chemistry 112 exam 3
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
chemical equliibrium
rate of forward reaction equals rate of reverse reaction
dynamic process
temperature dependent
equilibrium constant
Kc or Kp
relates molar concentrations of the products and reactants after the equilibrium has been reached
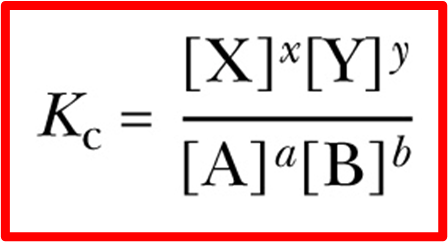
only include aqueous solutions and gases (not solids and liquids)
for Kp, use partial pressures
equilibrium constant evaluation
Kc > 1: reaction goes to completion, favors products
Kc < 1: no reaction, favors reactants
Kc value for reverse reactions
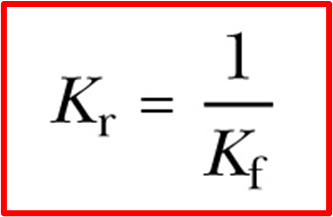
and vice versa
how to add reaction Kc
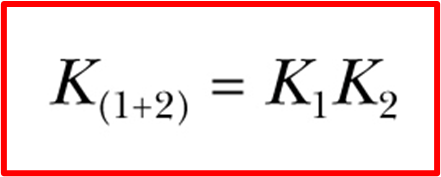
multiplying reactions K
(A+B = C+D) x (A+B = C+D) : K = (K1)²
how to compare Kp and Kc

if change in n = 0, Kp = Kc
ICE table
for solving equilibrium calculations
I: initial concentrations
C: change (+ for amount generated, - for amount used)
E: equilibrium concentrations
PAY ATTENTION TO STOICHIOMETRY FROM EQUATION
reaction quotient
Qc and Qp
predicts which direction the reaction will proceed
calculated like equilibirum constant
for concentrations not at equilibrium
reaction quotient analysis
Qc < Kc: reactions converted to products, forward reaction
Qc = Kc: system is at equilibrium
Qc > Kc: products converted to reactants, reverse reaction
le chatelier’s principle
reactions respond to change to minimize the change and reestablish equilibrium
temperature change: shift based on endothermic and exothermic reactions (if exothermic, moves towards reactants)
concentration change: high concentration of reactants, more products made and vice versa
volume/pressure change: shift based on # of gaseous moles (if pressure increased, moves to side with less moles) (if pressure decreased, moves to side with more moles)
arrhenius definitions of acid/base
acid: produced H+ in solution
base: produces OH- in solutions
bronsted lowry definitions of acid/base
acid: proton donor
base: proton acceptor
lewis definition of acid/base
acid: electron pair acceptor
base: electron pair donor
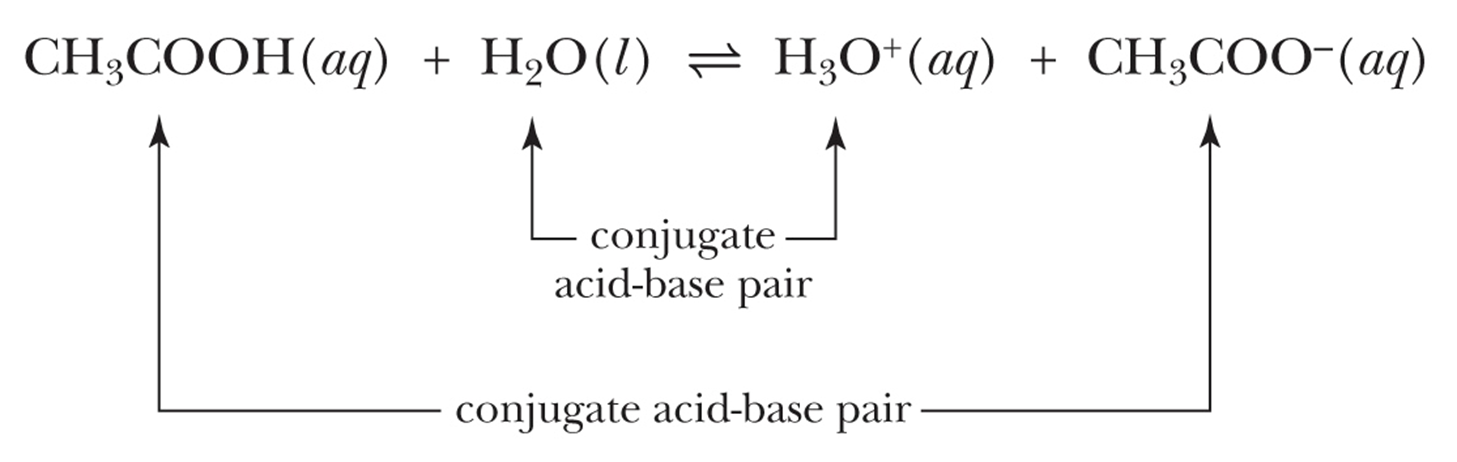
conjugate acid-base pair
acid and corresponding base on other side
base and corresponding acid on other side
strong acids and bases
acids: HCl, HI, HBr, HClO4, HNO3, H2SO4 (dissociate 100% in water) (Ka > 1)
base: LiOH, NaOH, KOH, Ca(OH)2, Sr(OH)2, Ba(OH)2
ion-product constant
Kw
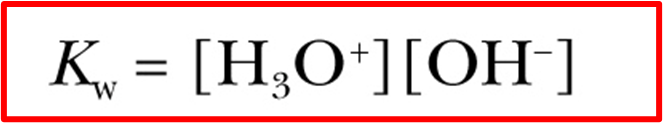
Kw = 1.0 × 10^-14 (at room temp, 25°C)
14 = pH + pOH
pH and pOH
small pH = acidic (<7)
large pH = basic (>7)
pH = -log([H+]) AND [H+] = 10^-pH
pOH = -log([OH-]) AND [OH-] = 10^-pOH
weak acids
dissociate less than 100%
any acid that isn’t strong
Ka < 1
% ionization
only for weak acids
( [H+] / [acid or base(initial)] ) x 100
acid dissociation/base protonation constant
Ka/Kb
HA(aq) + H2O(l) ⇌ H3O+(aq) + A-(aq)
Ka = [H3O+][A-] / [HA]
can determine pH of weak acid solution or pOH of weak base solution from this using ICE table
large Ka = strong acid
small Ka = weak acid
weak bases
react less than 100%
any base not listed as strong
EX: NH3, OH-, HCOO-, CH3COO-
acid-base reaction (neutralization rxn)
acid (aq) and base (aq) combine to create a salt (aq) and water (l)
ionization reaction
HA = H+ + A-
EX: HCOOH = HCOO- + H+
oxyacid strength
oxyacid: X - O - H
higher oxidation # or higher # of O’s = stronger oxyacid