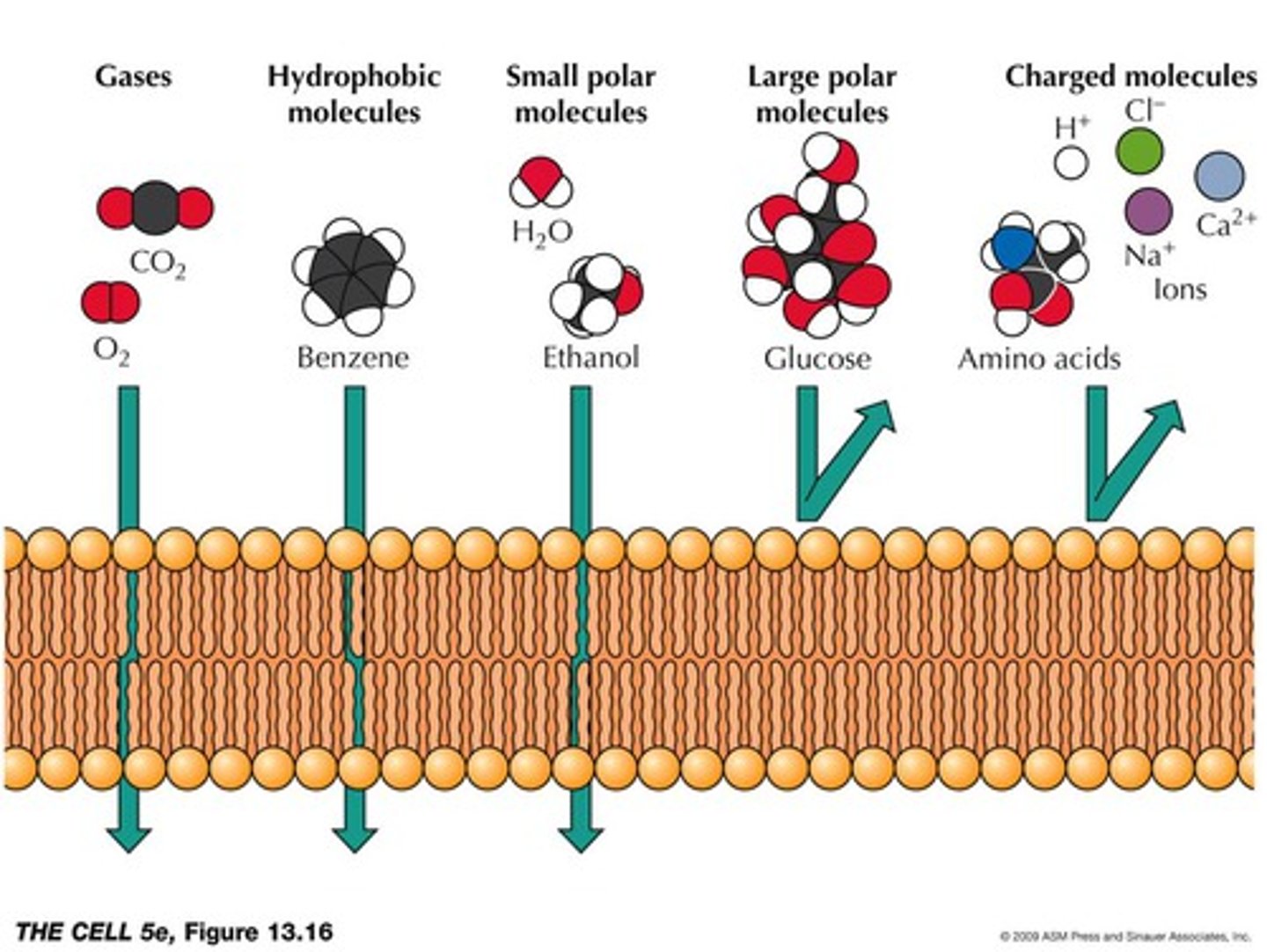
Membrane Permeability
-degree to which a membrane allows a substance to pass through it
-low for ions and most polar molecule s
-small molecules permeability based on solubility
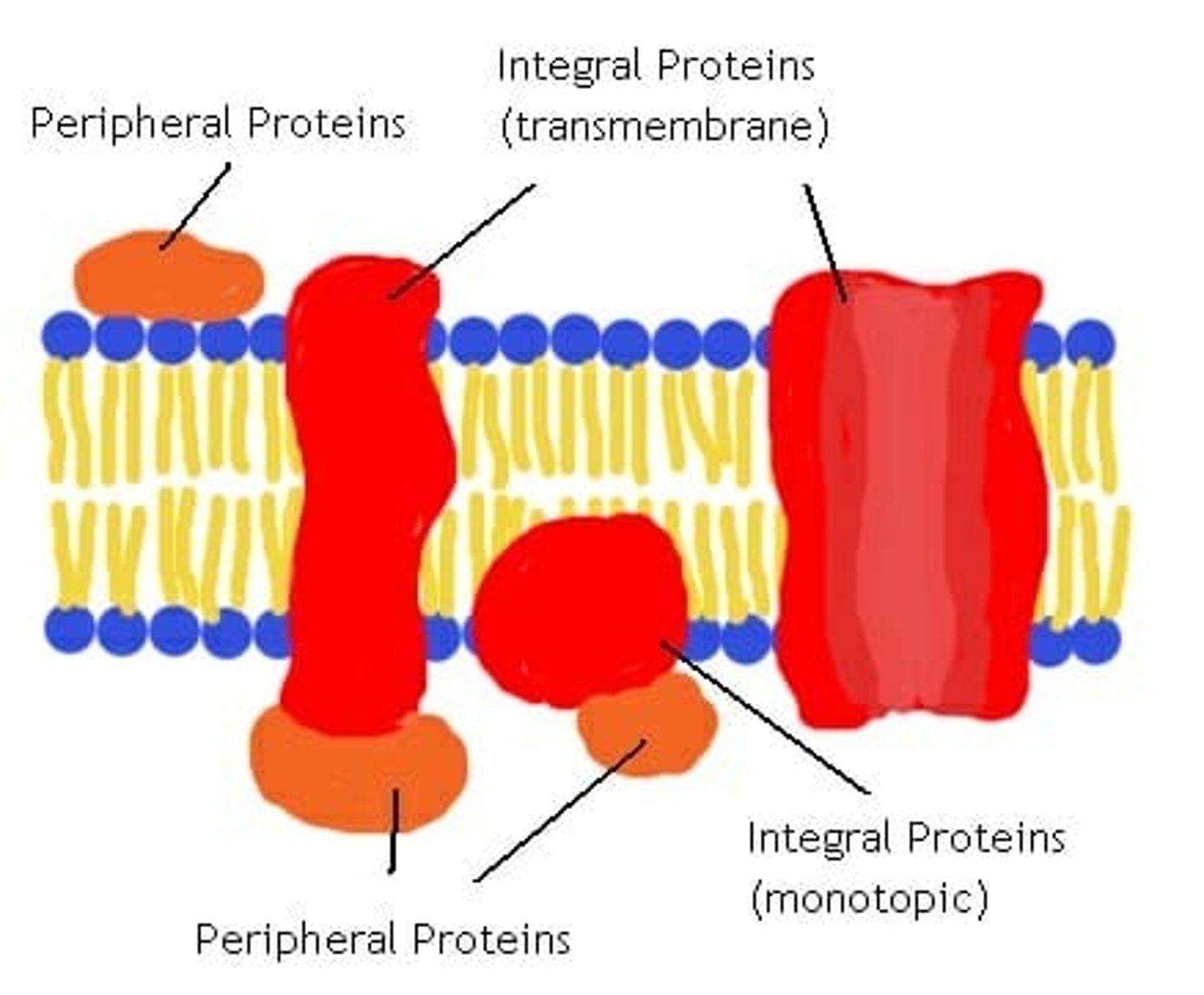
Membrane Proteins
-embedded proteins that perform specific functions for the cell membrane
-may be integral or periphreal depending on interaction with hydrophobic interior of membrane
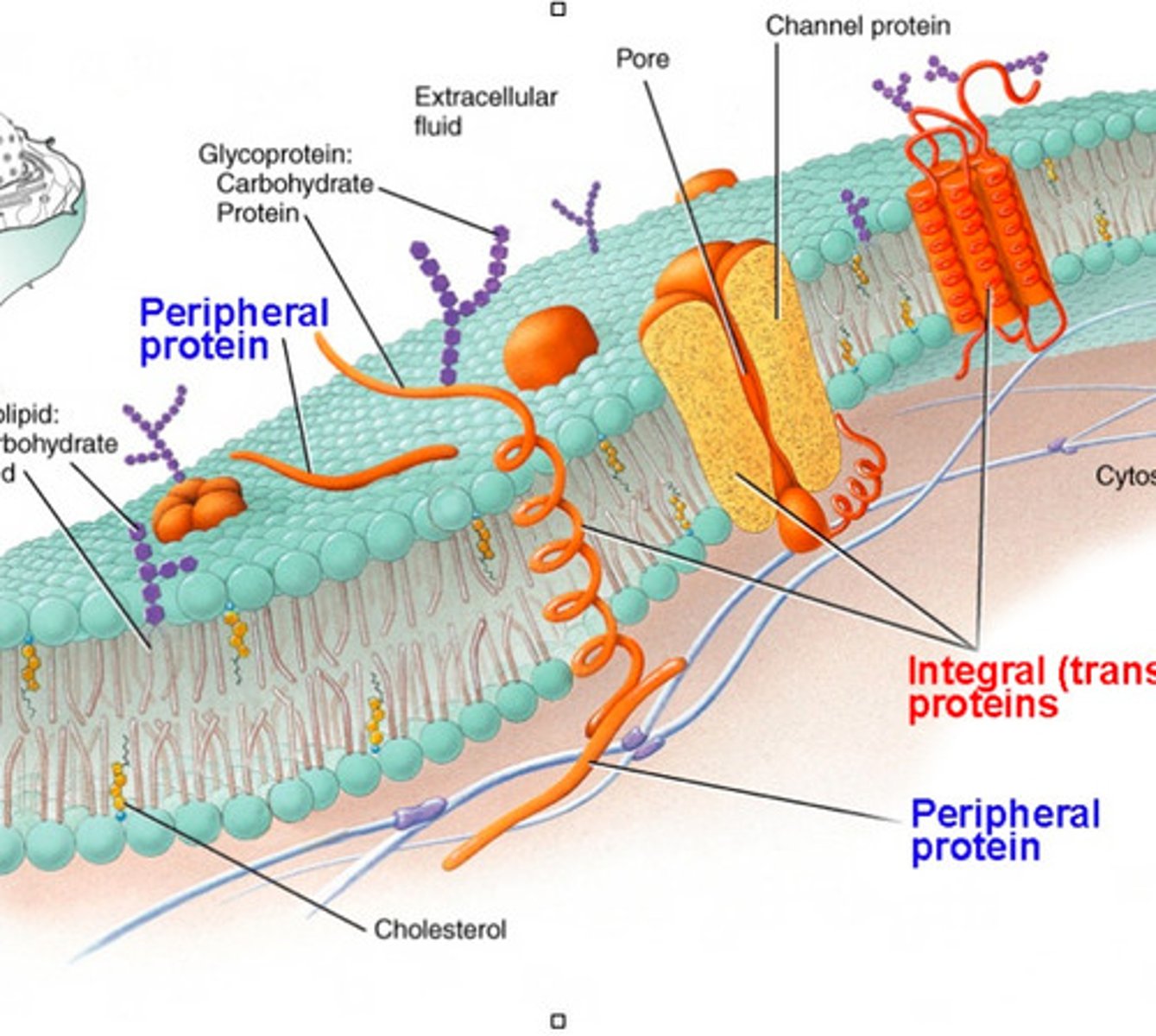
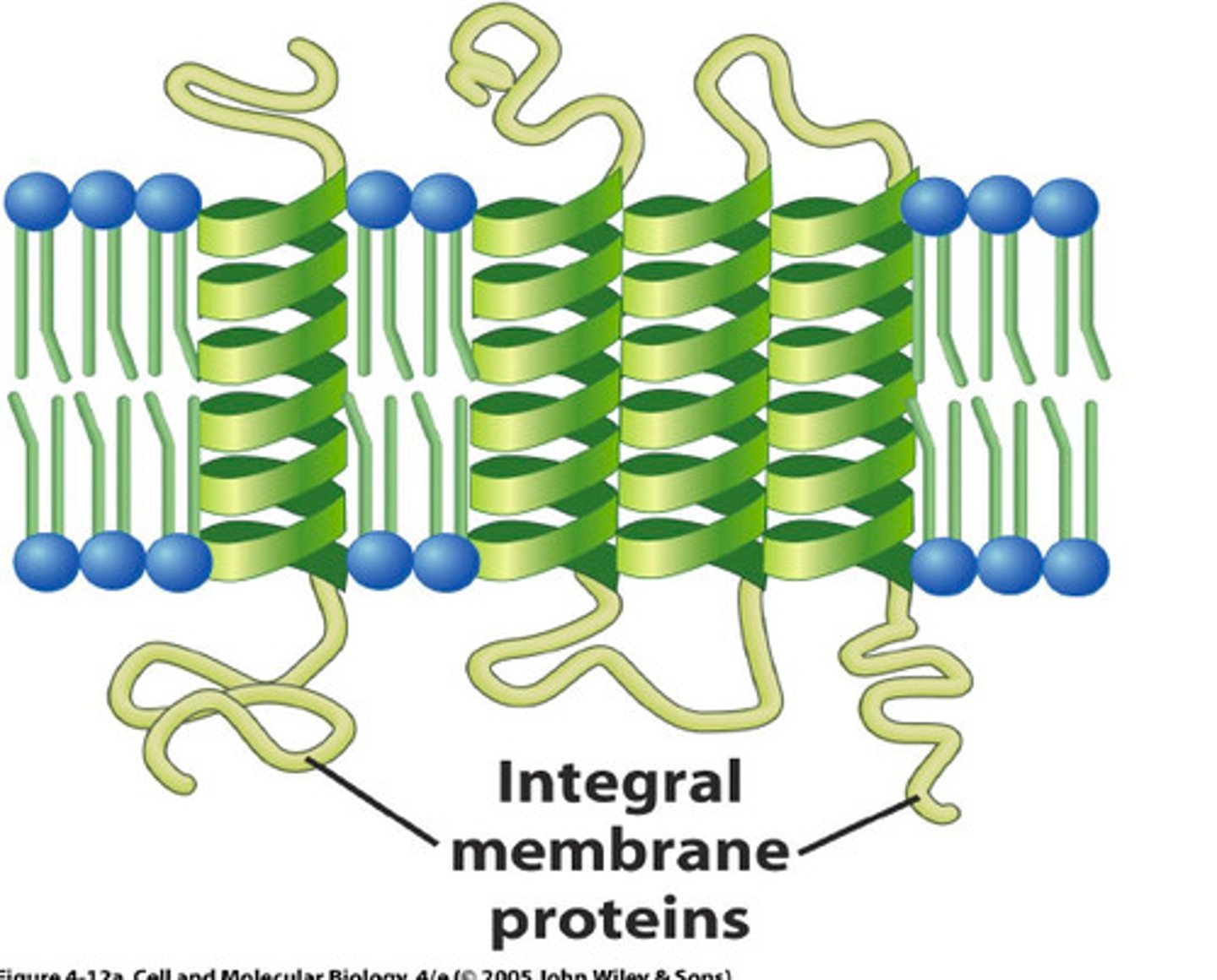
Integral Membrane Proteins
-embedded in hydrocarbons of membrane lipids
-transmembrane (span the bilayer)
-hydrophobic exterior and hydrophillic interior ("inside out" structure interacts with membrane)
Periphreal Proteins
-electrostatically and hydrogen bound to head groups of lipids or exposed portions of integral proteins
-may be on cytoplasmic or extracellular side of membrane
-hydrophilic exterior and hydrophobic core interact with fluid
Transport Proteins
-transmembrane/integral protein
-facilitates movement of specific molecules across membrane
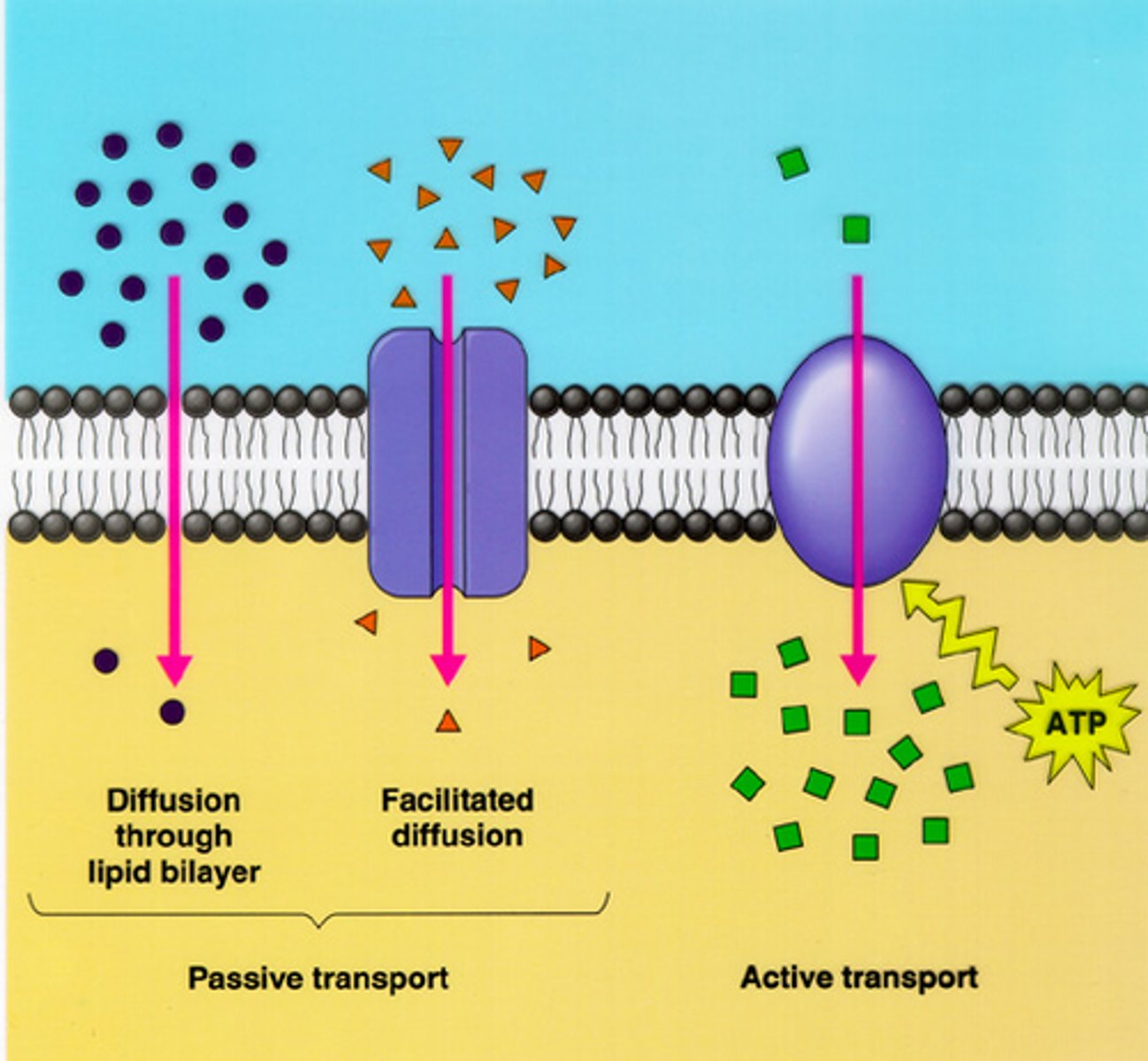
Simple Diffusion
-type of passive transport
-movement of a solute from an area of high concentration to an area of low concentration
-doesn't require energy input
-transports small, non-polar molecules
-doesn't use transport proteins
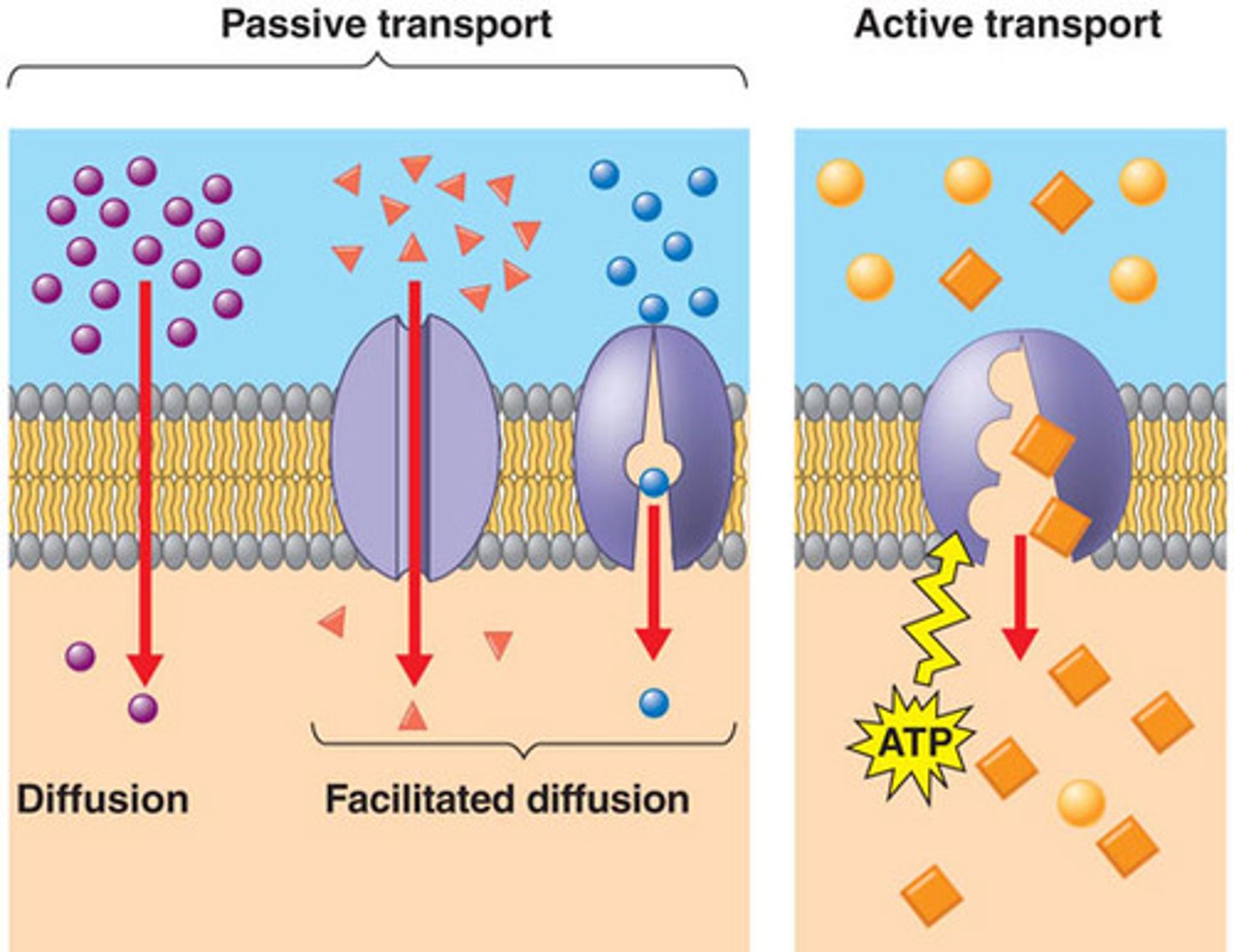
Facilitiated Diffusion
-type of passive transport
-driven by concentration gradient
-doesn't require an energy input
-uses tranport proteins (specific to certiain solutes)
-rate of diffusion depends on kinetics of tranport protein
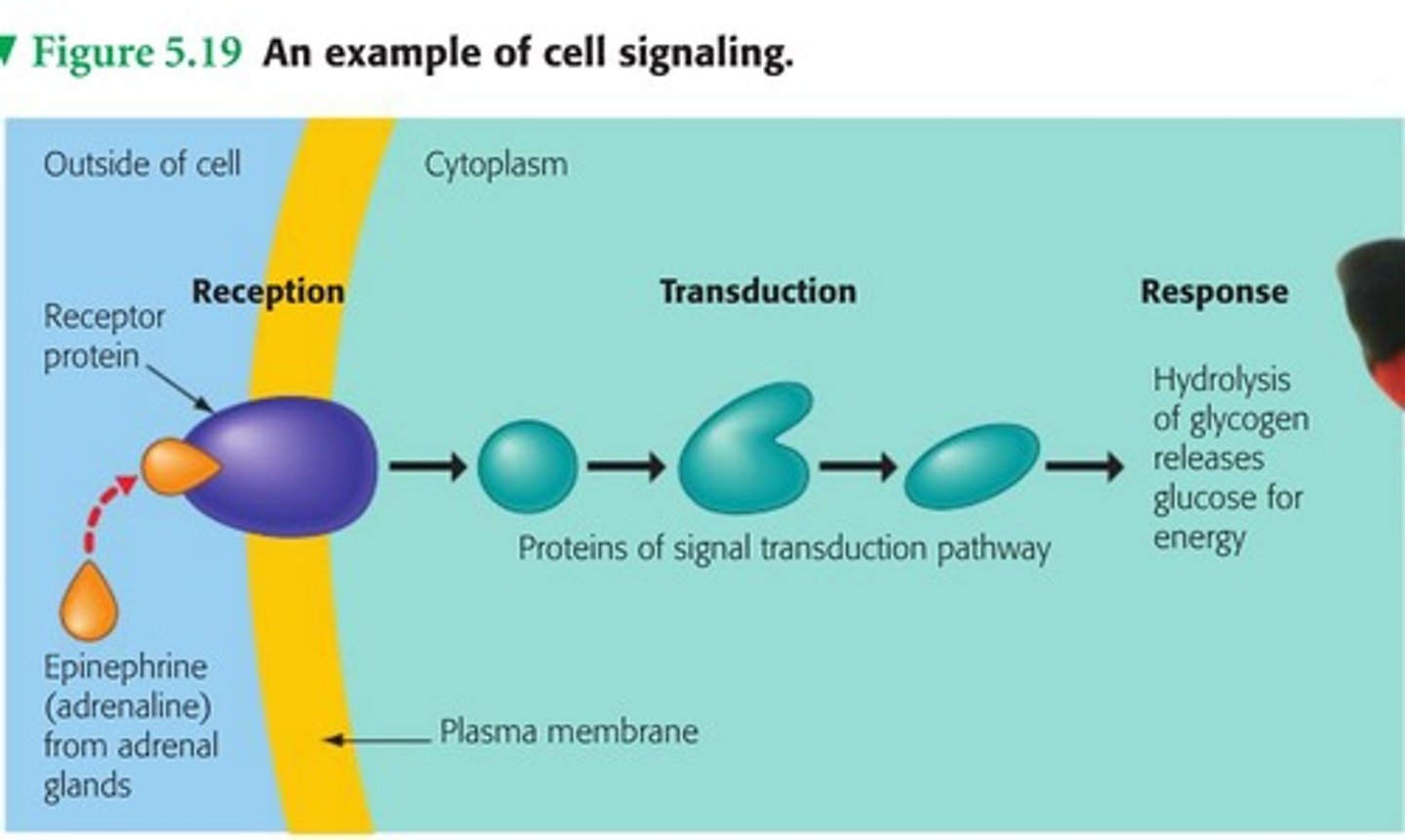
Signal Transduction Pathway
1. Reception
-primary messenger (ex: epinephrine) released
-binds to extracellular domain of receptor
-causes confomrational change
2. Transduction
-secondary messengers (ex: cAMP) released and amplify signal
3. Response
-effectors activate or inhibit cell processes to alter physiological response
Types of Membrane Receptor Proteins
Seven-Transmembrane-Helix (7TM)
Protein Kinase
Dimeric
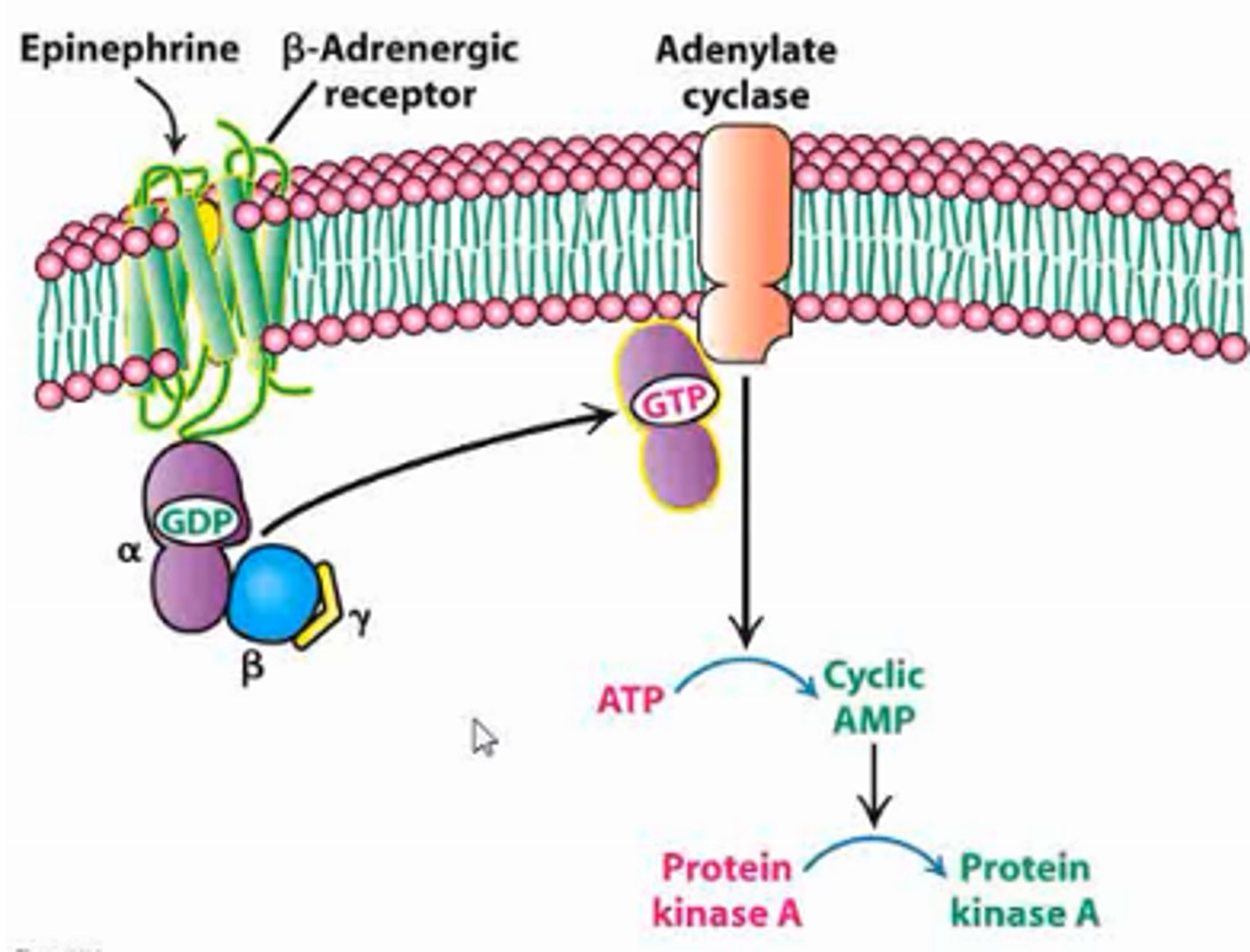
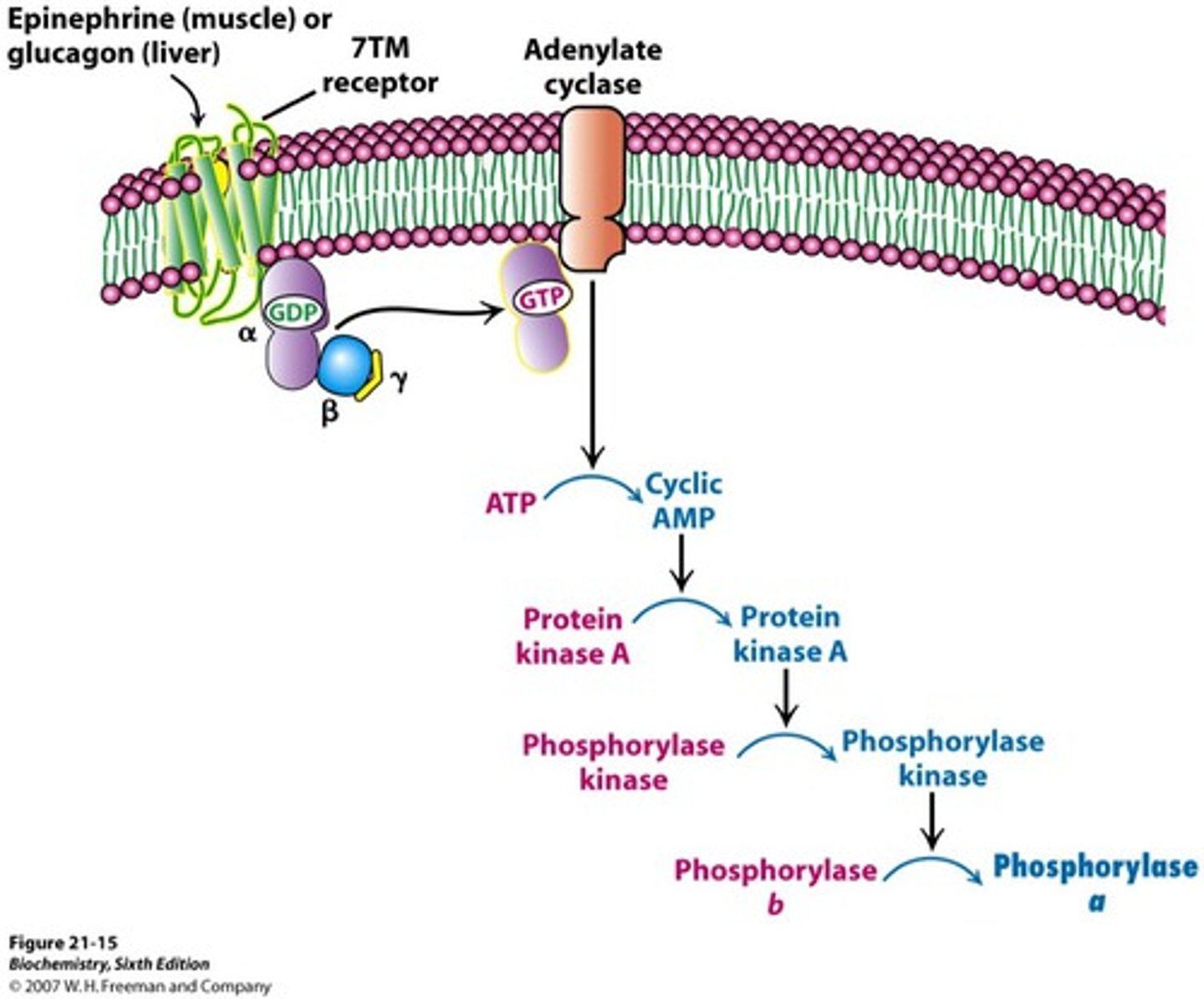
beta-adrenergic receptor
-type of 7TM receptor/G-protein coupled receptor
-binds epinephrine
-confromational change upon ligand bidning activated G-protein by phosphorylation (GDP -> GTP) AMPLIFIED
-activated G-protein stimulates adenylate cyclase AMPLIFIED
-adenylate cyclase releases cAMP (ATP -> cyclic AMP) AMPLIFIED
-cAMP activated protein kinase A
-activated protein kinase A phosphorylates target proteins
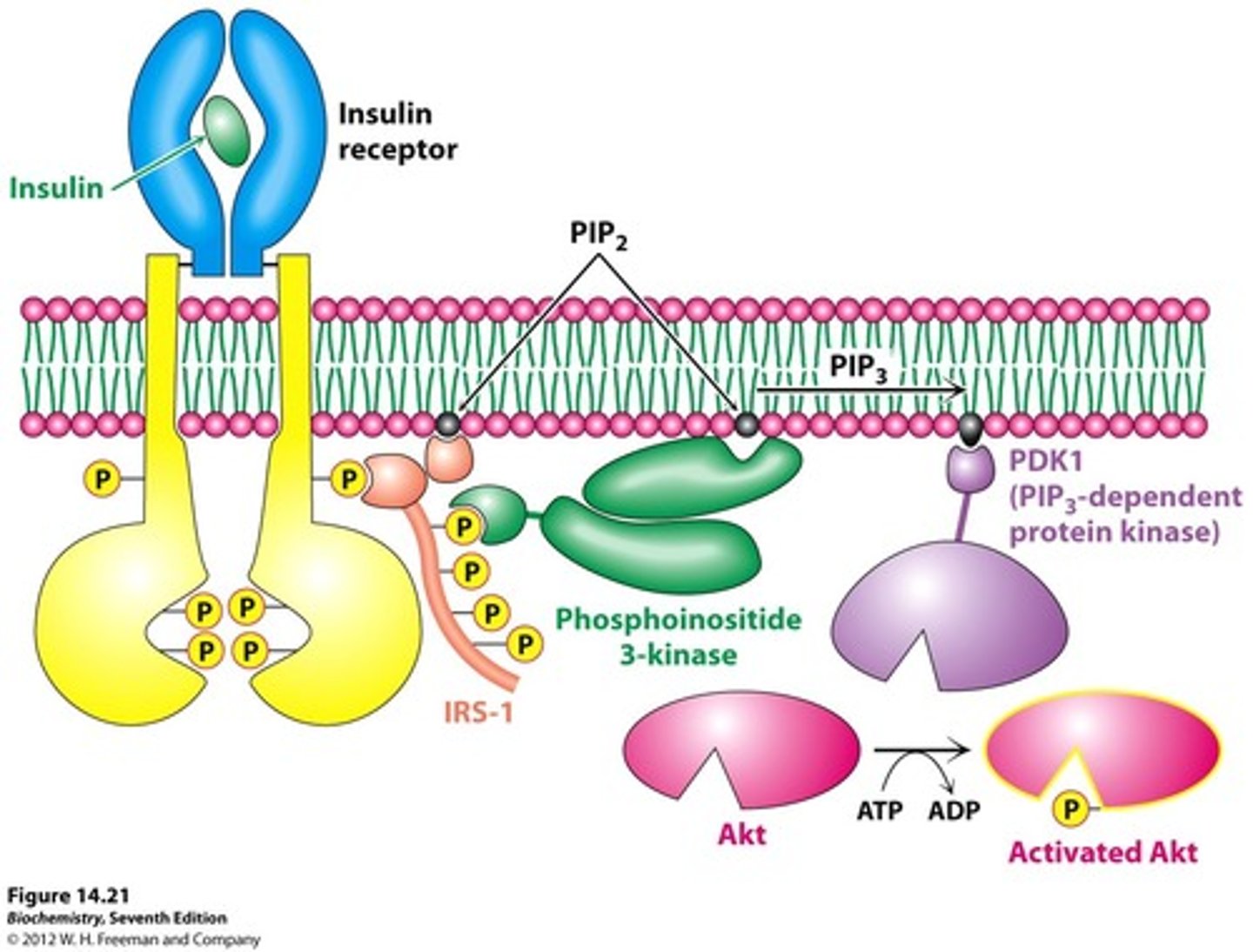
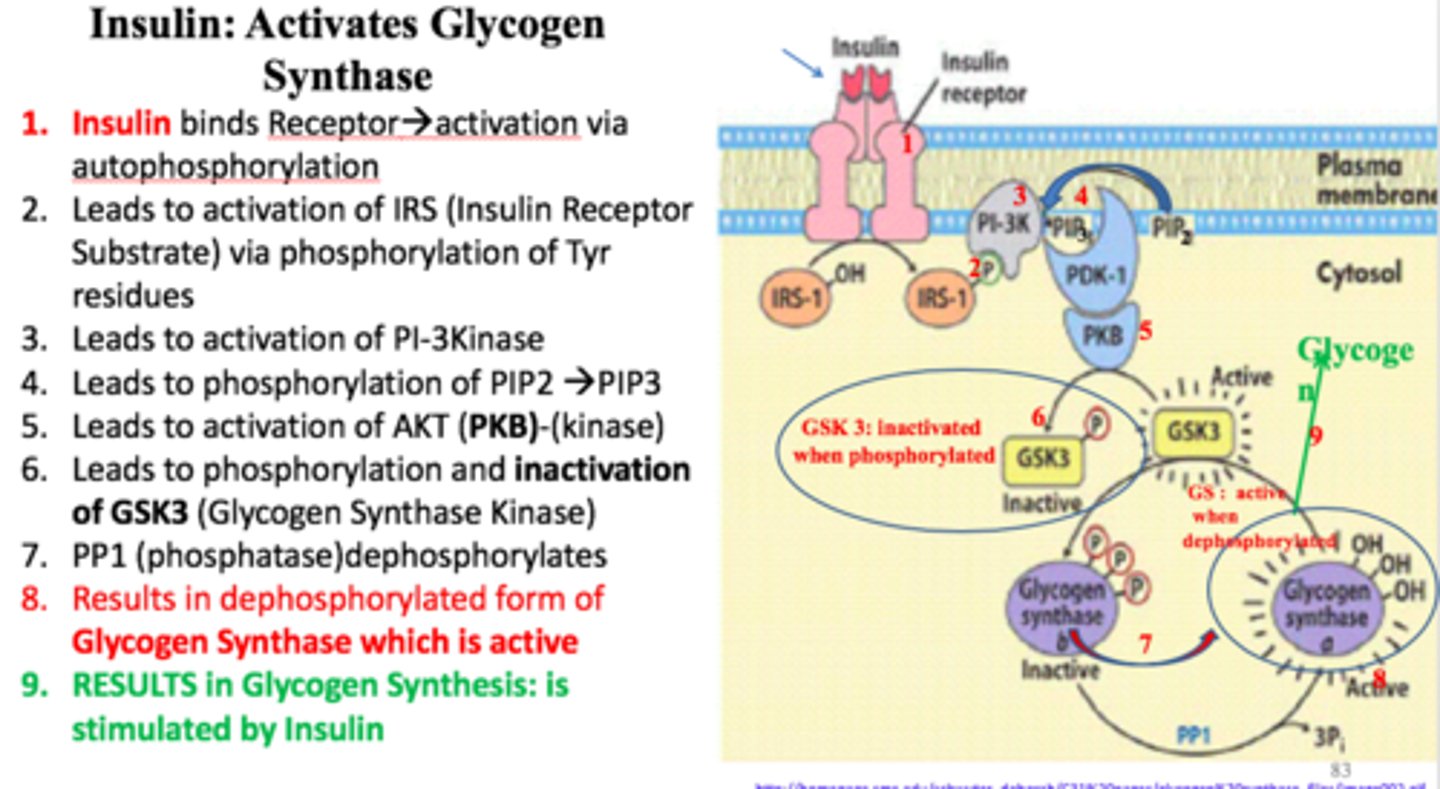
Insulin signaling
-tyrosine kinase membrane protein receptor
-receptor is a dimer
-insulin binding causes cross phosphorylation and activaiton of the receptor
-activated receptor phosphorylates insulin receptor substrates (IRS) that act as adaptor proteins AMPLIFIED
-IRS-1 phosphorylates PIP (PIP2 -> PIP3) AMPLIFIED
-PIP3 activates PDK-1
-PDK-1 activates Akt protein kinase by phosphorylation AMPLIFIED
-Activated Akt causes increased glucose transporters on cell surface
True or False: Elevated temperature increases membrane fludity.
True
True or False: Cholesterol increases membrane fluidity.
True
True or False: Saturated fatty acids increase membrane fluidity.
False
True or False: Phopsholipids readily flip from one side of the bilayer membrane to the other.
False
(Phospholipids can readily move laterally or rotationally but rarely flip flop from one layer to the another due to the fact that flip flopping requires higher energy to move hydrophilic head through hydrophobic core.)
True or False: Many membrane spanning proteins readily tumble in a lipid bilayer.
False
Like membrane lipids, membrane proteins do not tumble (flip-flop) across the lipid bilayer, but they do rotate about an axis perpendicular to the plane of the bilayer (rotational diffusion). In addition, many membrane proteins are able to move laterally within the membrane (lateral diffusion).
The rate limiting step for non-mediated passage (simple diffusion) of a molecule through a membrane bilayer is its solubility in the membranes's core. A molecule which is very soluble in the core of a bilayer will exhibit greater permeability than a molecule which is less soluble in the lipid core. Using your textbook as a guide (Tymozcko4e Fig 12.4; Berg9e Fig. 12.15), rank the following in order of increasing permeability (1 = least permeable; 5 = most permeable).
CO2
Ethanol
Glucose
Glucose 6-phosphate
H+
1. H+ (ion)
2. Glucose 6-phosphate (large, polar)
3. Glucose (large, polar)
4. Ethanol (small, uncharged)
5. CO2 (gas)
Cells have evolved elaborate signaling cascades to deliver messages from hormones that cannot pass through the cell membrane. In the process, the signal is amplified to coordinately regulate multiple cellular processes. Your book shows how insulin signaling is amplified at enzymatic steps in its cascade (Tymoczko4e Figure 13.20; Berg9e Figure 14.26). Identify the steps at which amplification occurs in the cyclic AMP signaling pathway (Tymoczko4e Ch. 13.2; Berg9e Ch. 14.1)
1. Epinephrine + β-adrenergic receptor -> activated hormone-receptor
2. GDP-bound G protein + GTP -> GTP-bound G protein + GDP
3. GTP-bound G protein + adenylyl cyclase -> activated adenylyl cyclase
4. ATP -> cAMP + Pi
5. cAMP + Protein Kinase A -> activated Protein Kinase A
6. effector protein + ATP -> phosphorylated effector protein + ADP
1. no
2. yes
3. no
4. yes
5. no
6. no
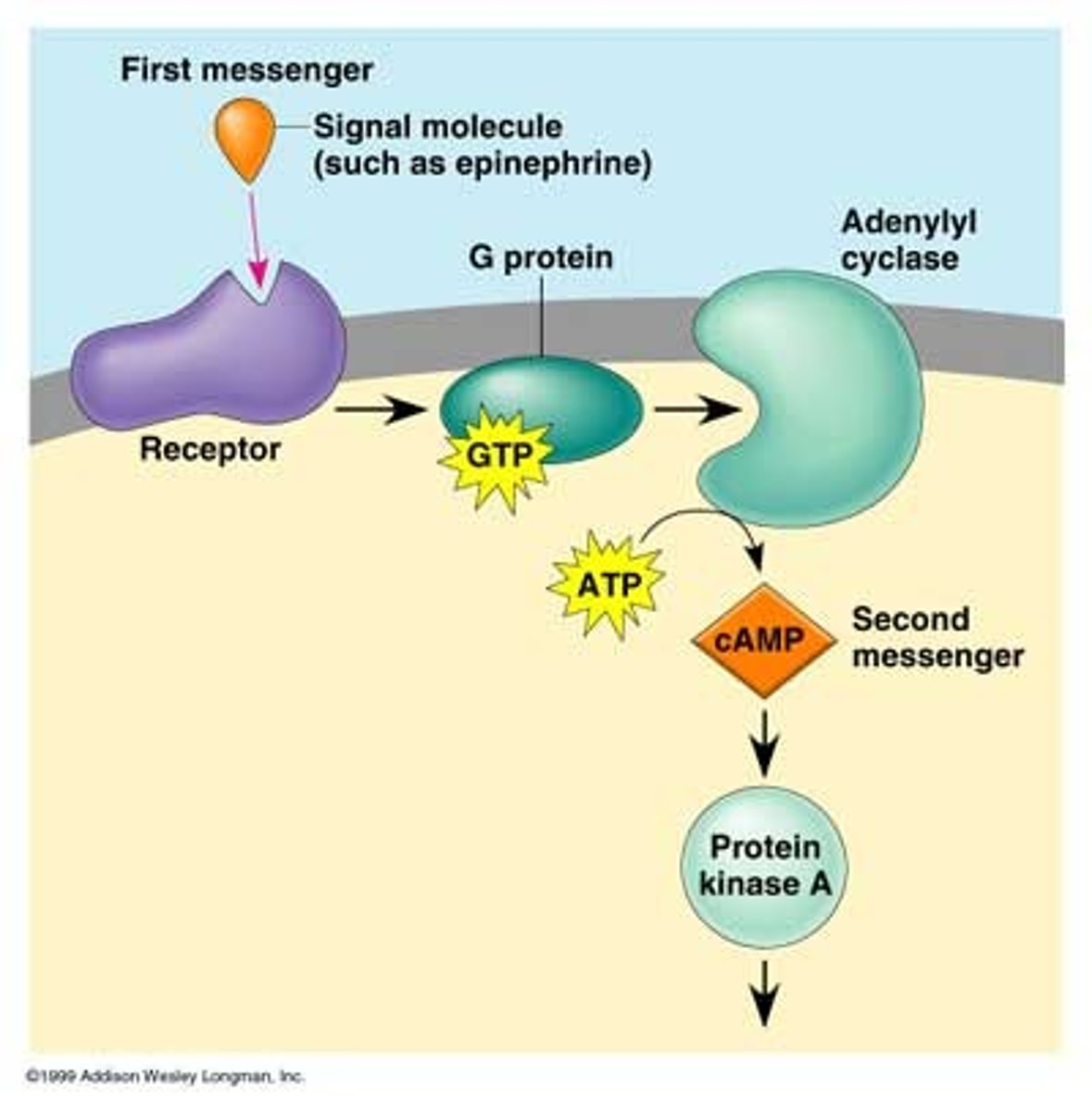
Identify activating and terminating enzyme activity or binding event in the cyclic AMP signaling cascade.
1. Adenylyl cyclase
2. cyclic AMP phosphodiesterase
3. G protein GDP/GTP exchange
4. G protein GTP hydrolysis
5. Hormone-receptor binding
6. Hormone-receptor dissociation
7. Phosphoprotein phosphatase
8. Protein kinase
1. activating (ATP -> cAMP)
2. terminating (cAMP -> AMP)
3. activating (GDP -> GTP)
4. terminating (GTP -> GDP)
5. activating (initiates cAMP cascade)
6. terminating (diminshes cAMP cascade)
7. terminating (inhibits cAMP cascade)
8. activating (cAMP binds regulatory unity and causes dissociation and activation of PKA)
Which amino acids are phosphorylated in insulin signaling?
serine or tyrosine
What functional group is modified in insulin signaling?
hydroxyl
What is the target protein of the insulin receptor?
IRS
What is the target protein of PDK1?
Akt
What is the target protein of Akt?
downstream proteins
Which enzymes turn off insulin signaling?
phosphoprotein phosphates (remove phosphoryl groups from proteins to restore hydroxyl group)
Glycogen
-Storage form of glucose
-found in liver and muscle
-glucose units linked by alpha 1,4 bonds
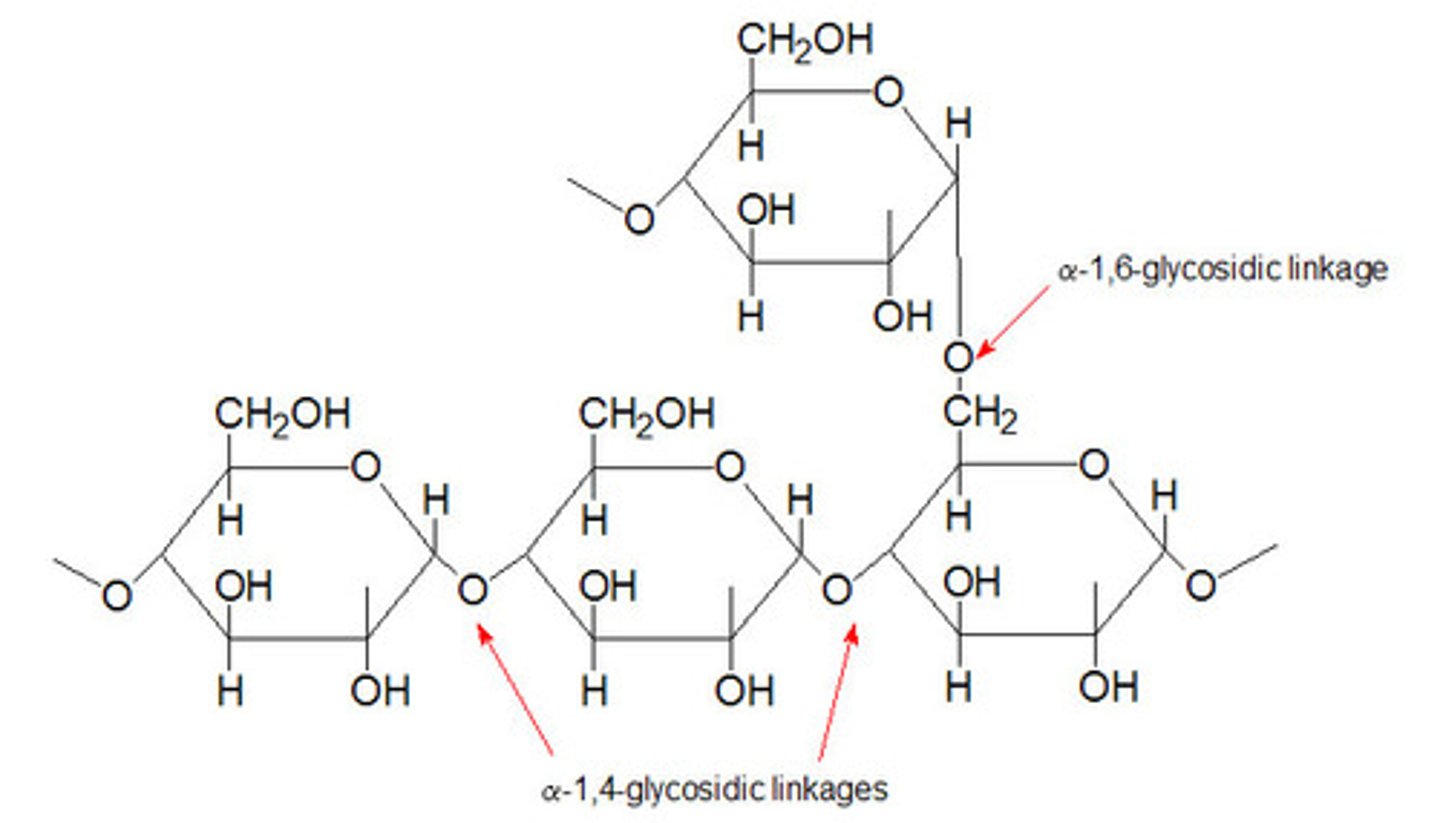
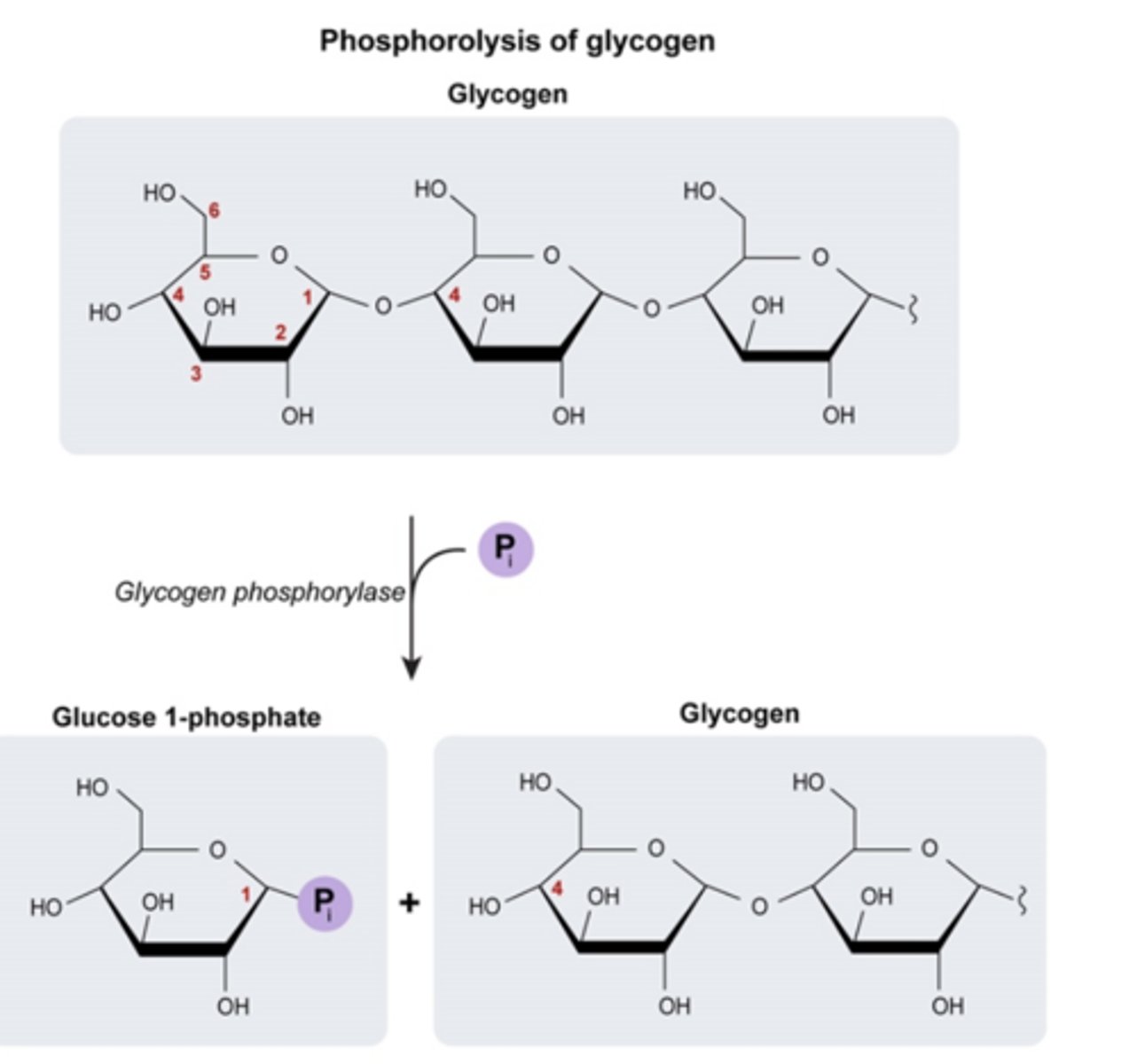
Glycogen Phosphorylase
-key regulatory enzyeme of glycogen breakdown
-enzyme that cleaves glucose from the non-reducing end (free -OH) of a glycogen branch by phosphorylating it
-glycogen is more branched (branches occur more frequently) --> more points where enzymes can come in & break down glycogen
-permits rapid release of glucose from glycogen stores
-phosphorolysis of terminal alpha-1,4-linkage yield glucose 1-phosphate and a glycogen molecule that is one glucose unit shorter
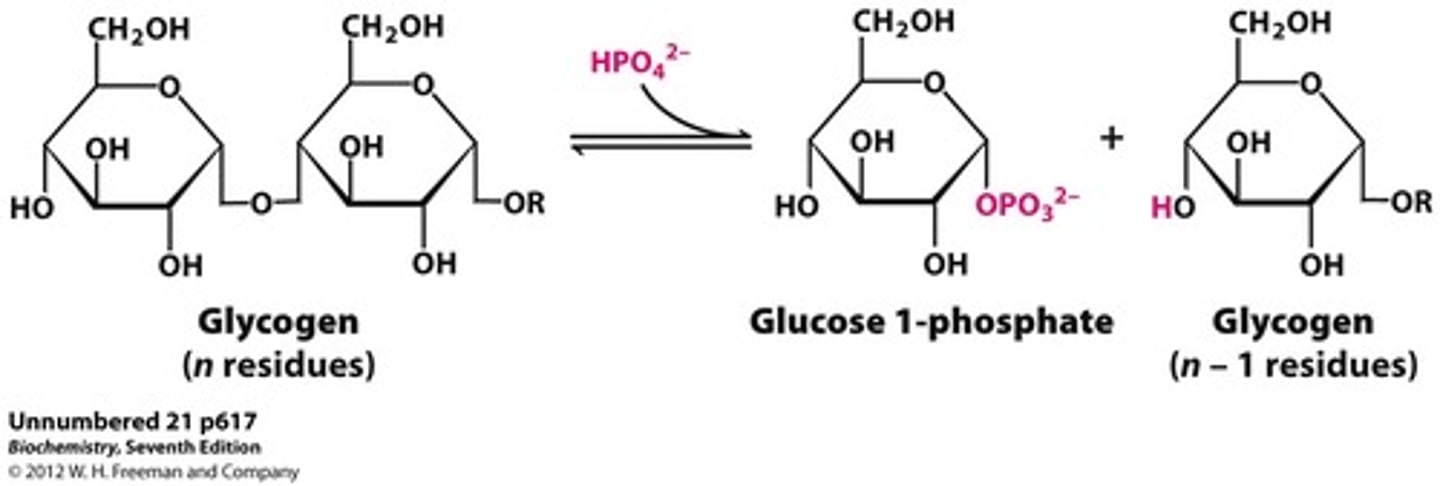
Glycogen Phosphorylysis
addition of a phopshate to glycogen results in release of glucose 1-phosphate and a glycogen molecule that is one glucose unit shorter
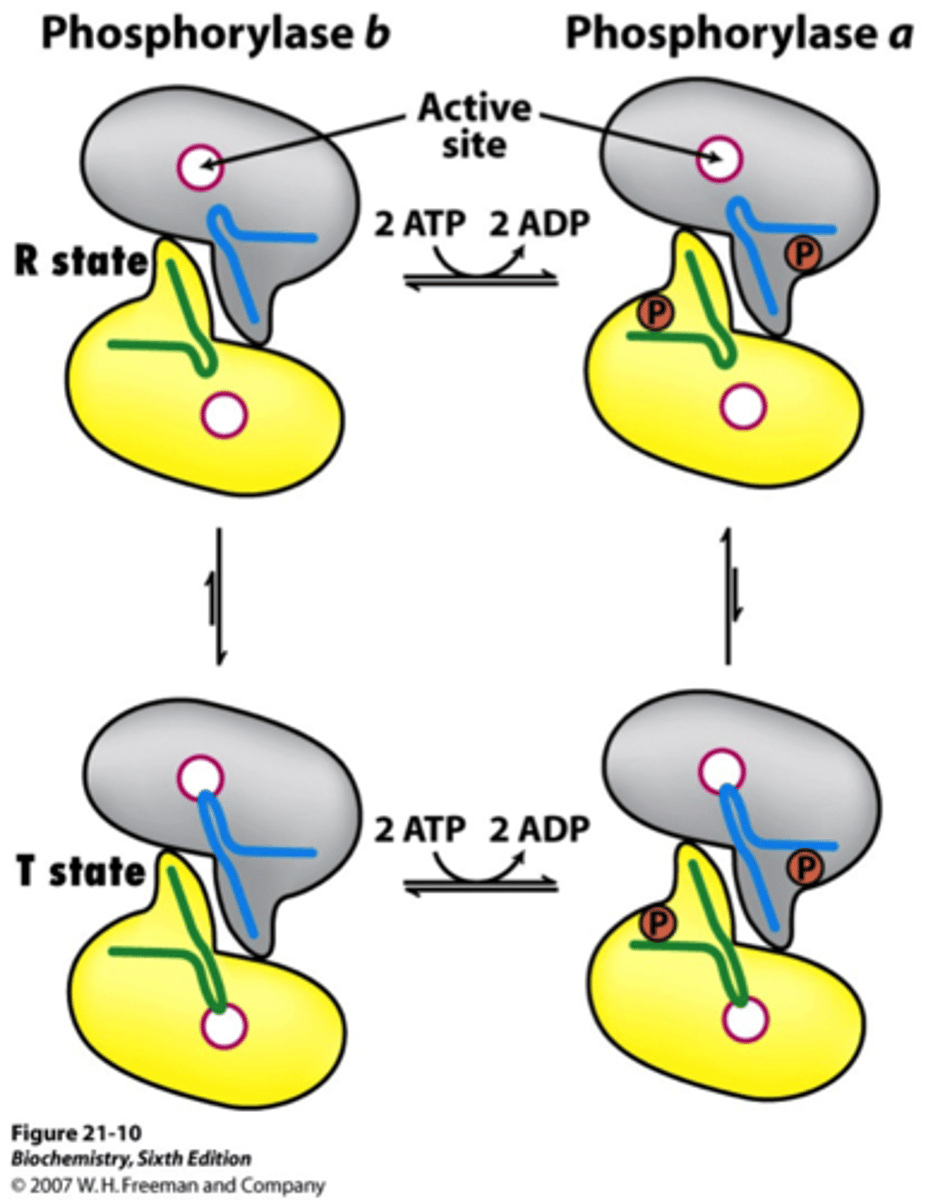
Forms of Glycogen Phosphorylase
active (a) = phosphorylated = R state
inactive (b) = unphosphorylated = T state
Allosteric Regulation of Glycogen Phosphorylase
-related to energy charge
-inhibited by ATP (high energy charge = high ATP = sufficient glucose = glycogen not degraded)
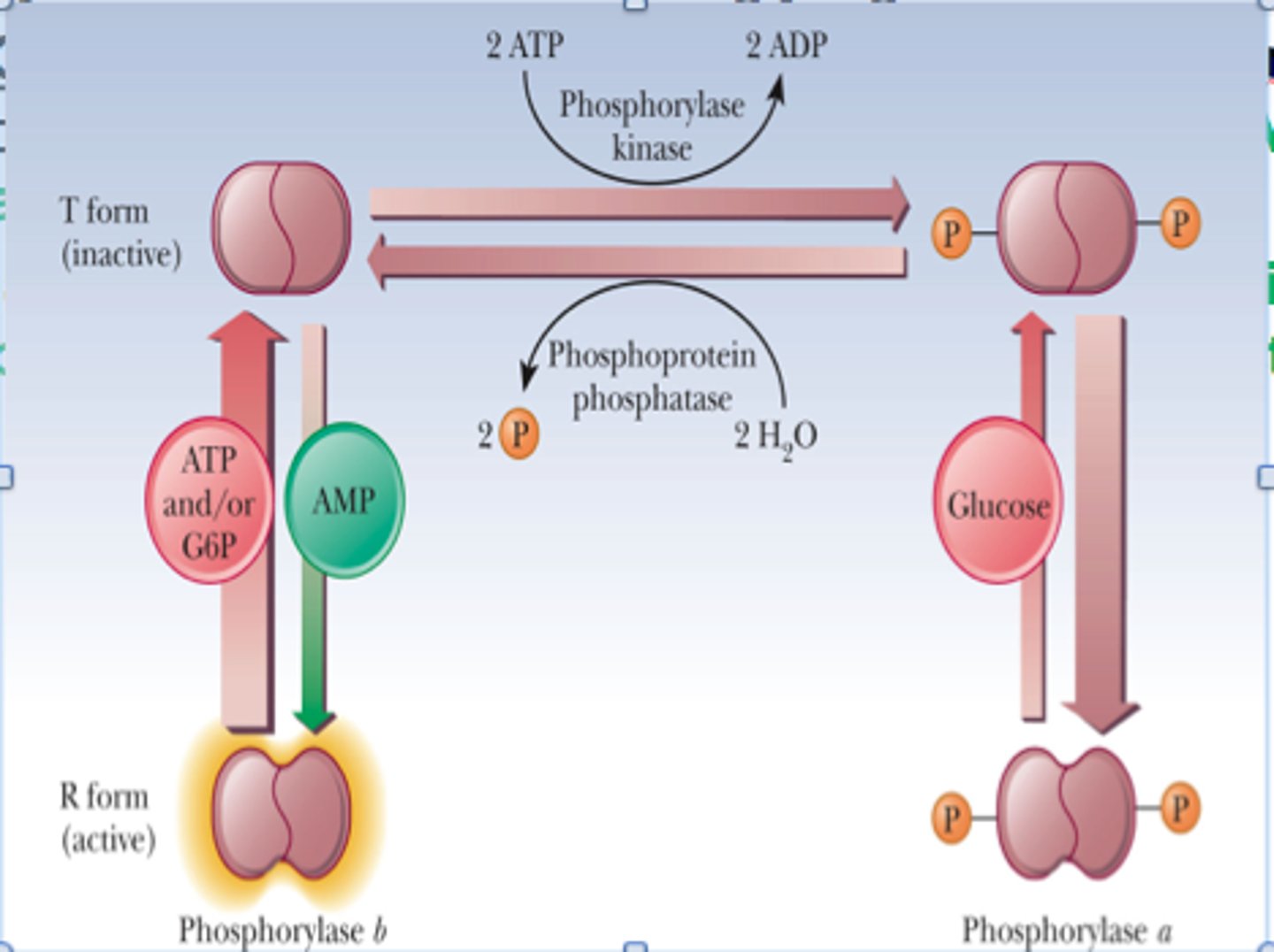
Allosteric Regulation of Liver Glycogen Phosphorylase
-inhibited by glucose (binding of glucose a -> b form)
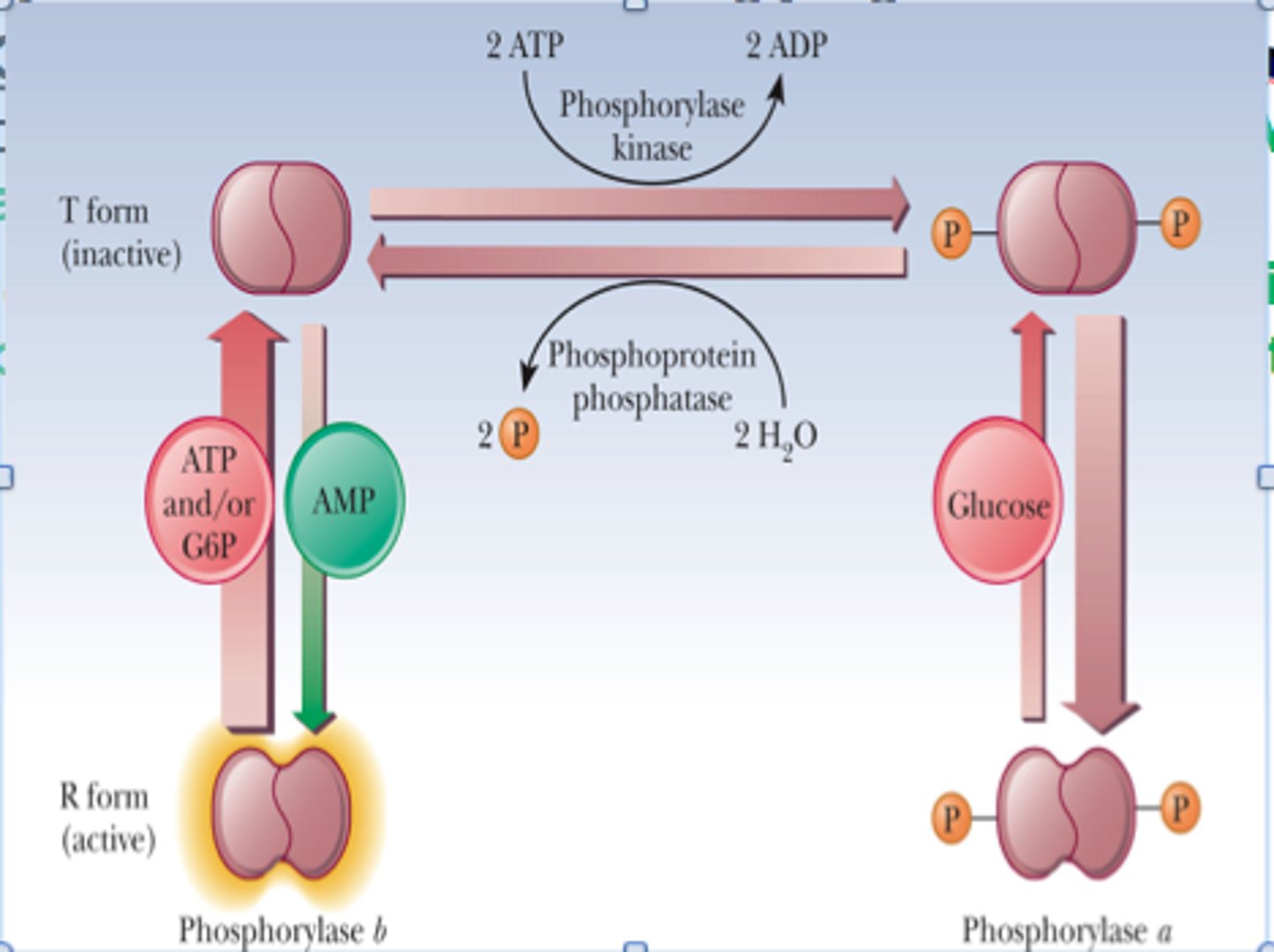
Allosteric Regulation of Muscle Glycogen Phosphorylase
-stimulated by AMP (low energy charge = low ATP/high AMP = glucose insufficient = bind and stabilize R state = glycogen brokendown)
-inhibited by ATP (high energy charge = high ATP = sufficient glucose = glycogen not degraded)
-inhibited by glucose 6-phosphate (binds same site as ATP and inhibits through feedback inhibtion)
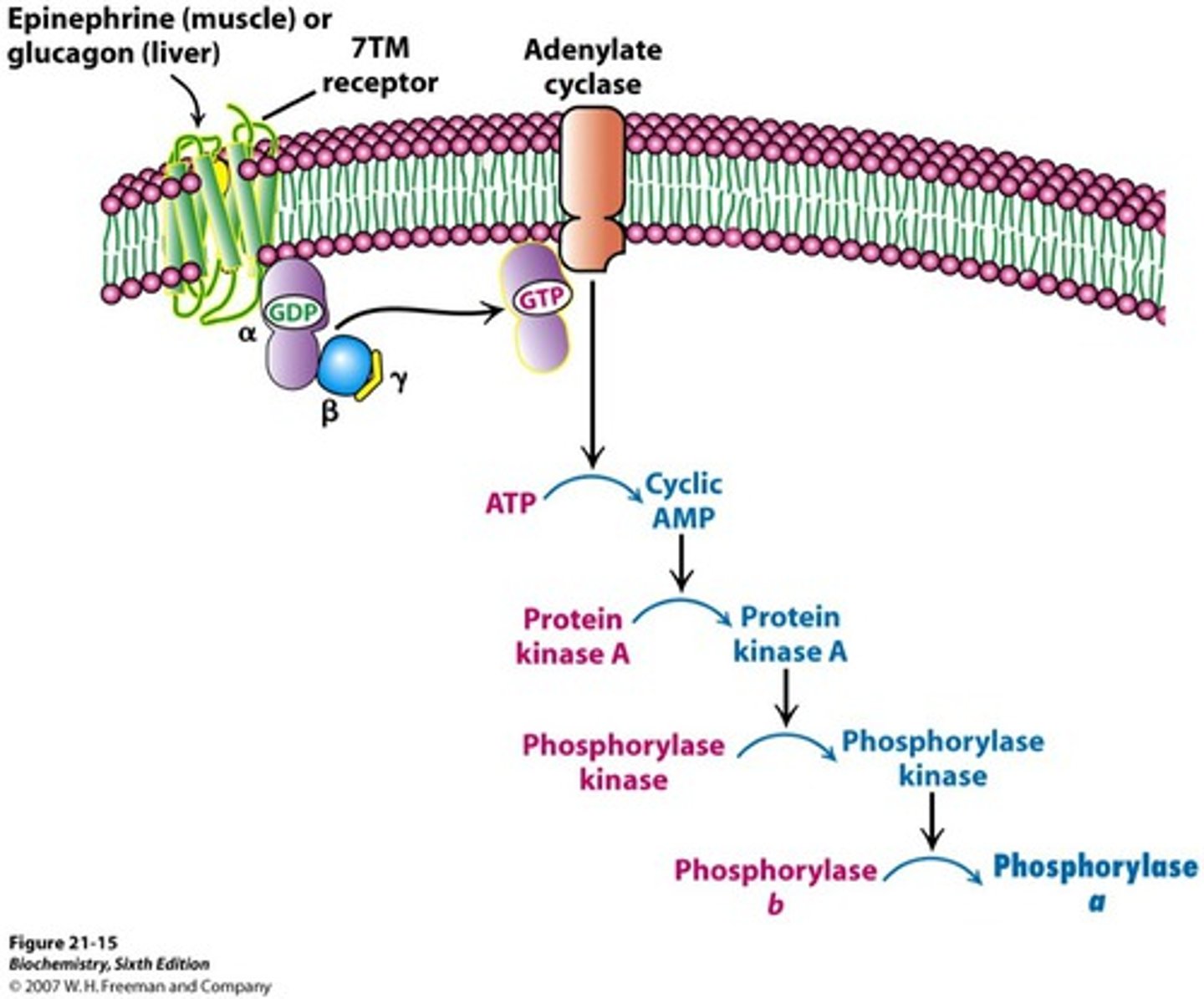
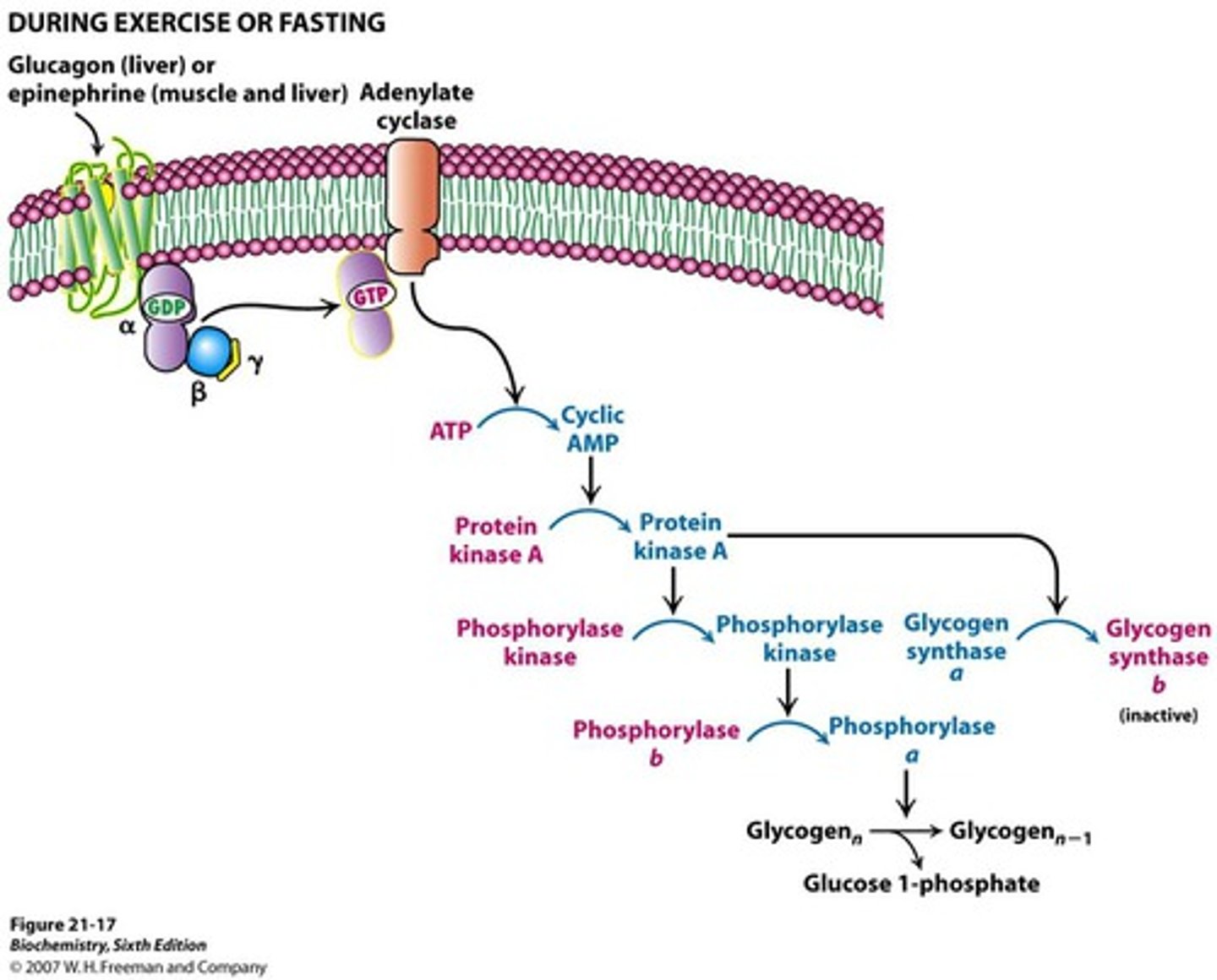
Hormonal Regulation of Glycogen Phosphorylase
-reversible phosphorylation of serine converts b -> a (activates enzyme)
-regulated by phosphorylase kinase
-main hormones include glucagon and epinephrine (low blood glucose = glucagon secreted by liver)
(exercise = epinephrine secreted by muscle)
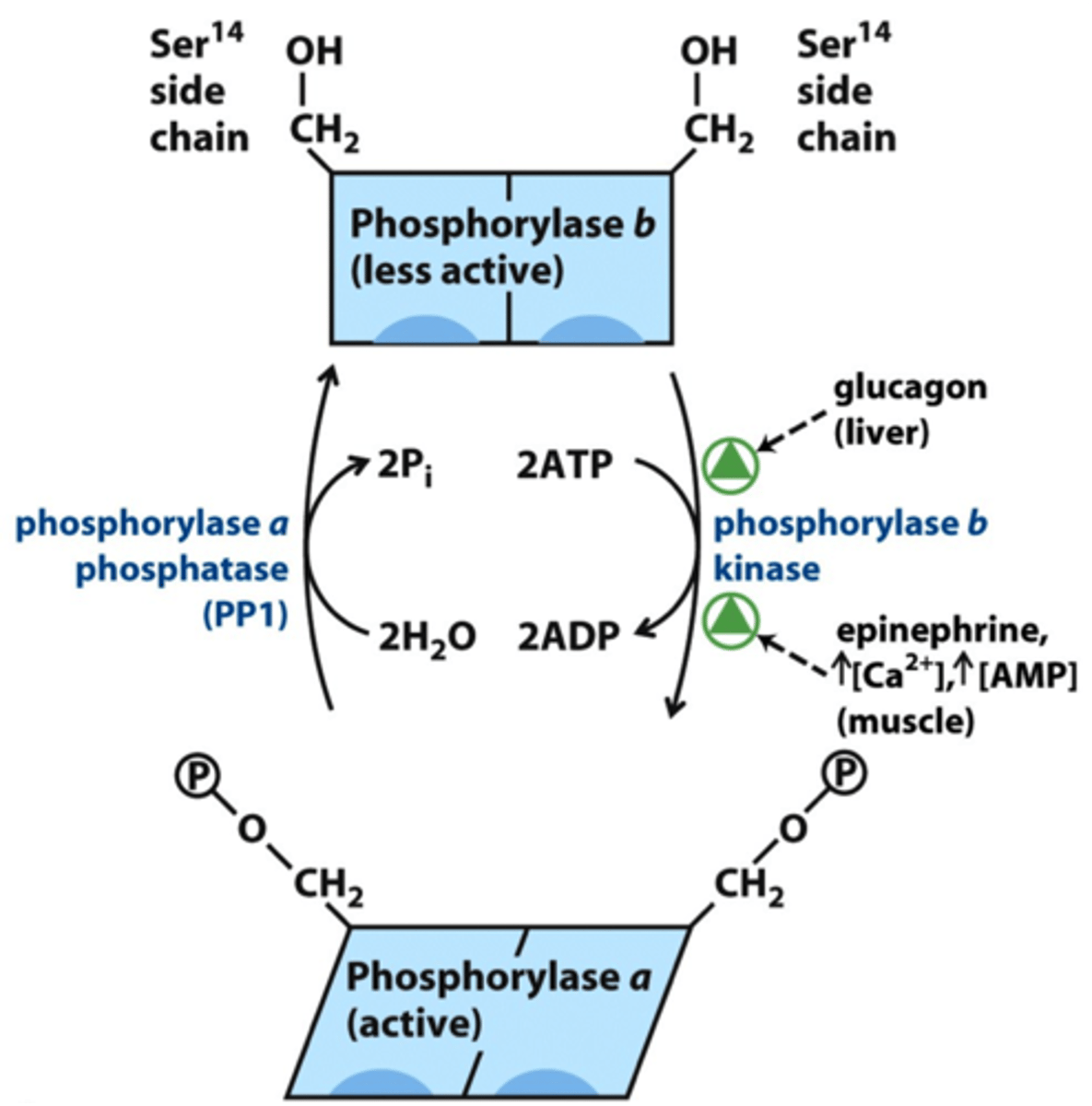
Mechanism of Hormonal Regulation of Glycogen Phosphorylase
-protein kinase A activates phosphorylase kinase and initiates of cAMP signal transduction cascade
-phosphorylation of phosphorylase by PKA makes it more suscpetible to dephosphorylation (inactivation by PP1)
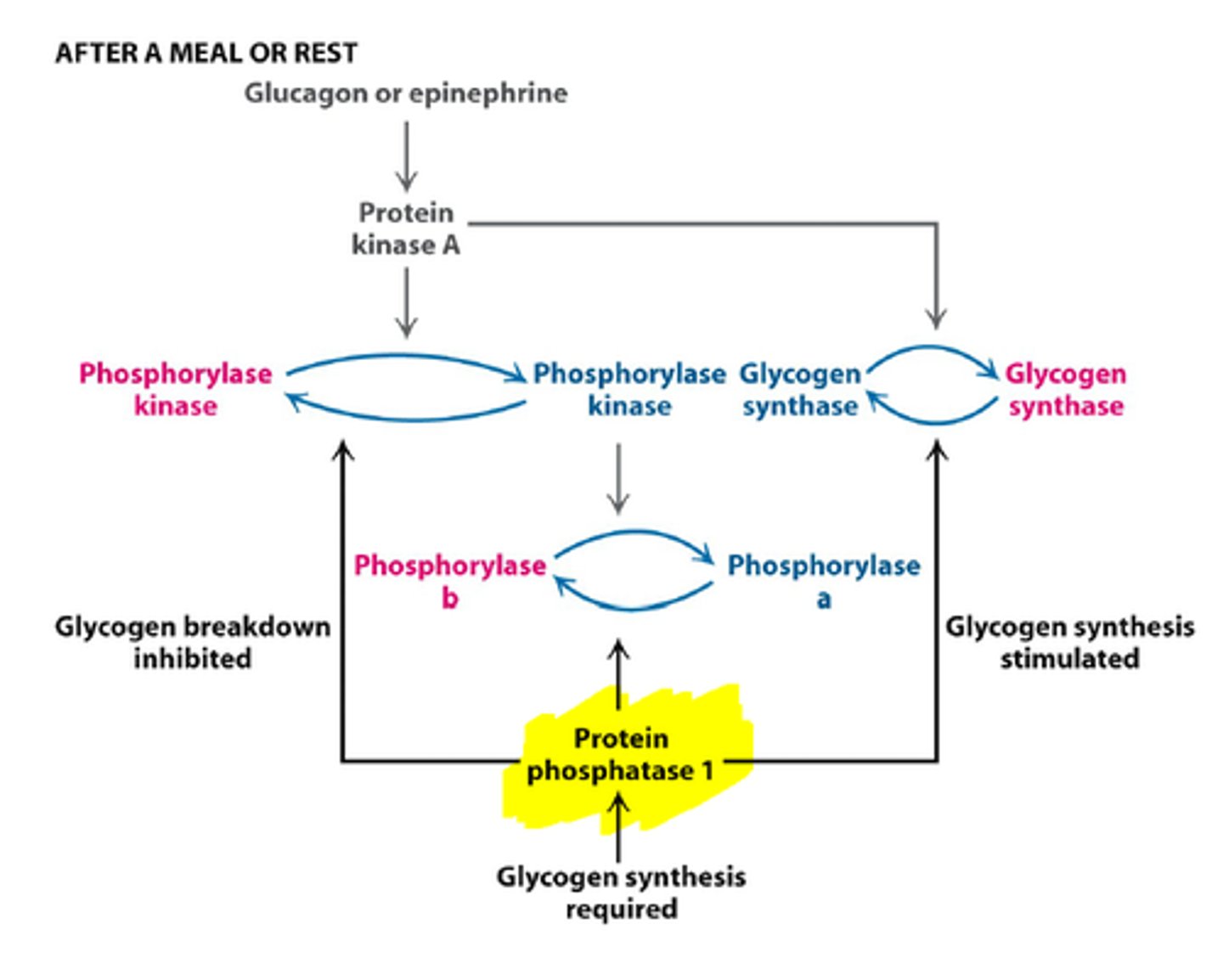
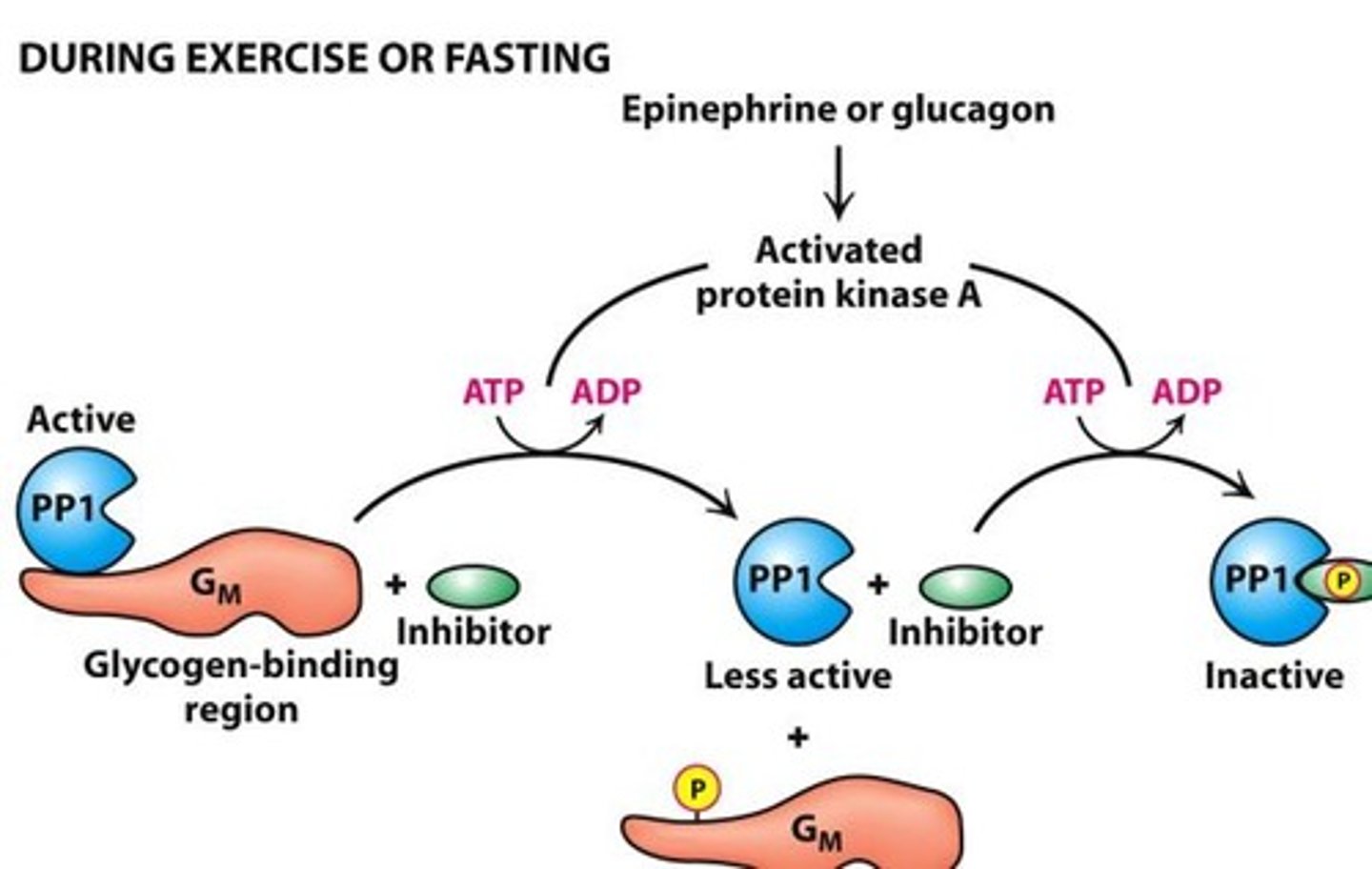
Protein Phosphatate 1 (PP1)
-inactive PP1 promotes glycogen degradation
-binding of epinphrine/glucagon activates PKA and causes
-PP1 active when bound to Gm
-PPI inactive when bound to inhibitor
Glycogen Synthase
-key regulatory enzyme in glycogen synthesis
-new glycolysis units added to nonreducing terminal residues of glycogen
-activated glycosyl units of UDP is transfered to hydroxyl group of C4 to form alpha 1,4-glycosidic linkages ONLY
-UDP is displaced by terminal hydoxyl group of growing glycogen molecule
-isozymic forms specific to liver and muscle
-can add glycosl resides to polysacchardie chain containign 4+ residues
Forms of Glycogen Synthase
-active (a) = unphosphorylated
-inactive (b) = phosphorylated
Allosteric Regulation of Glycogen Synthase
-activated by glycogen 6-phosphate (stabilizess R state) increased blood glucose = increase glucose 6-phosphate = increase glycogen synthesis
-inhibited by glycogen synthase kinase (GSK) and PKA (phosphrtylate and inactive a -> b)
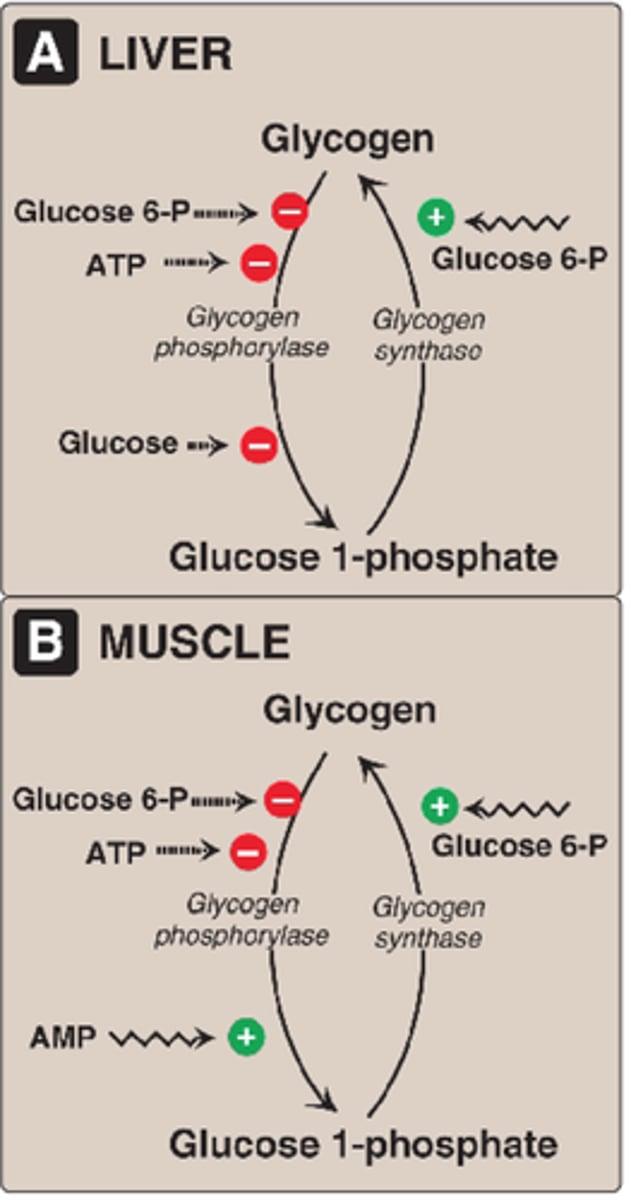
Hormonal Regulation of Glycogen Synthase
-inhibited by glucagon and epinephrine
-glycogen synthase kinase (GSK) and protein kinase A (PKA) add a phosphoryl group to glycogen synthase and decrease its enzymatic activity
-PP1 removes phosphoryl groups from glycogen synthase (b -> a)
-insulin stimulates synthesis of glycogen when blood-glucose conc is high (inactivates GSK)
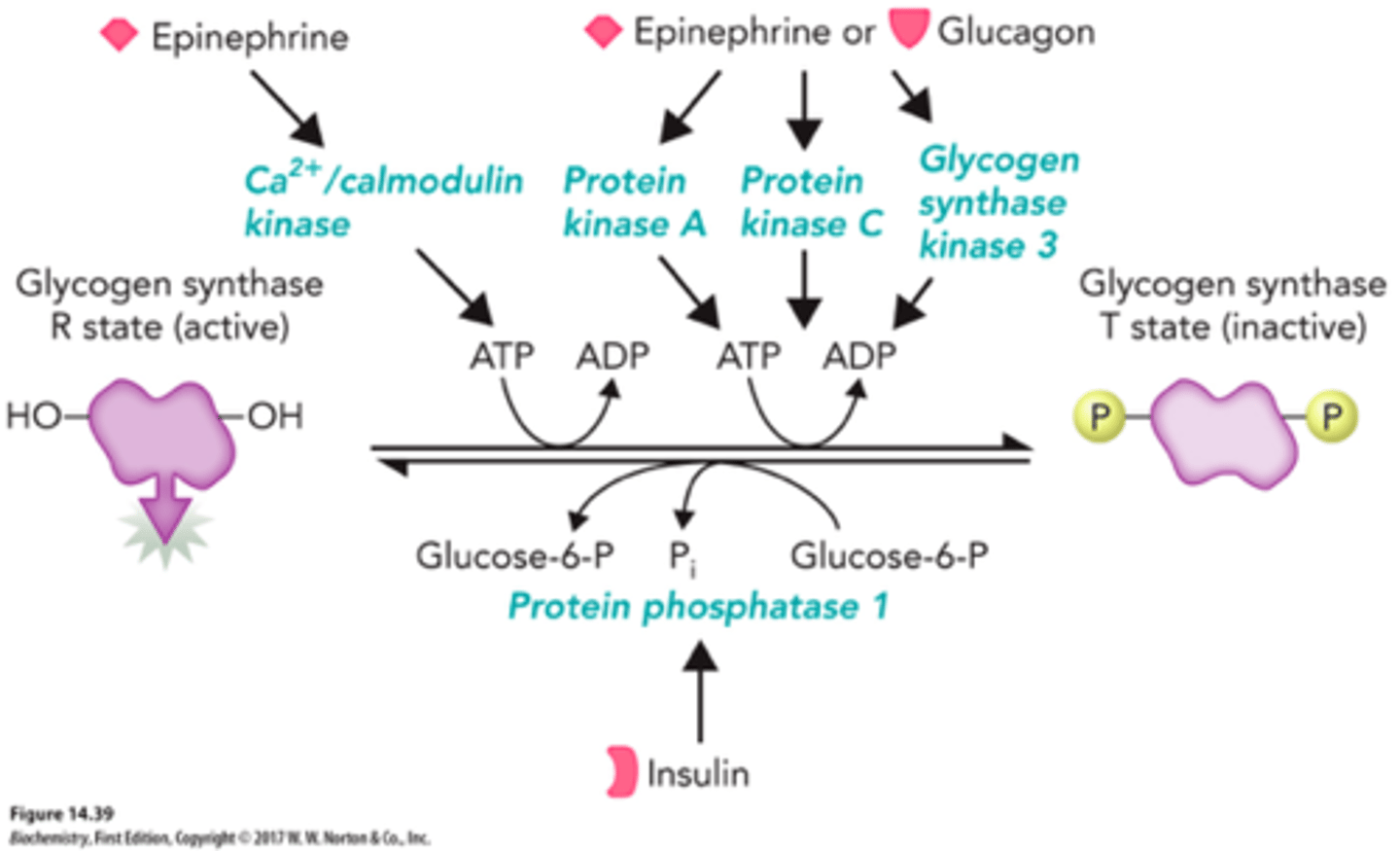
Glycogen Synthase Kinase
phosphorylates and inhibits glycogen synthase
Is phosphorylase kinase a substrate for phosphorylation by PKA?
yes
PKA upon cAMP mediated activation phosphorylates the alpha and beta subunit of phosphorylase kinase and activates it.
Is phosphorylase b a substrate for phosphorylation by PKA?
no
Is glycogen synthase kinase a substrate for phosphorylation by PKA?
no
Is glycogen synthase a a substrate for phosphorylation by PKA?
yes
What is the more active form of glycogen phosphorylase?
a
What is the phosphorylated form of glycogen phosphorylase?
a
What is the more active form of glycogen synthase?
a
What is the phosphorylated form of glycogen synthase?
b
What is the role of AMP on glycogen phosphorylase?
activates muscle phosphorylase b (inhibits degradation)
What is the role of ATP on glycogen phosphorylase?
inhibits muscle phosphorylase b
What is the role of glucose on glycogen phosphorylase?
activates liver phosphorylase a
What is the role of glucose 6-phosphate on glycogen phosphorylase?
inhibits muscle phosphorylase b
What is the effect of epinephrine on liver phosphorylase?
increase
What is the effect of epinephrine on muscle phosphorylase?
increase
What is the effect of epinephrine on liver glycogen synthase?
decrease
What is the effect of epinephrine on muscle glycogen synthase?
decrease
What is the effect of glucagon on liver phosphorylase?
increase
What is the effect of glucagon on muscle phosphorylase?
no effect
What is the effect of glucagon on liver glycogen synthase?
decrease
What is the effect of glucagon on muscle glycogen synthase?
no effect
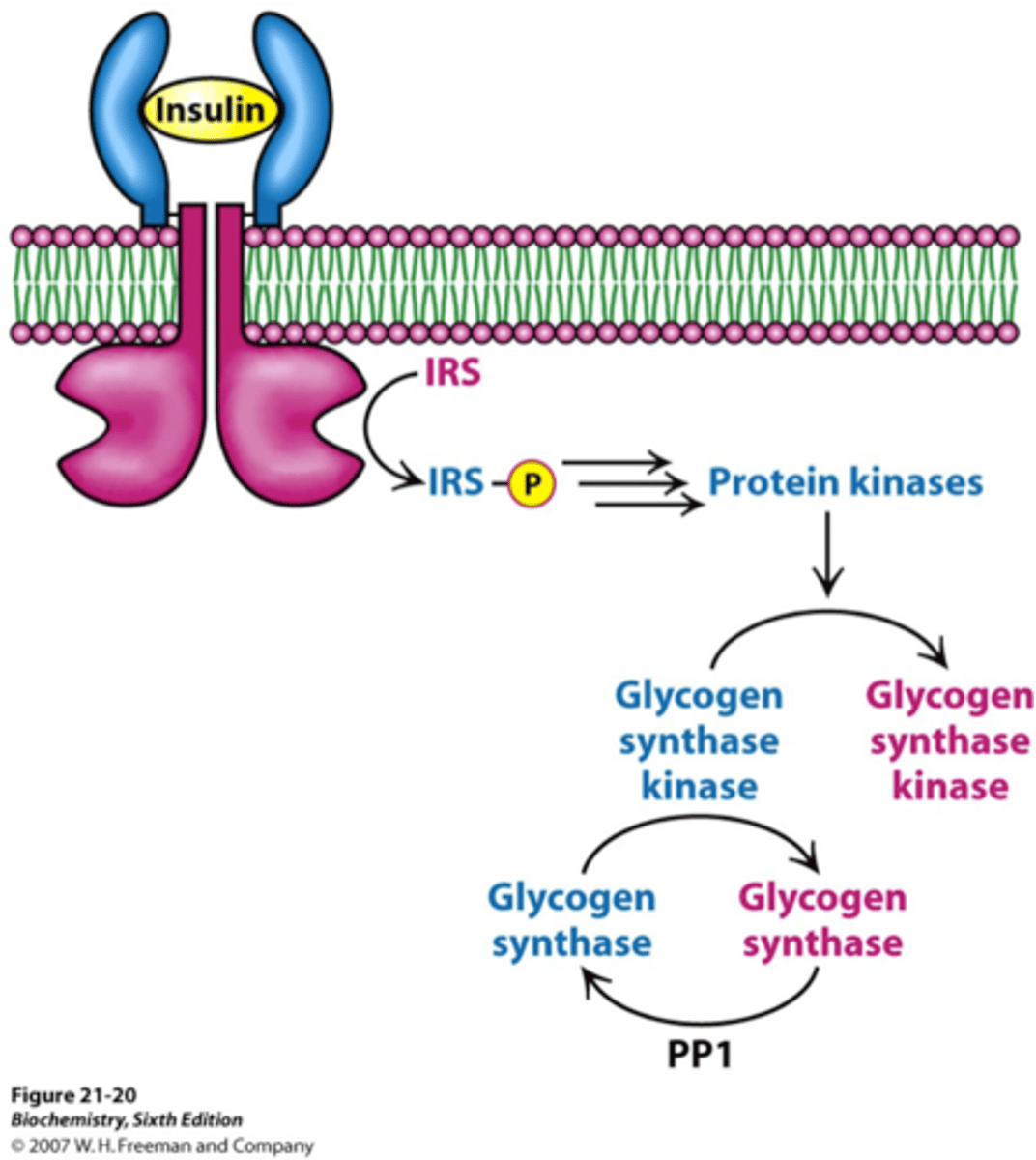
How does insulin activate glycogen synthase?
activates a protein kinase cascade that leads to dephosphorylation of glycogen synthase
Phosphorylase kinase is activated when it is phosphorylated by ____ in a signaling cascade initiated by _____ in muscle.
PKA
epinephrine
Insulin promotes gylcogen synthesis. One of the main effects of insulin is to activate Akt, a protein kinase which catalyzes the phosphorylation and inactivation of ____________________. This halts the phosphorylation of ___________________. The enzyme _________________ catalyzes the dephosphorylation of _________________ to its ________ state.
glycogen synthase kinase
glycogen synthase a
phosphoprotein phosphate 1
glycogen synthase b
active
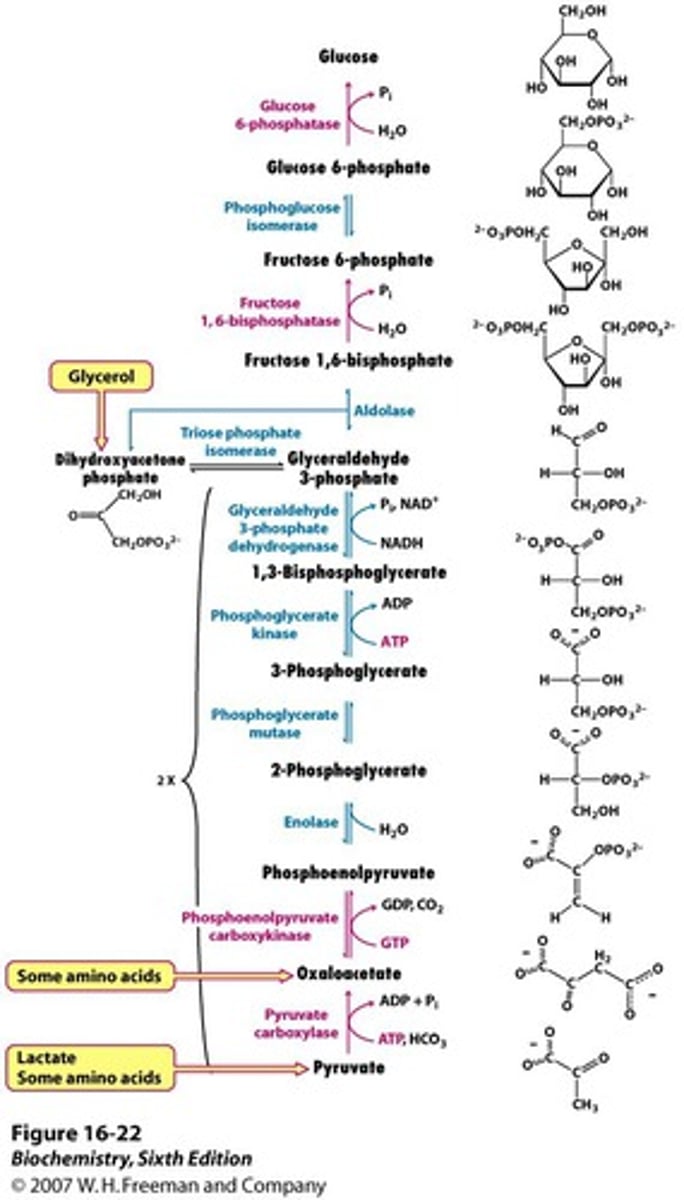
Gluconeogenesis
formation of glucose from 2 molecules of pyruvate
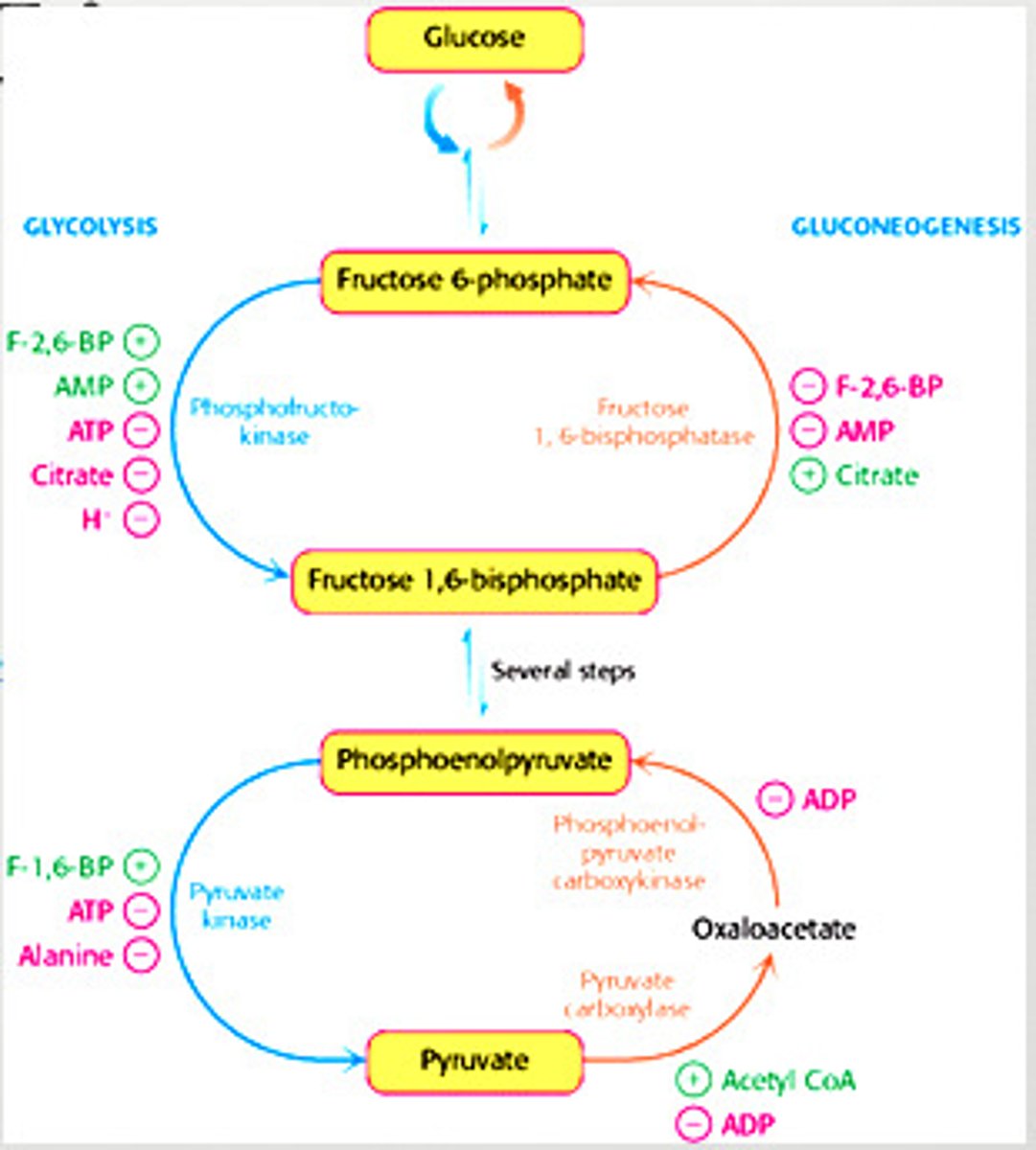
Regulation of Glycolysis
-controlled by enzymes that catalyze irreversible reactions (large, negative delta G) = hexokinase, phosphofructokinase, and pyruvate kinase
-also controlled by energy charge (inhibtied at high level)
Regulation of Glycolysis in the Muscle - Phosphofructokinase
-main site of control (comitted step)
-allosterically inhibited by ATP
-allosterically stimulated by AMP (can compete with ATP)
-inhibited by a decrease in pH (increases inhibitory effect of ATP)
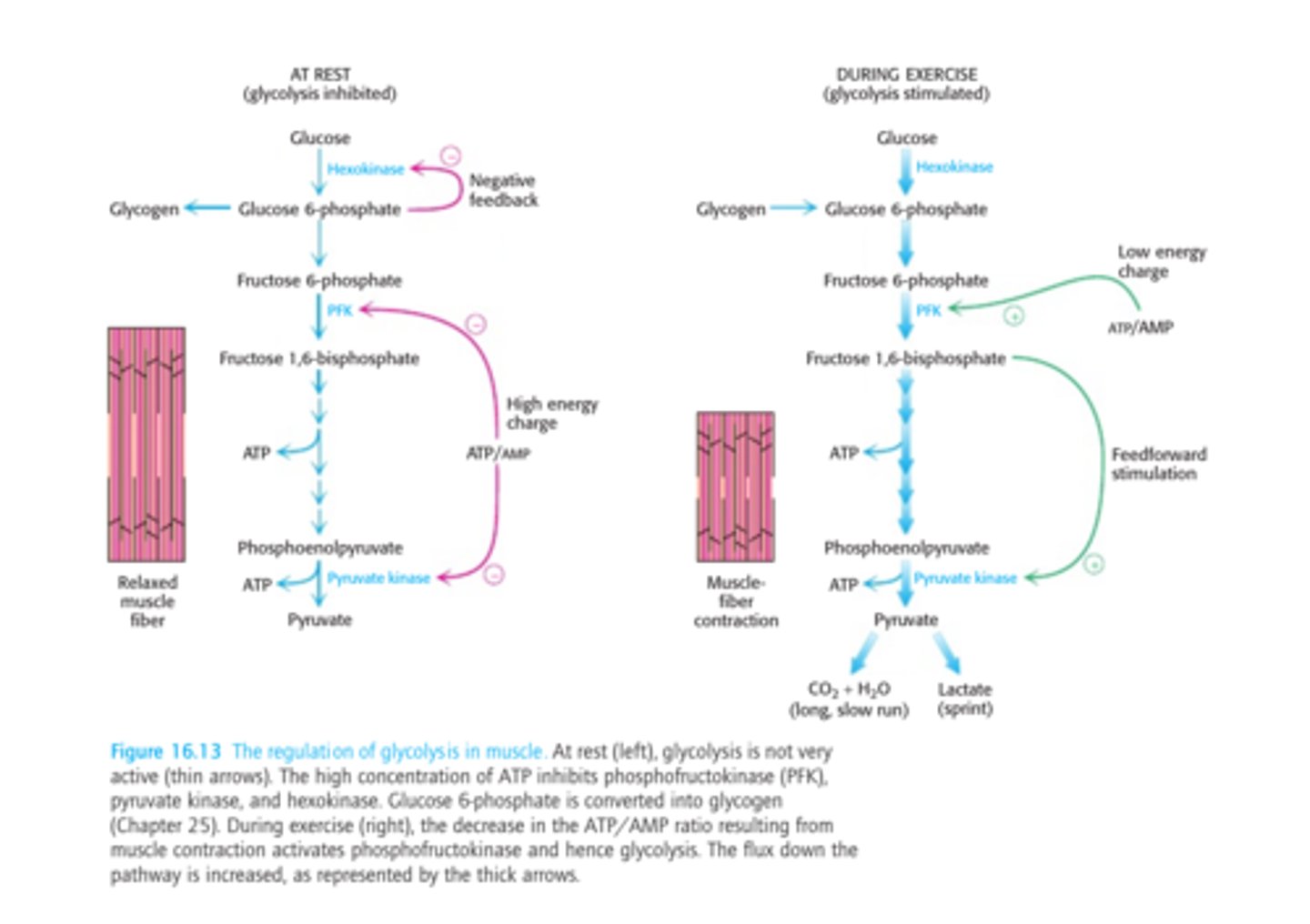
Regulation of Glycolysis in the Muscle - Hexokinase
-catalyzes the first step in glycolysis
-feedback inhbition by glucose 6-phosphate
-indirectly inhibited by phosphofructokinase (inactive PFK = increased fructose and glucose 6-phosphate)
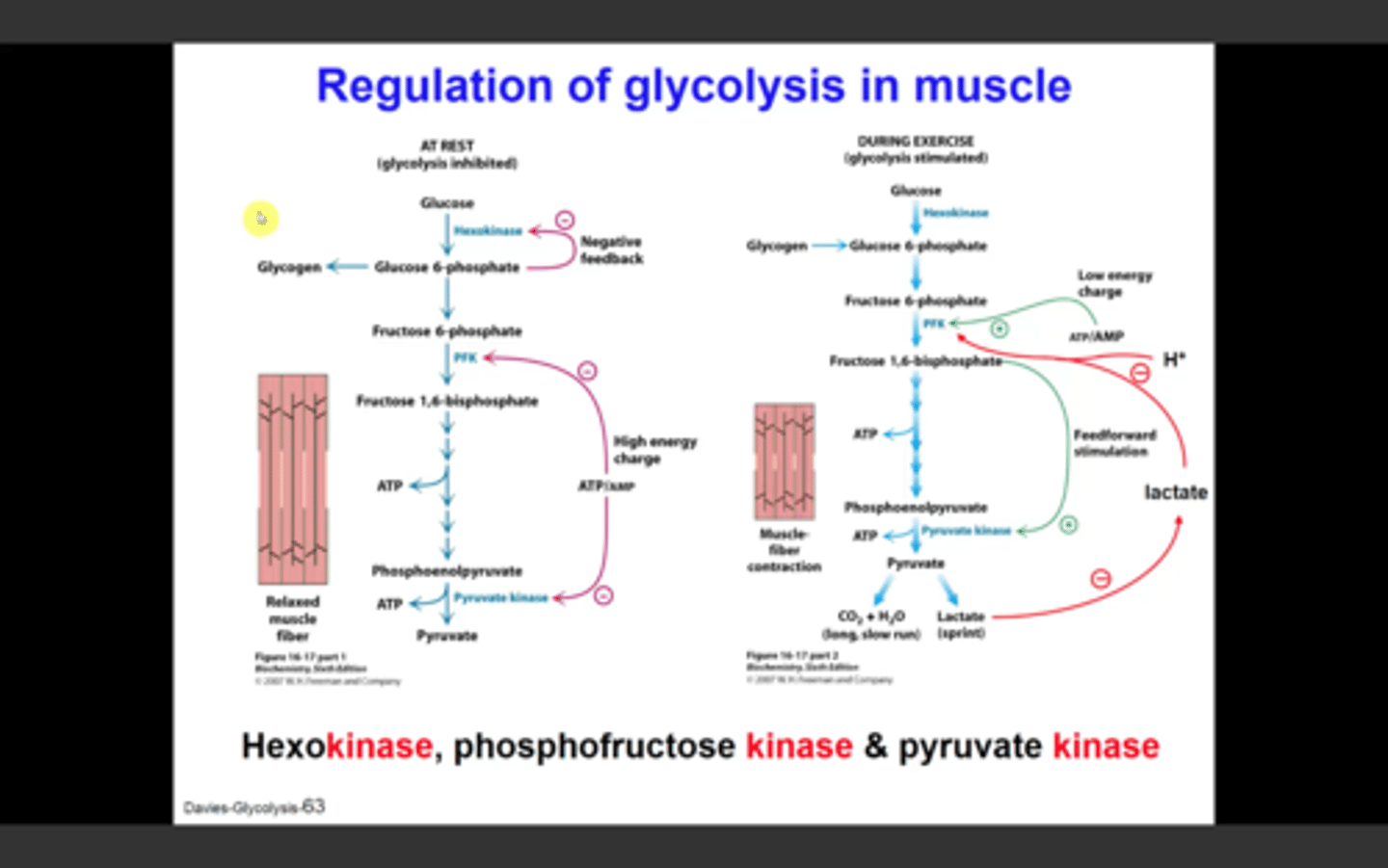
Regulation of Glycolysis in the Muscle - Pyruvate Kinase
-allosterically inhibited by ATP (high energy charge)
-feedforward stimulation by fructose 1,6-bisphosphate
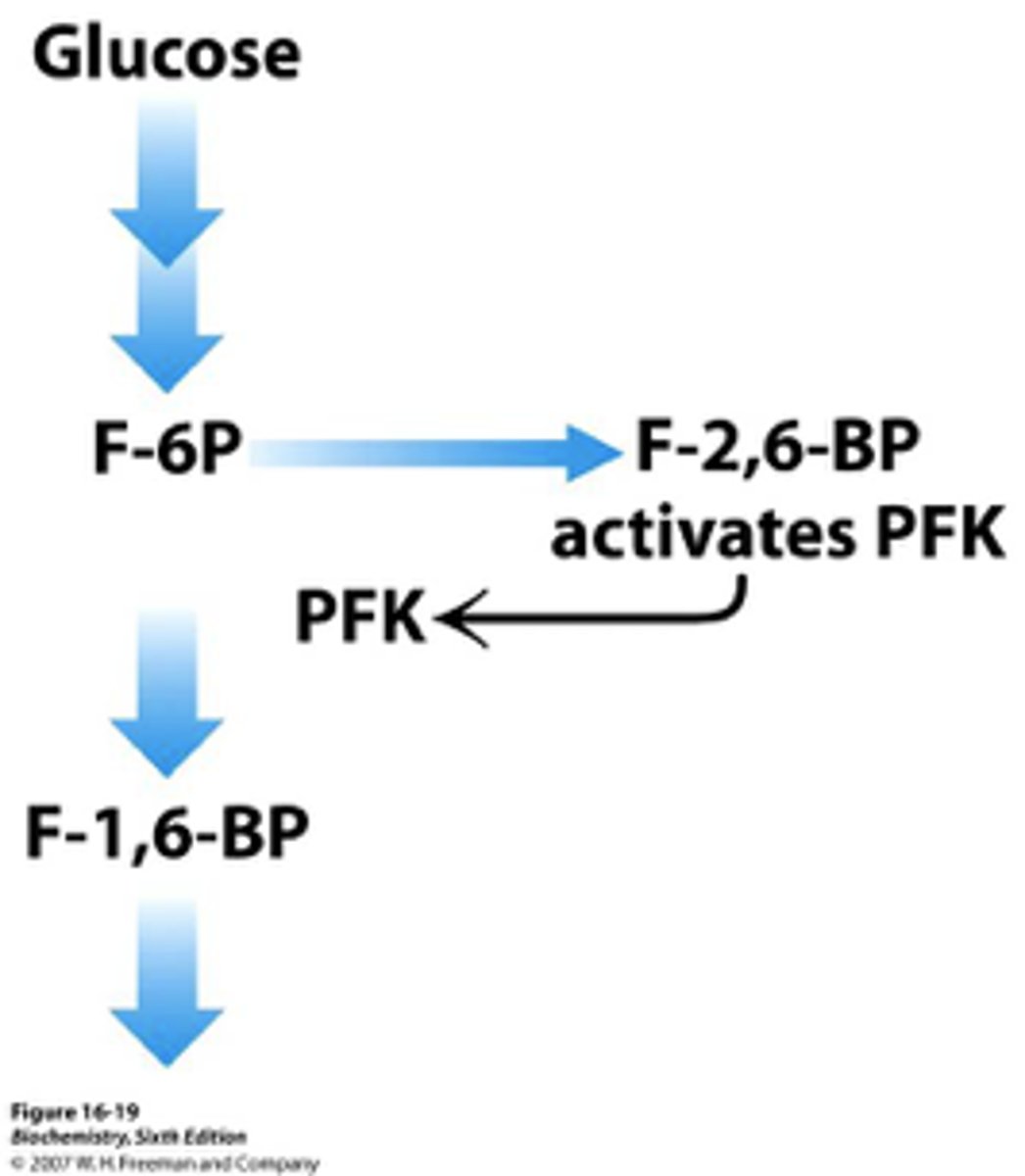
Regulation of Glycolysis in the Liver - Phosphofructokinase
-allosterically inhibited by ATP (not as much as muscle)
-inhibited by citrate
-stimulated by fructose 2,6-bisphosphate and fructose 6-phosphate (feedforward stimulation)
Regulation of Glycolysis in the Liver - Glucokinase
-inhibited by glucokinase regulatory protein (GKRP) at low glucose conc
-can only form glucose 6-phosphate when glucose is abundant
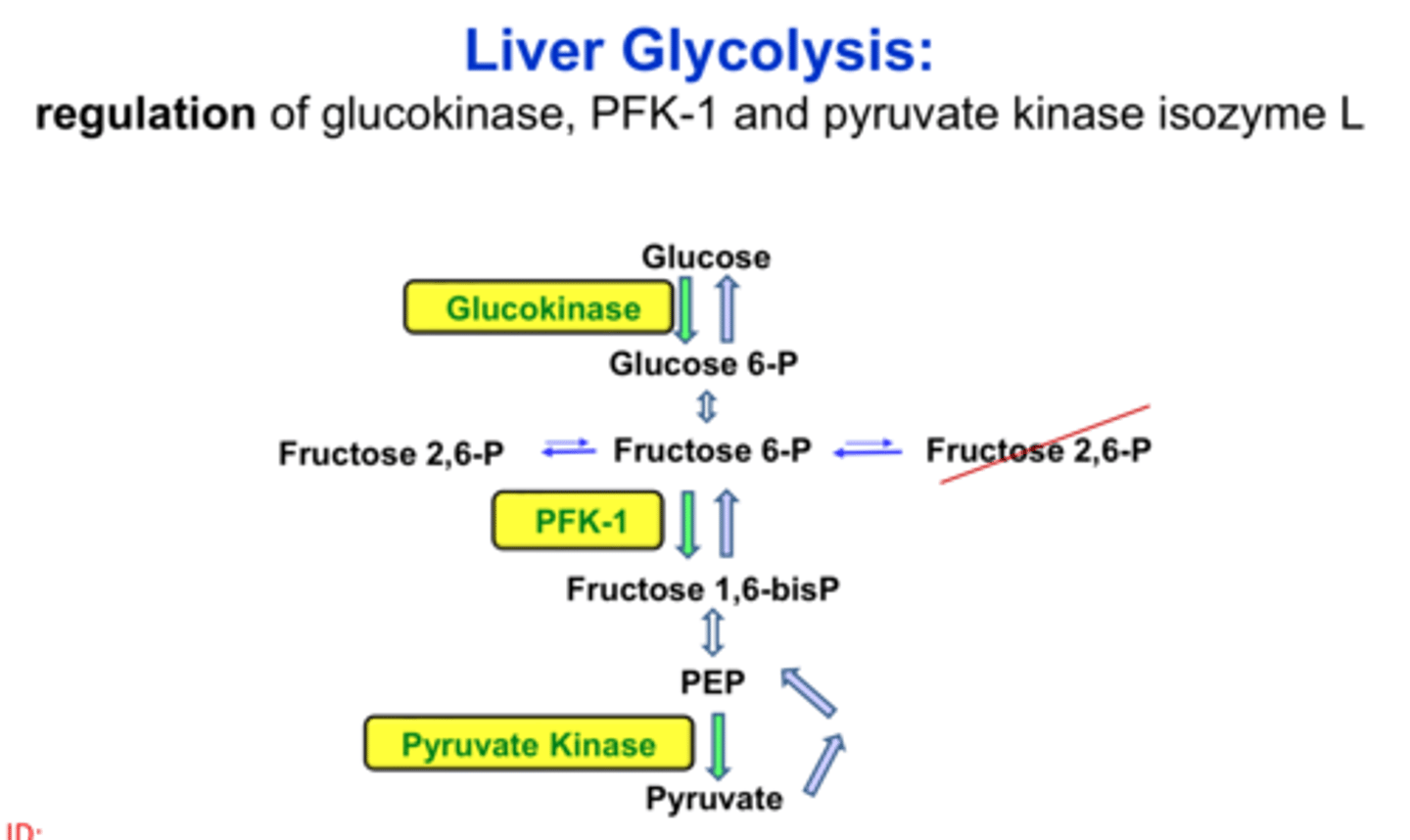
Regulation of Glycolysis in the Liver - Pyruvate Kinase
-allosterically inhibited by ATP
-activated by fructose 1,6-bisphophate (feedforward stimulation)
-inhibited by alanine
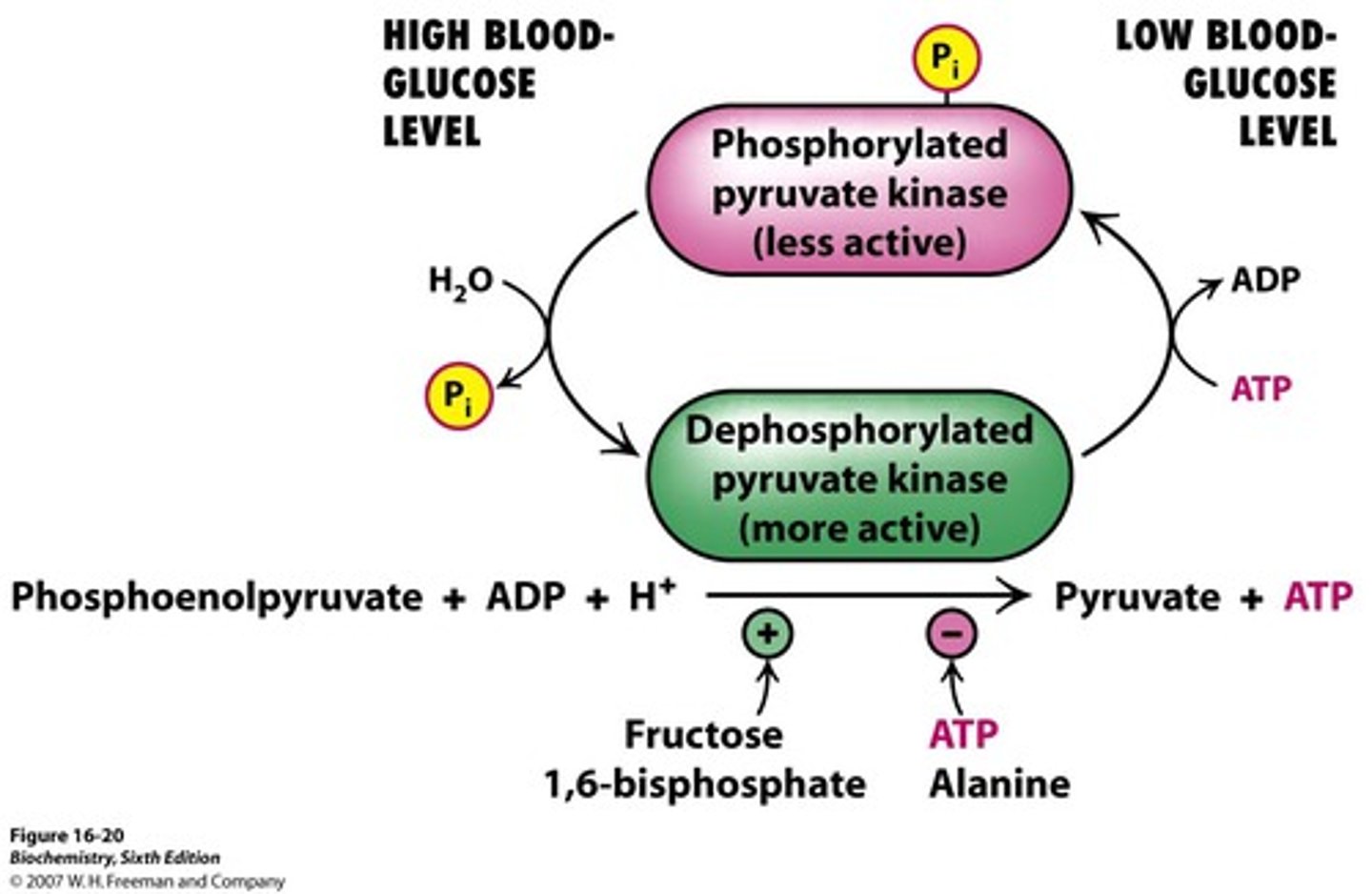
-phosphorylated and inactivated by cAMP cascad when blood glucose concentation is low (avoids consumption by liver)
Regulation of Gluconeogenesis
- Regulation is allosteric
- Primarily on enzymes that bypass the irreversible reactions in Glycolysis
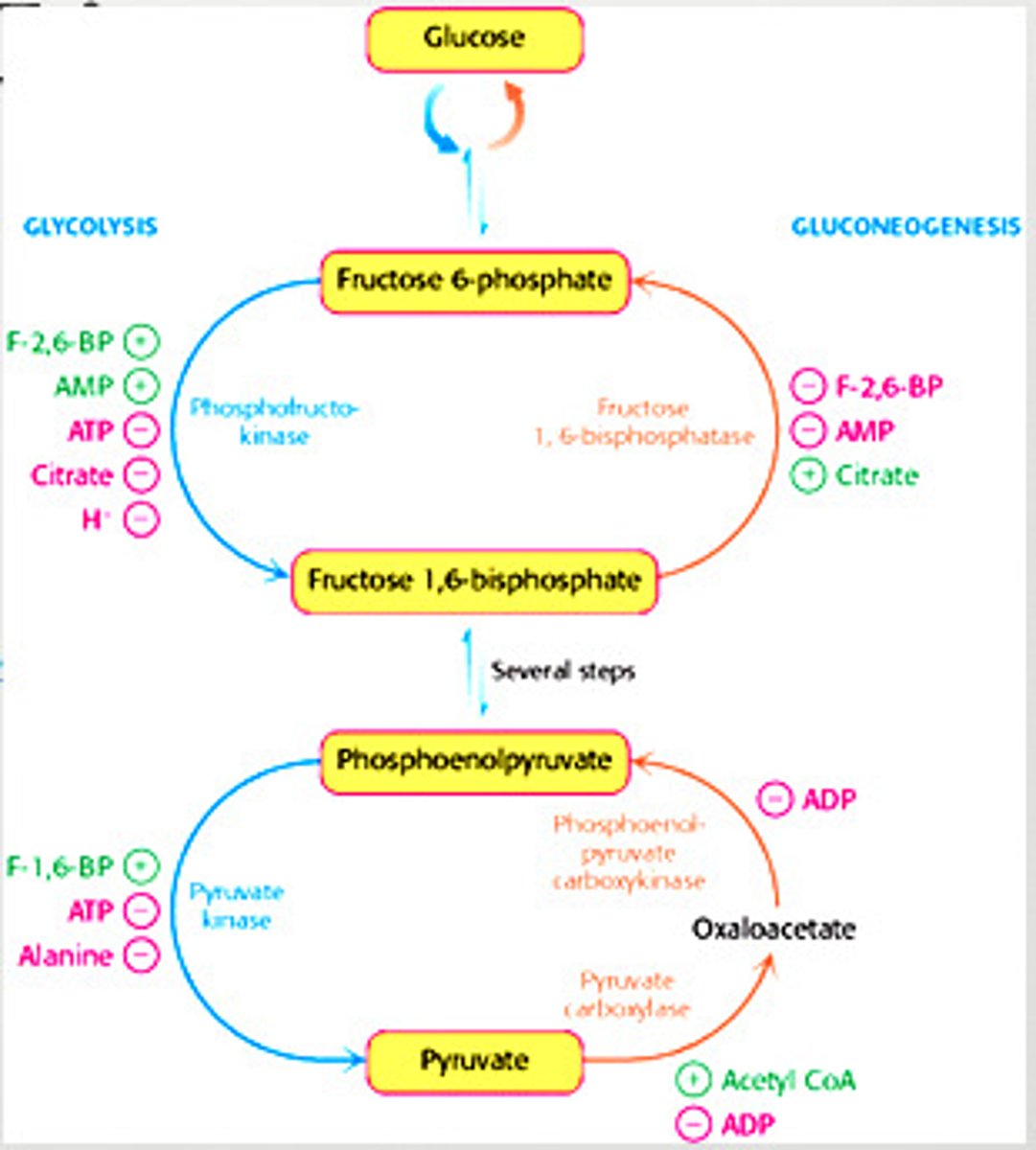
Distinct Enzymes of Gluconeogenesis
regulated in gluconeogenesis
pyruvate carboxylase (pyruvate -> oxaloacetate)
phosphoenolpyruvate caboxykinase (oxaloacetate -> phosphoenolpyruvate)
fructose 1,6-bisphosphate (F 1,6P -> F6P)
Regulation of Gluconeogenesis - Fructose 1,6-Bisphosphate
-inhibited by AMP
-inhibited by F-2,6-BP
-stimulated by citrate
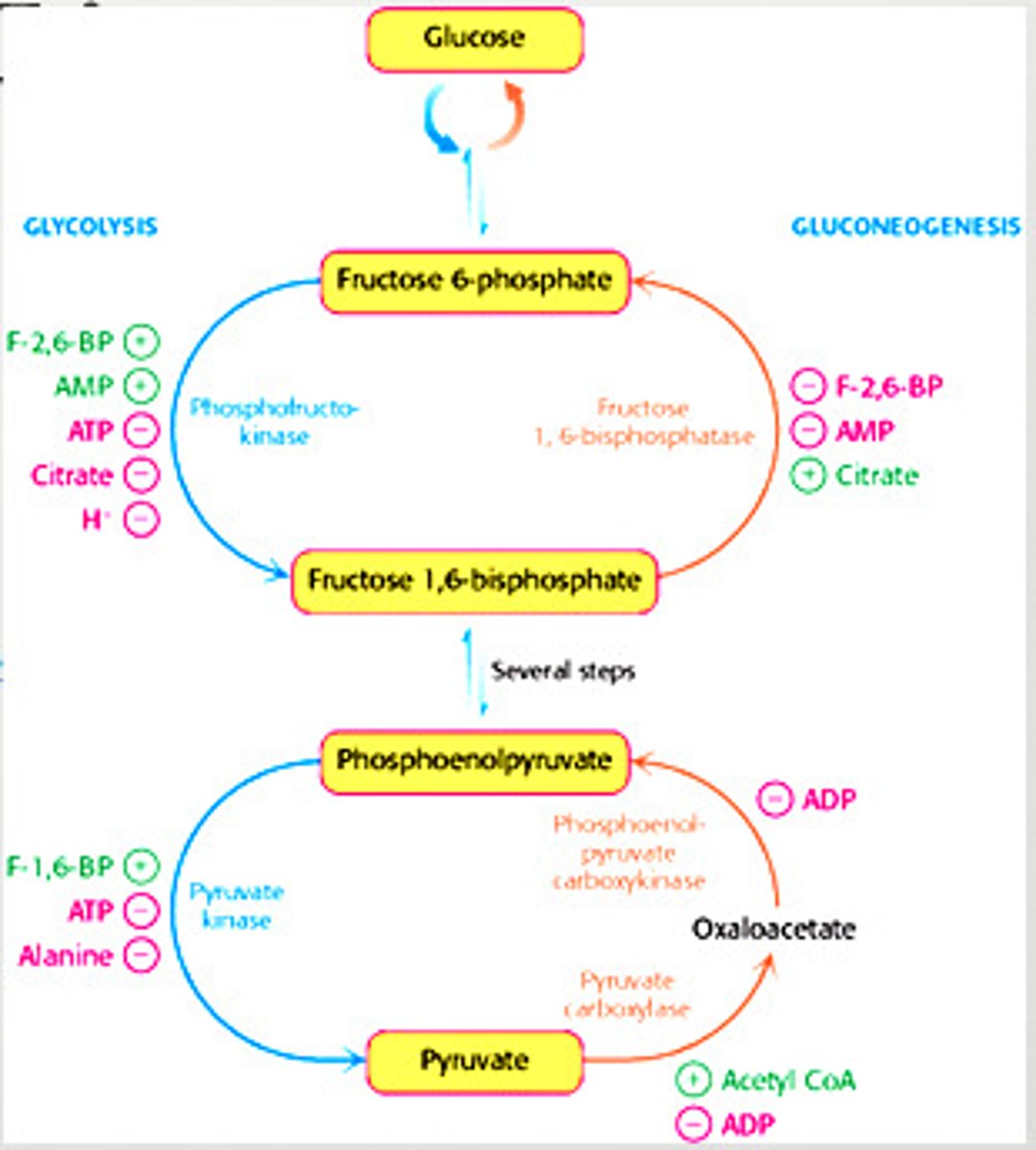
Regulation of Gluconeogenesis - Pyruvate Carboxylase
-inhibited by ADP
-activated by acetyl CoA
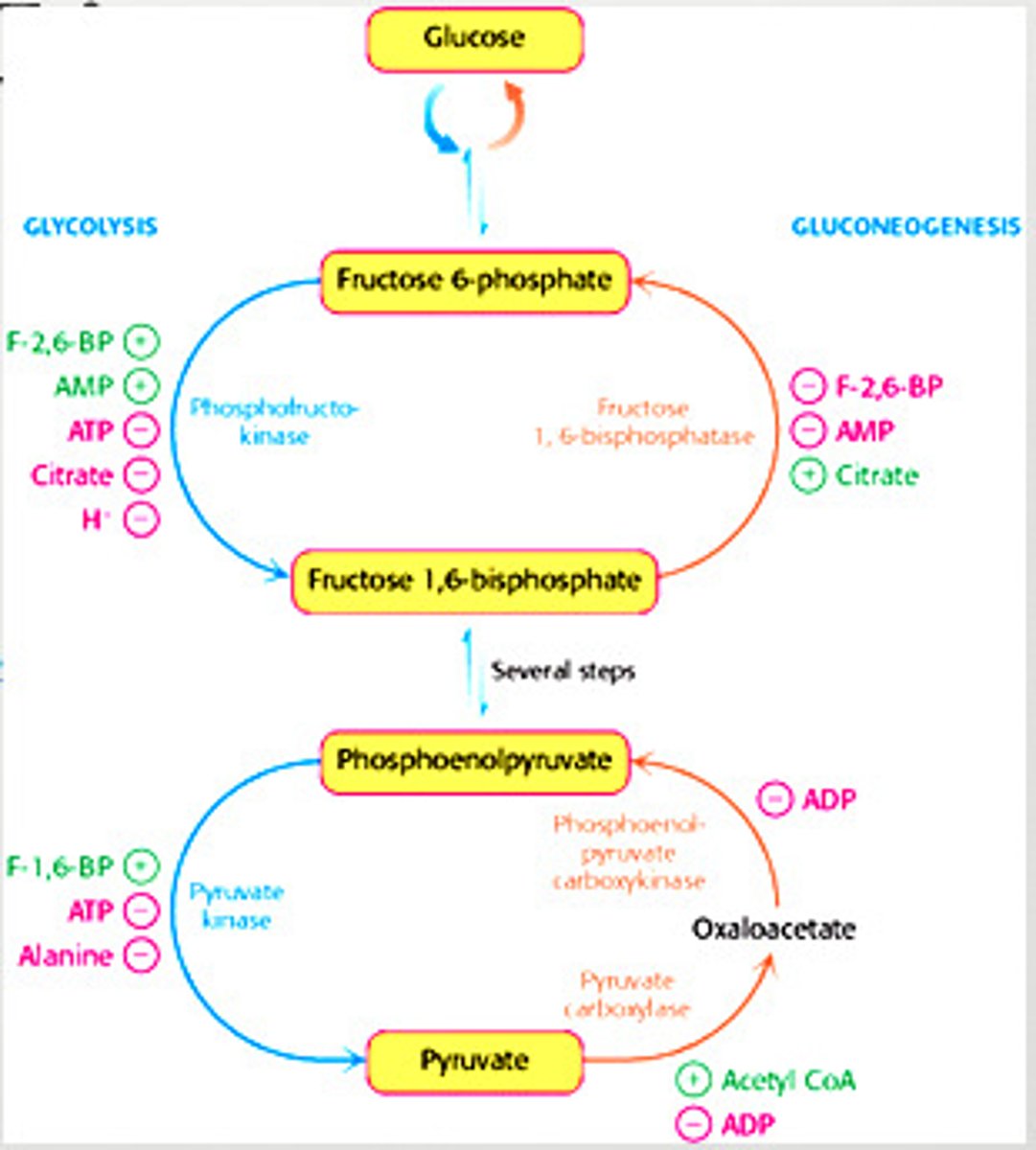
Regulation of Gluconeogenesis - Phosphoenolpyruvate Carboxykinase
-inhibited by ADP
What is the rate of glycolysis dependent on?
concentration of glucose
What is the rate of gluconeogenesis dependent on?
lactate and other precursors of glucose
How does energy charge reciprocally regulate gluconeogensis and glycolysis?
low energy charge stimulates glycolysis and inhibits gluconeogenesis (PFK stimulated and F-1,6-B inhibited)
high energy charge inhibits glyoclysis and stimulates gluconeogenesis (PFK inhibited and F-1,6-B stimulated)
How do glucose levels reciprocally regulate gluconeogensis and glycolysis?
glucose abundant = glycolysis
glucose scarce = gluconeogensis
What enzymes are involved in balancing the rate of glycolysis and gluconeogensis in the liver?
PFK and F-2,6B