BIOC 4331 Lecture 5
1/44
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
What is horizontal (lateral) gene transfer?
Transfer of genes between species, most common in bacteria, which can falsely suggest close evolutionary relationships.
What are homologs?
Proteins with similar sequences due to shared evolutionary origin.
What are paralogs?
Homologous proteins found within the same species.
What are orthologs?
Homologous proteins found within different species.
What does hypervariable mean in protein sequences?
Positions with no functional or structural role, free to mutate.
What does invariant mean in protein sequences?
Positions required for function or 3D structure that must not change.
What does a “conservative” substitution mean?
A residue changes to another with similar properties (e.g., Asp -> Glu)
What does sequence identity mean in protein sequence alignment?
The percentage of positions with identical amino acids between aligned sequences.
Which substitutions give the highest alignment scores?
Identical amino acids and conserved substitutions (e.g., Asp -> Glu)
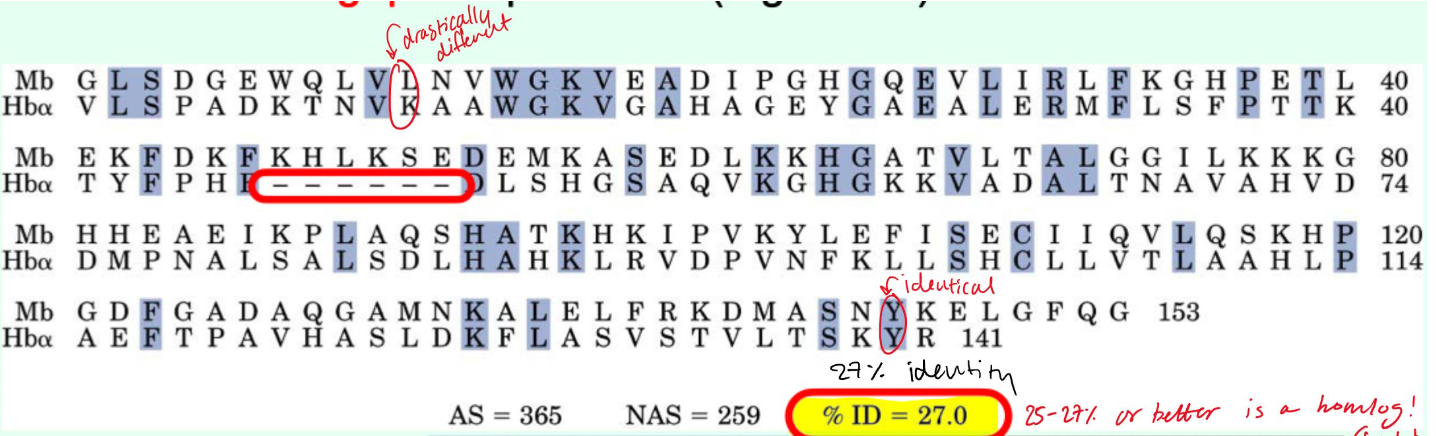
How are gaps treated in sequence alignments?
They are penalized
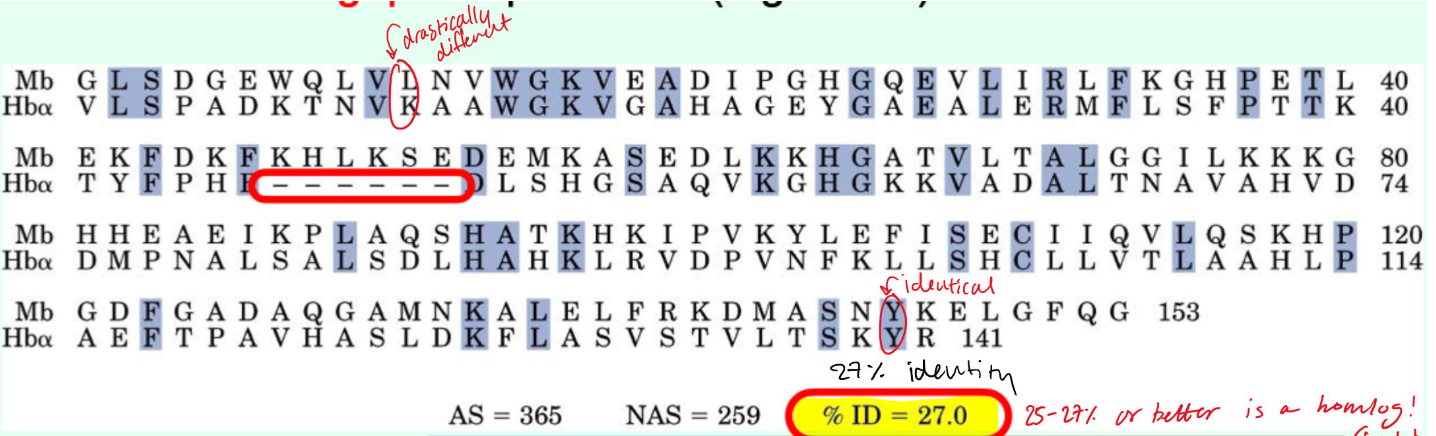
Roughly what percent identity suggest proteins are homologs?
~25-27% identity or higher
What is a cofactor?
A non-protein molecule required for protein function.
What amino acids commonly form ionic interactions?
Negatively charged Asp/Glu (acidic) with positively charge Lys/Arg (basic)
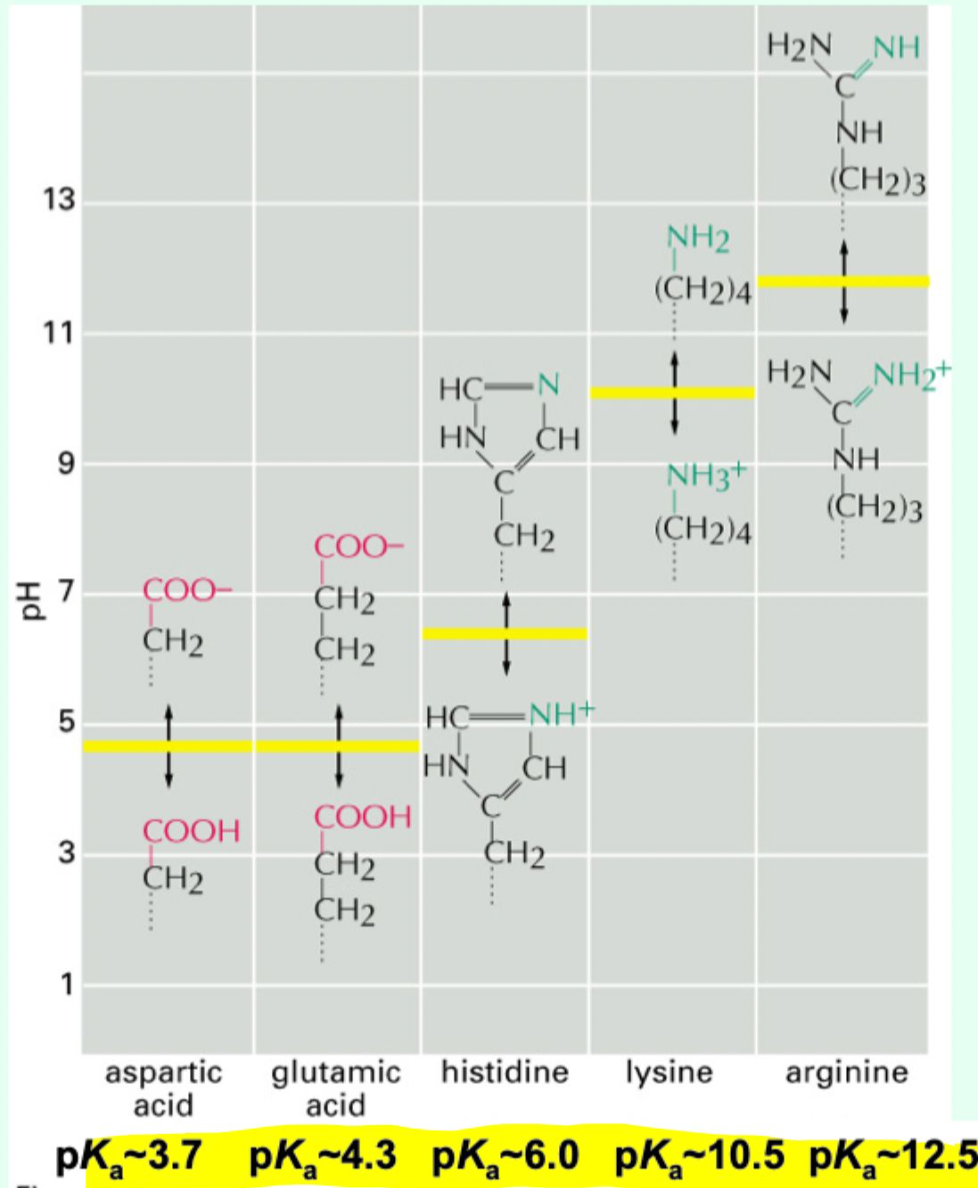
What other parts of protein can participate in ionic interactions?
N- and C- termini and post-translational modifications (glycosylation, phosphorylation)
What is secondary structure in proteins?
The stable, energetically favorable 3D conformations that a polypeptide adopts as building blocks (e.g., α-helices and β-sheets).
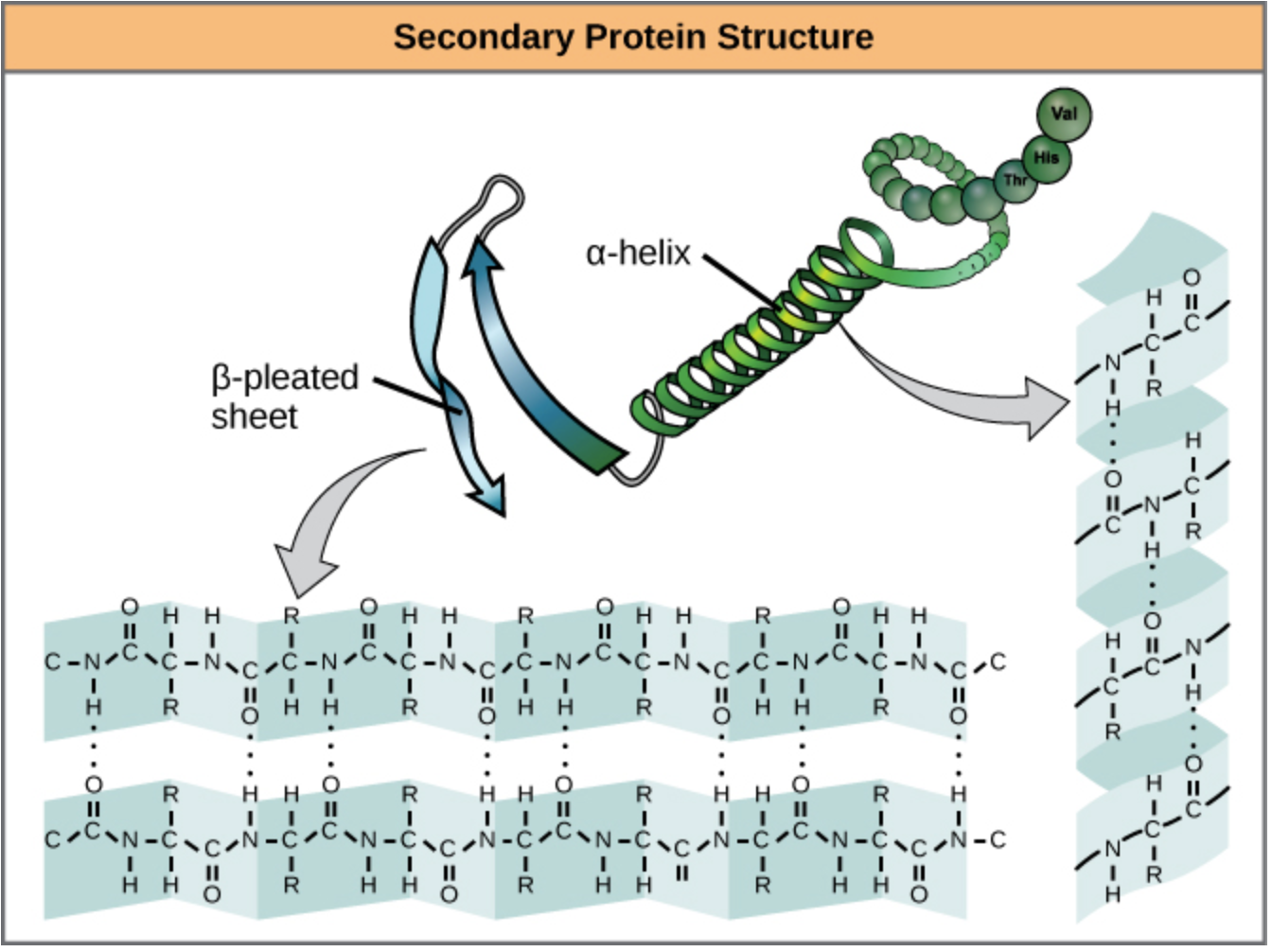
What are the two principle contributors that constrain protein secondary structure?
The peptide bond and the amino acid side-chain
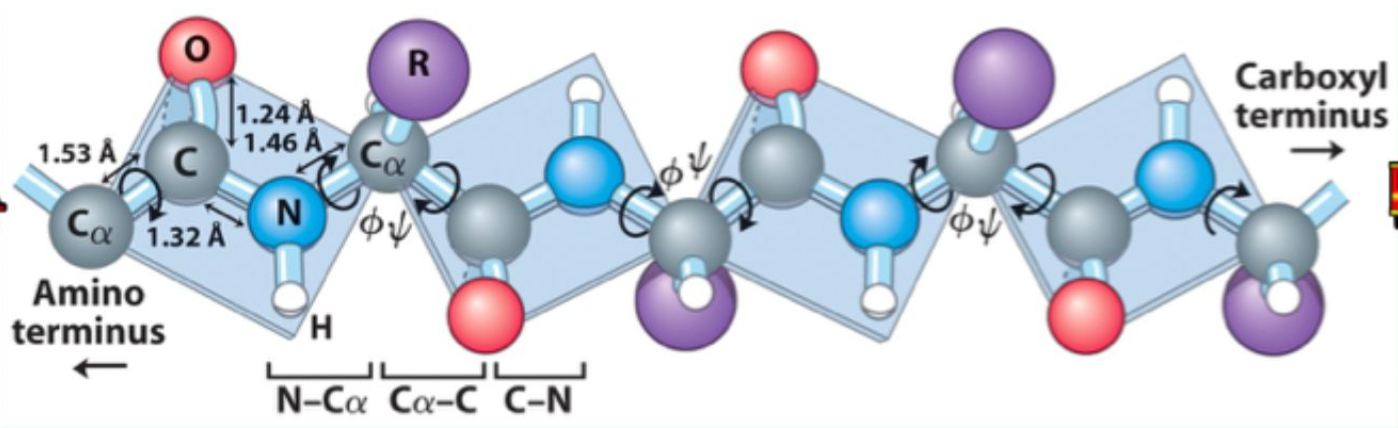
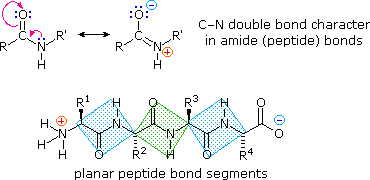
What key property does the peptide bond have?
Partial double-bond character, making it planar.
Why can't the peptide C-N bond rotate freely?
Because electrons are delocalized, giving the bond double-bond character.
Are most peptide bonds cis or trans?
Trans (lower energy, less steric hinderance)
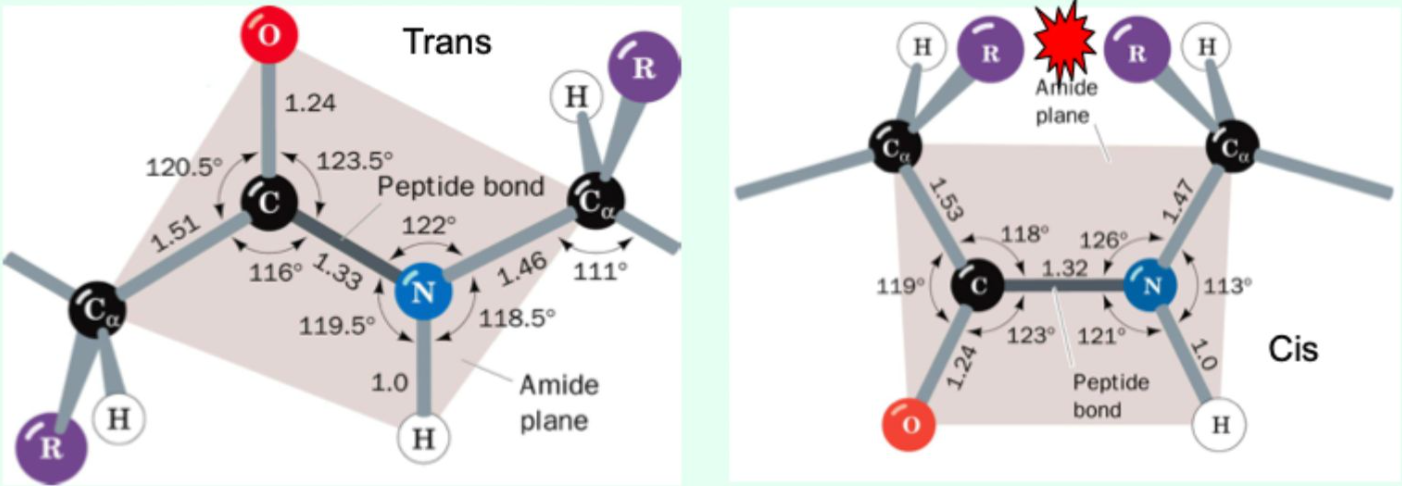
Which amino acid commonly allows cis peptide bonds?
Proline (<10% of Pro are in cis configuration)
What backbone bonds allow rotation?
φ (phi): N–Cα
ψ (psi): Cα–C(=O)
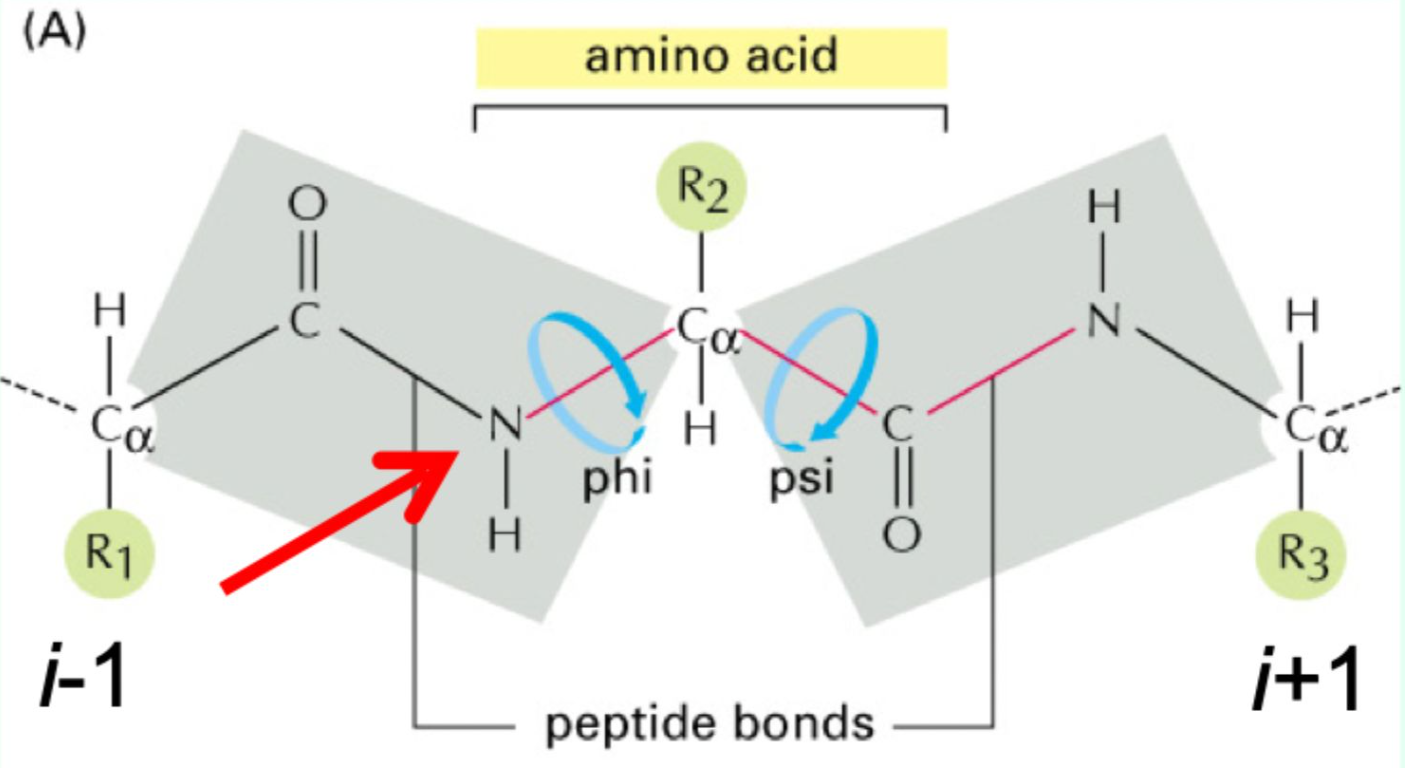
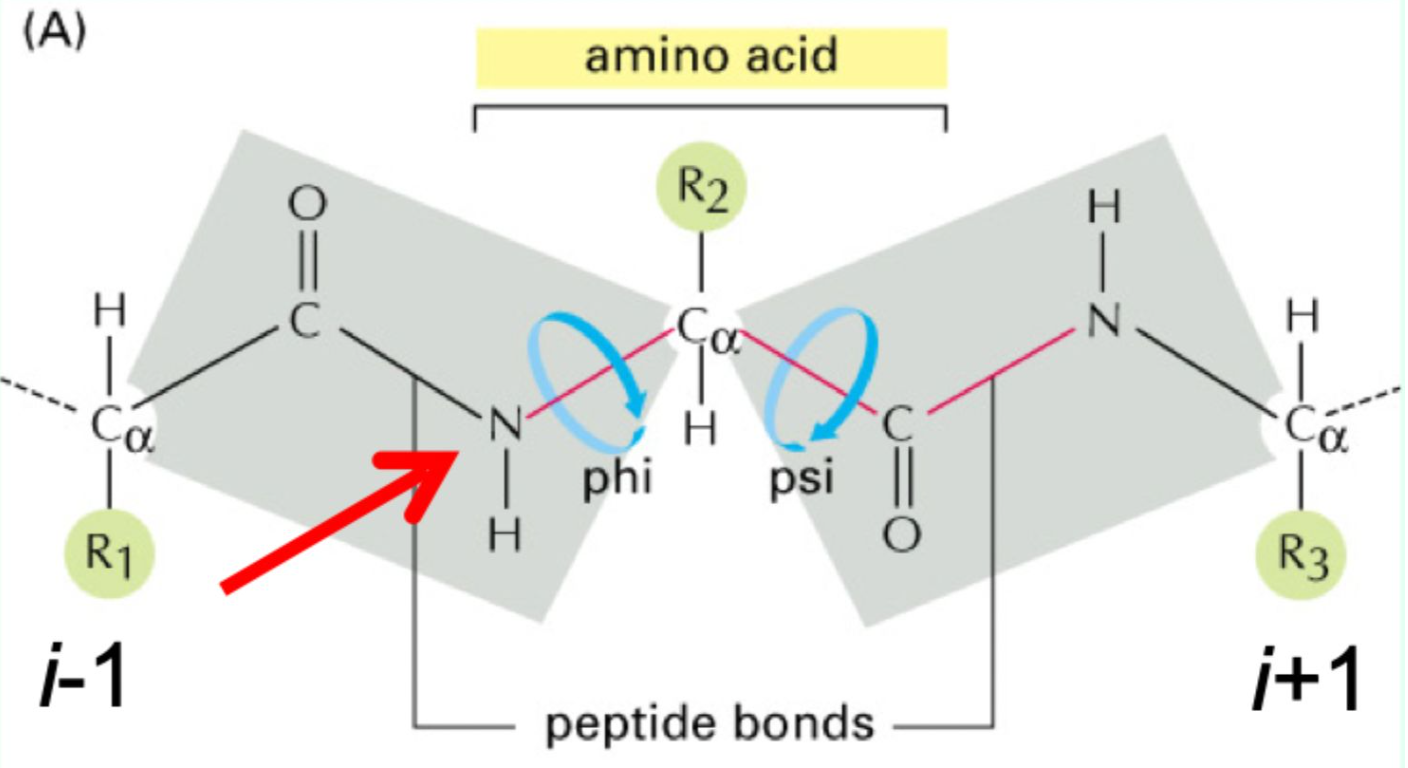
What do φ and ψ angles define?
The 3D conformation of the protein backbone.
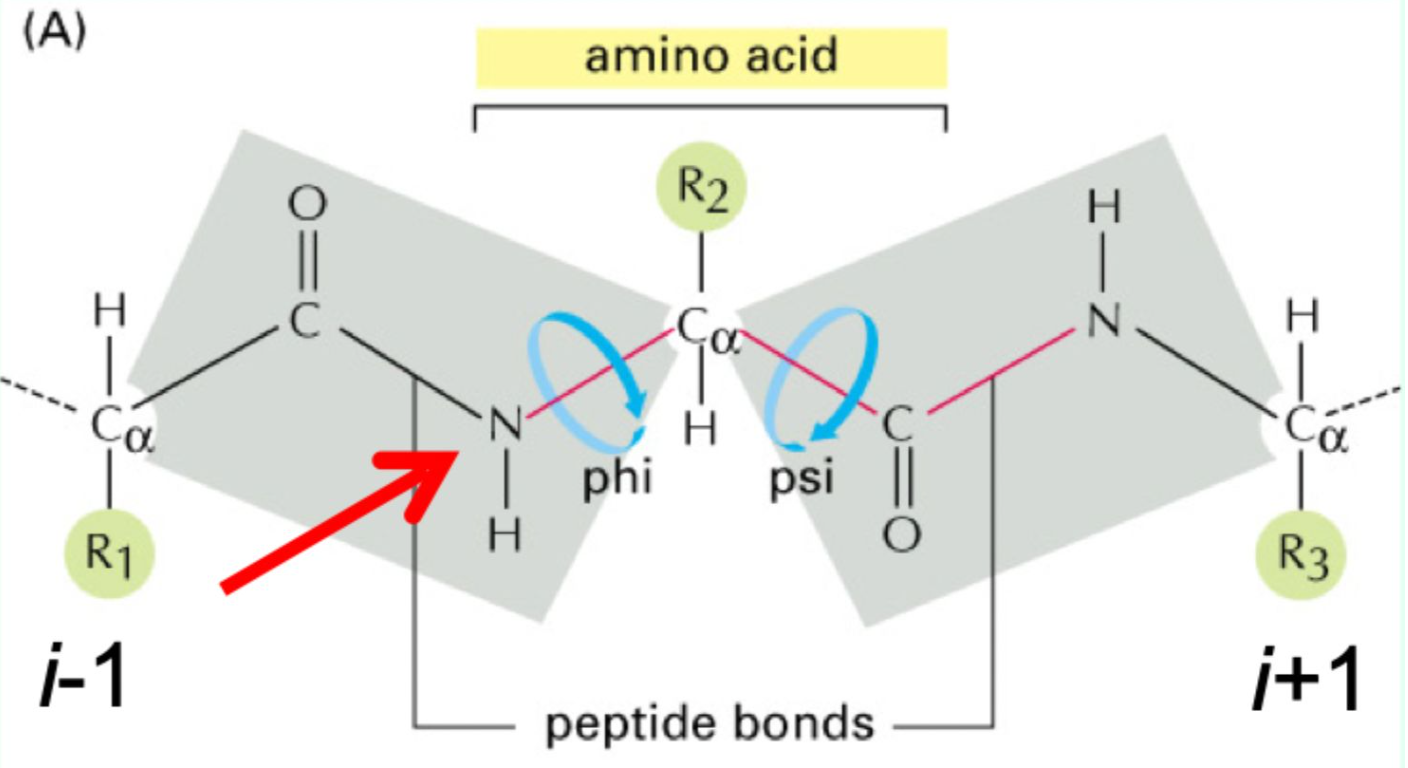
What is the repeating backbone order of atoms and torsion angles in a polypeptides chain?
N → φ (phi)→ Cα → ψ (psi) → C=O → peptide bond, repeated along the chain (Never fi(phi)ght the Alpha ps(i)yChO).
What is Ramachandran plot?
A plot of φ vs. ψ angles showing allowed backbone conformations.
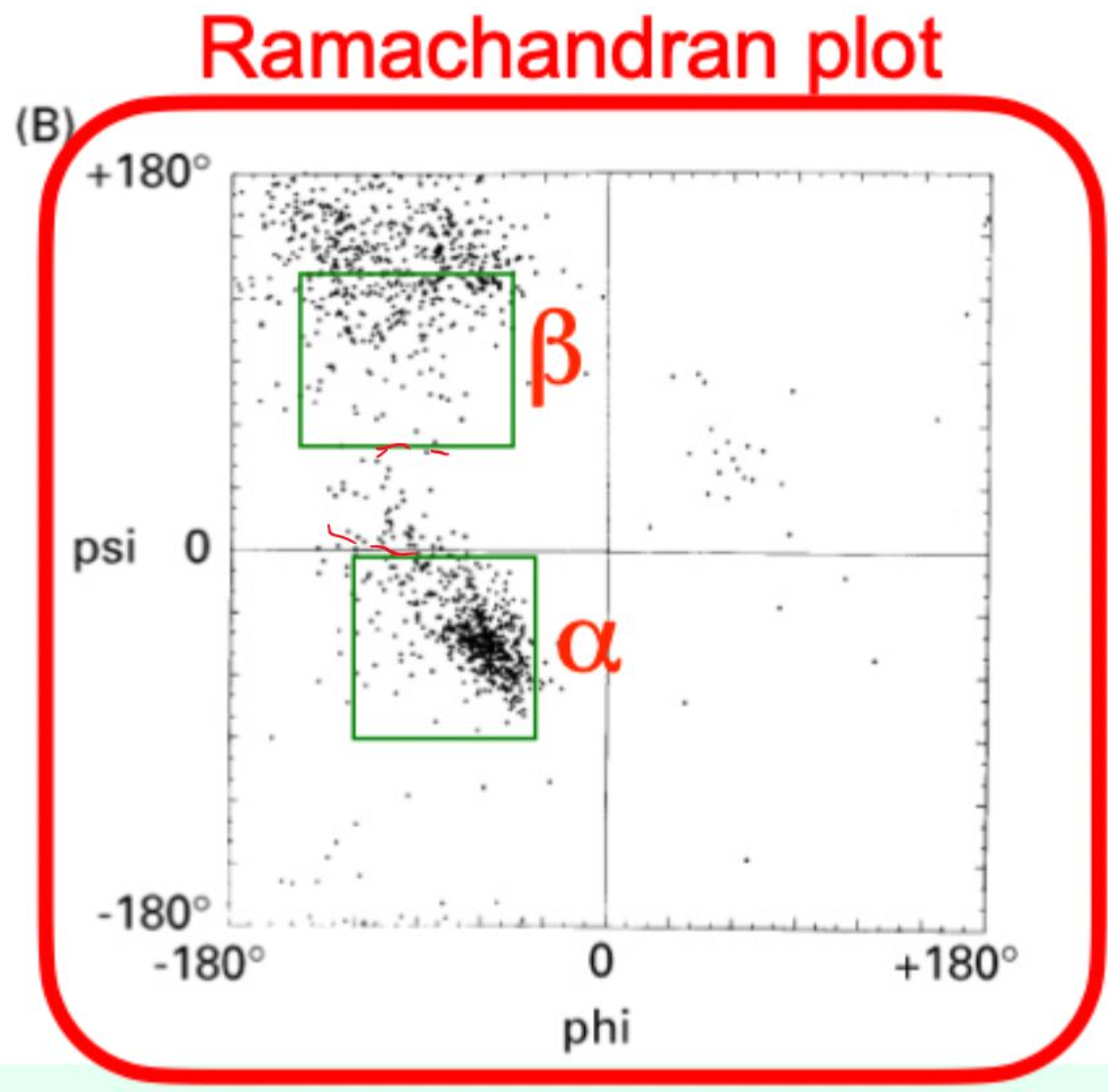
Which amino acid is most restricted on the Ramachandran plot?
Proline (φ ≈ −60° due to side-chain covalent bonding)
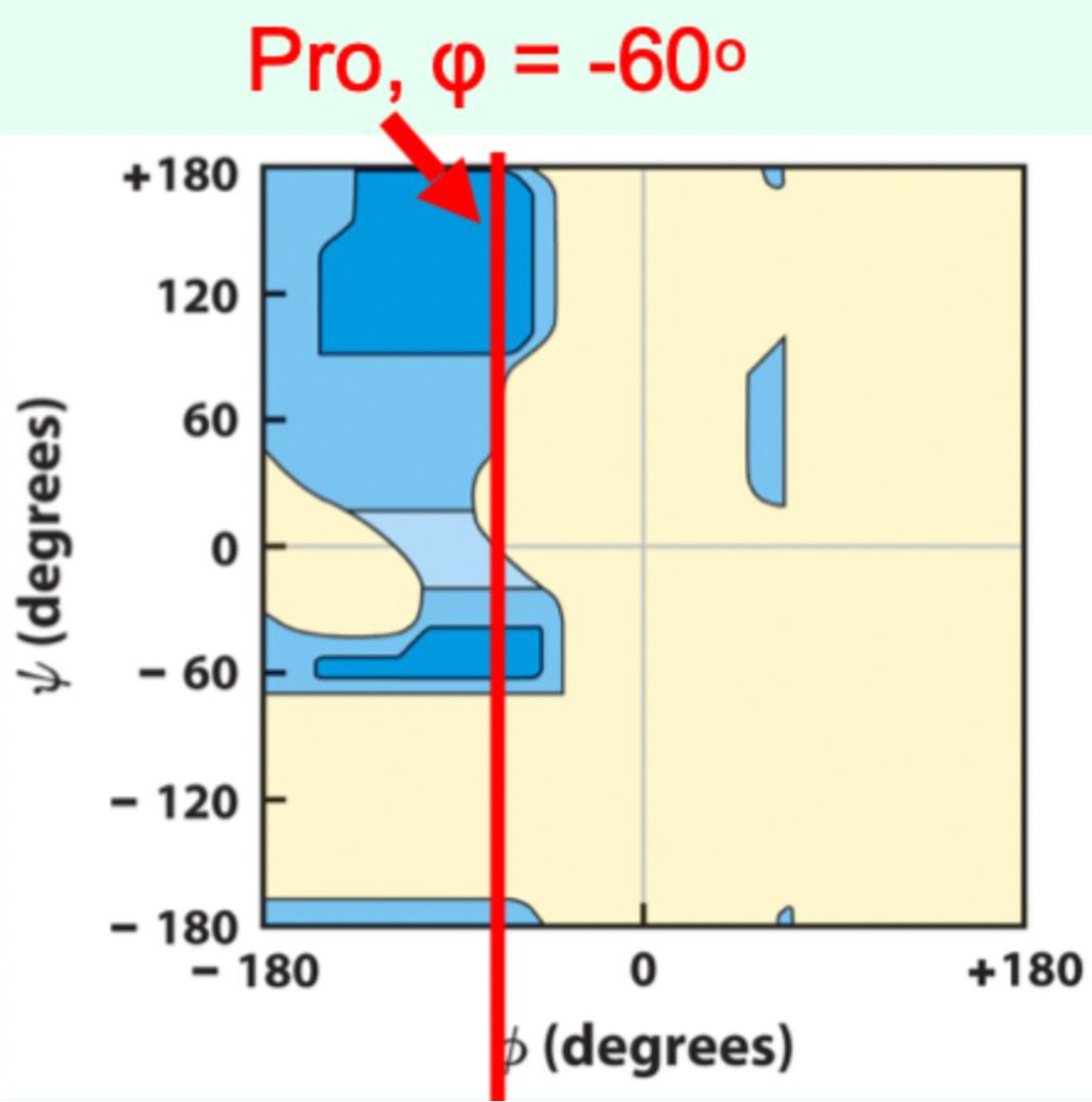
Which amino acid is most flexible on the Ramachandran plot?
Glycine (no side chain)
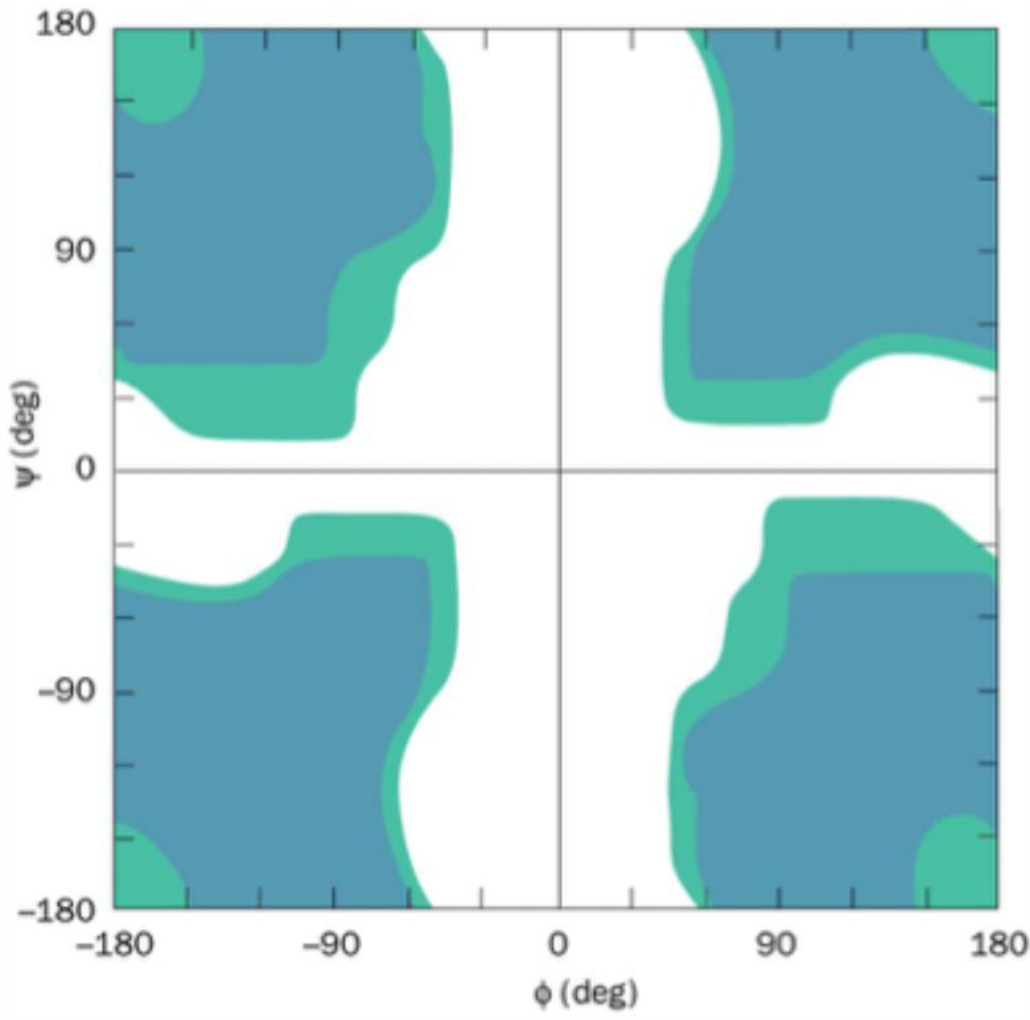
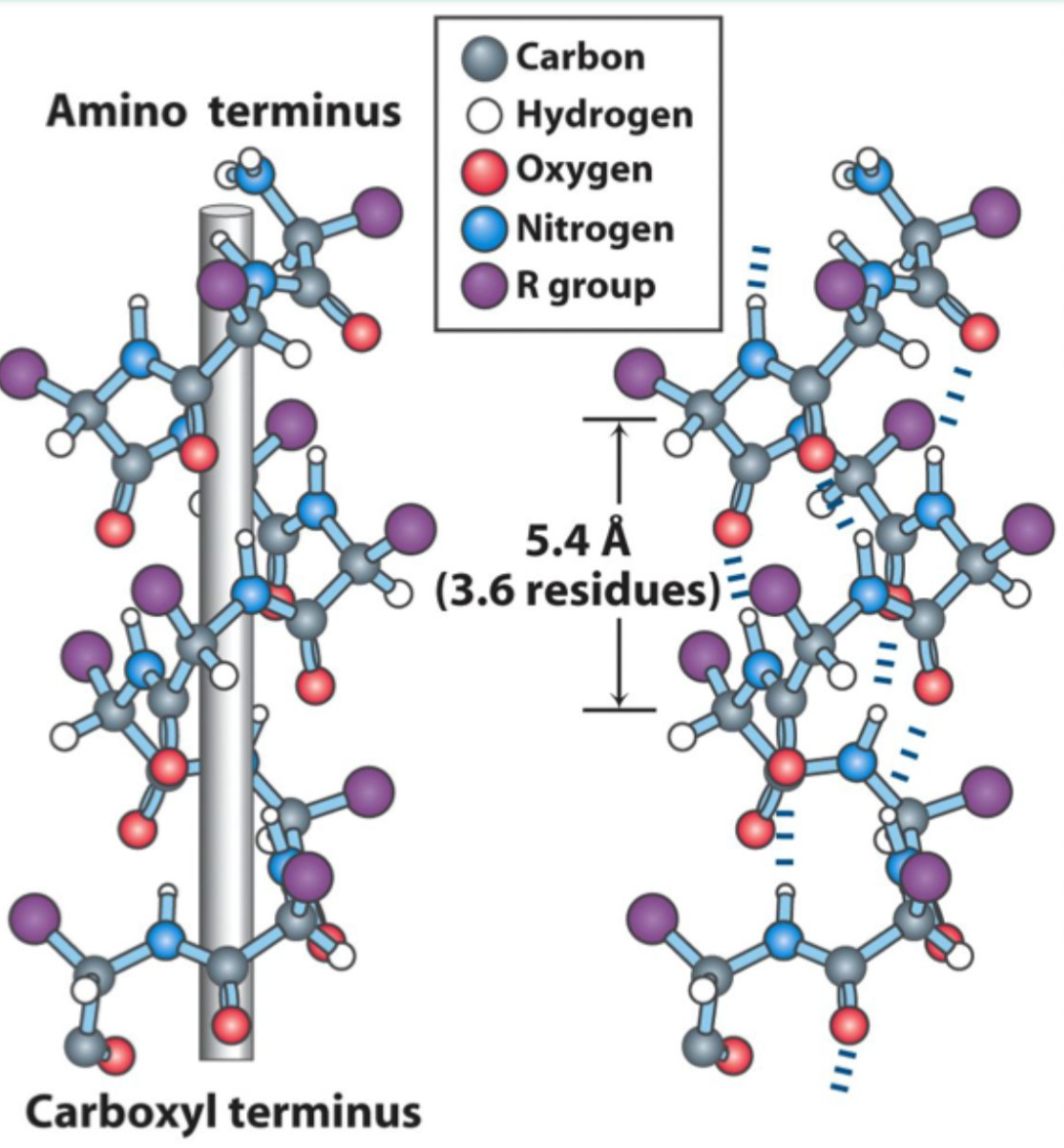
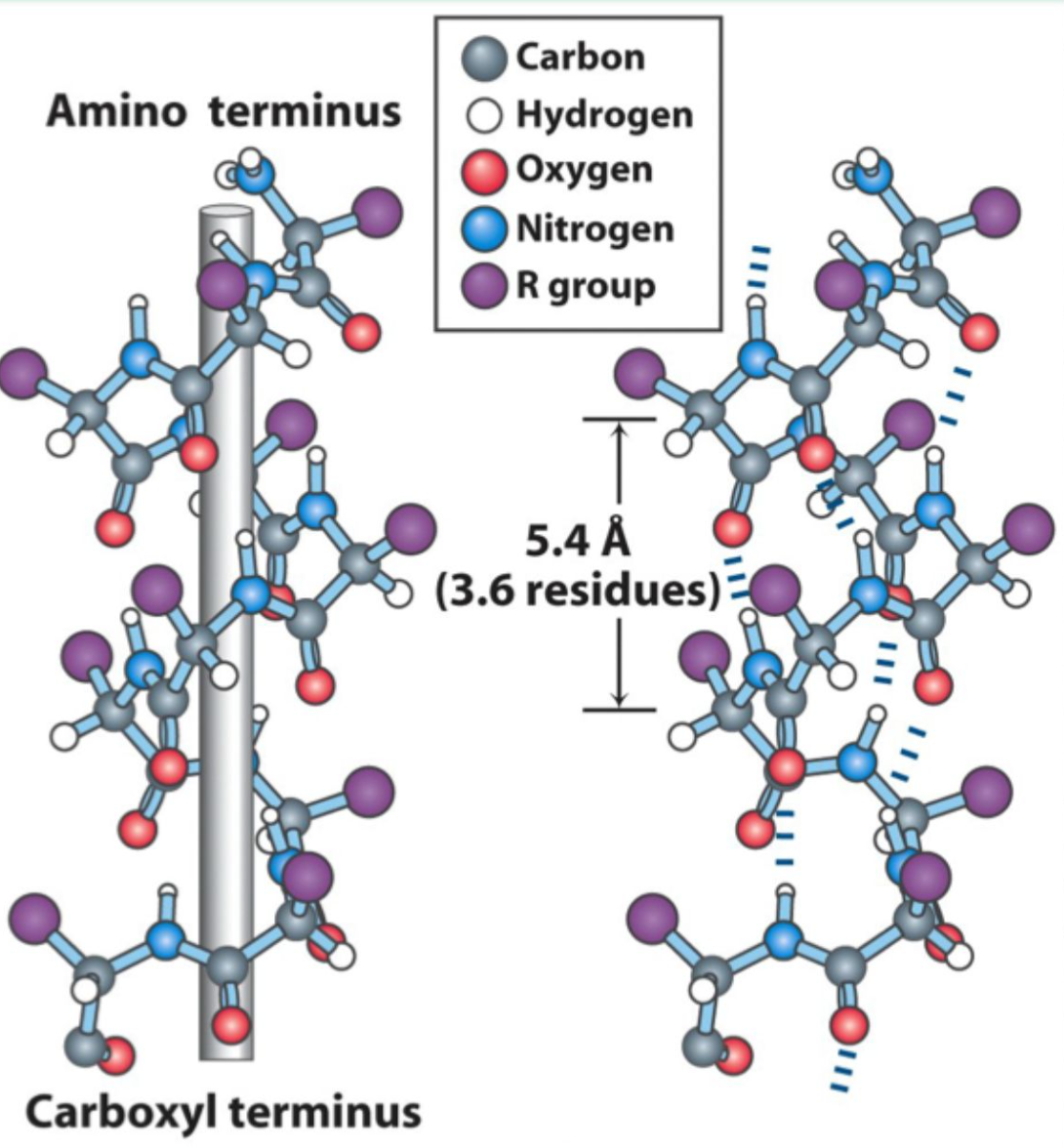
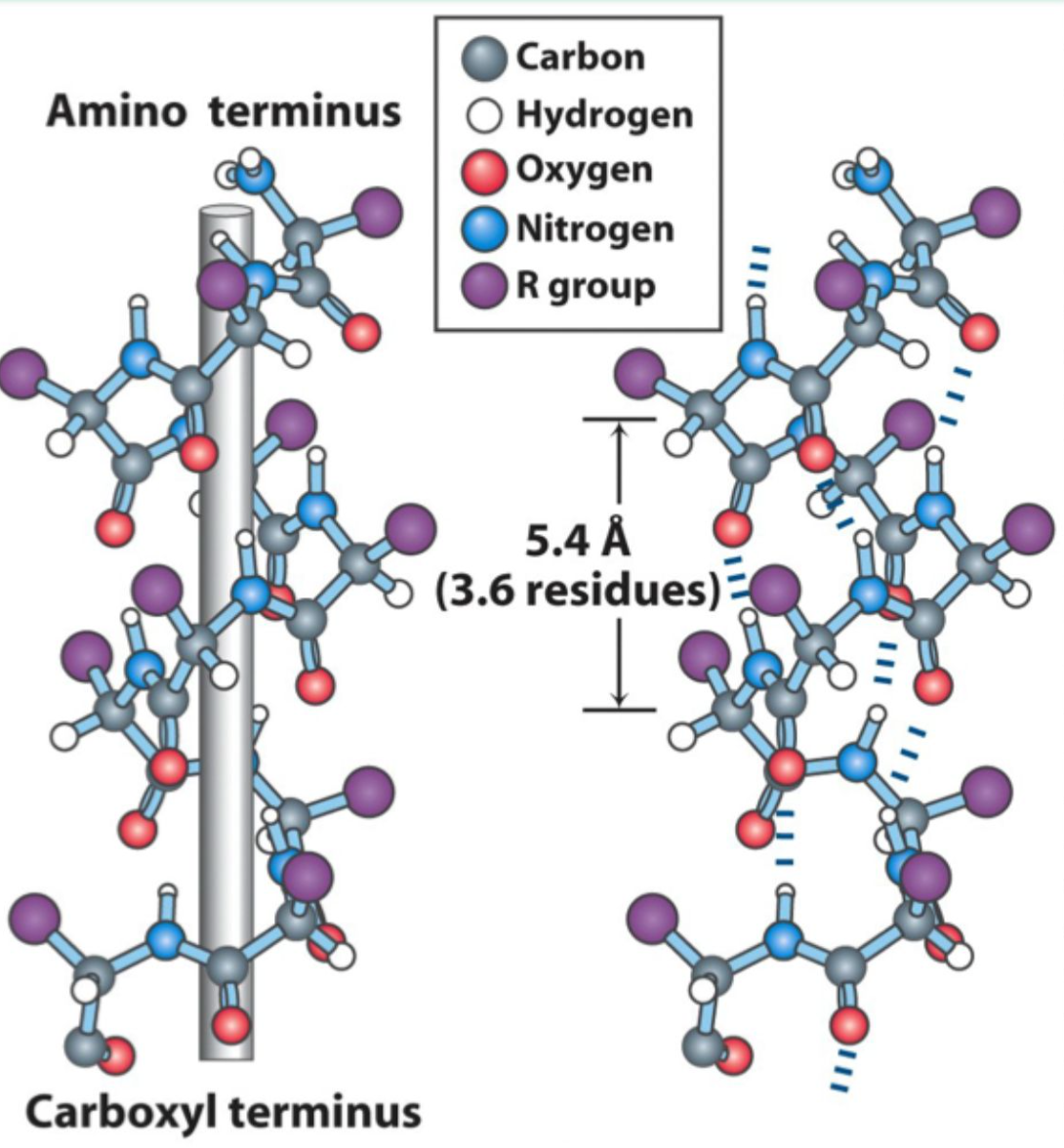
How many residues per turn are in an α-helix?
3.6 residues (5.4 Å) per turn.
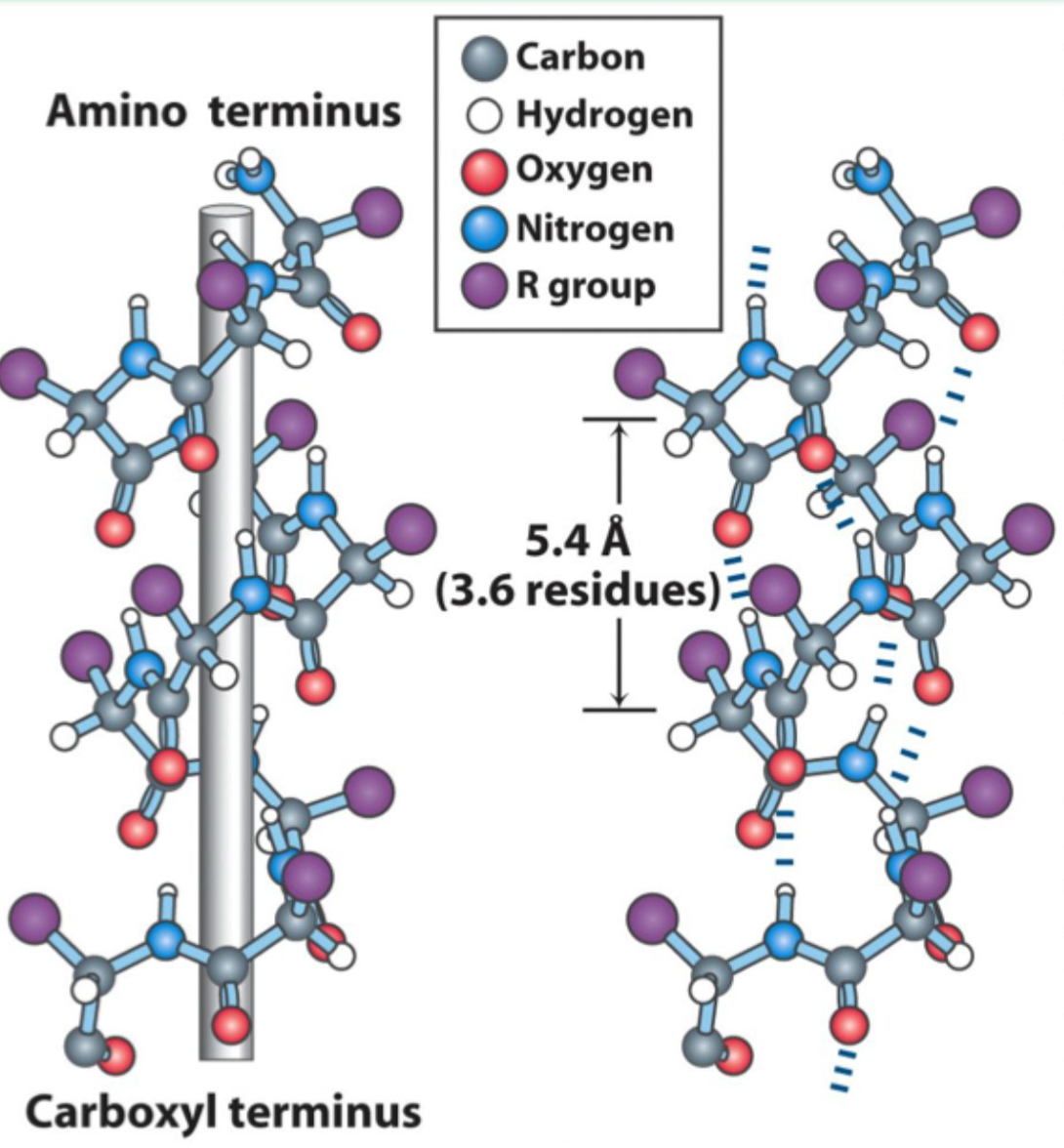
What hydrogen bonding pattern stabilizes an α-helix?
C=O of residue i hydrogen bonds to N–H of residue i+4.
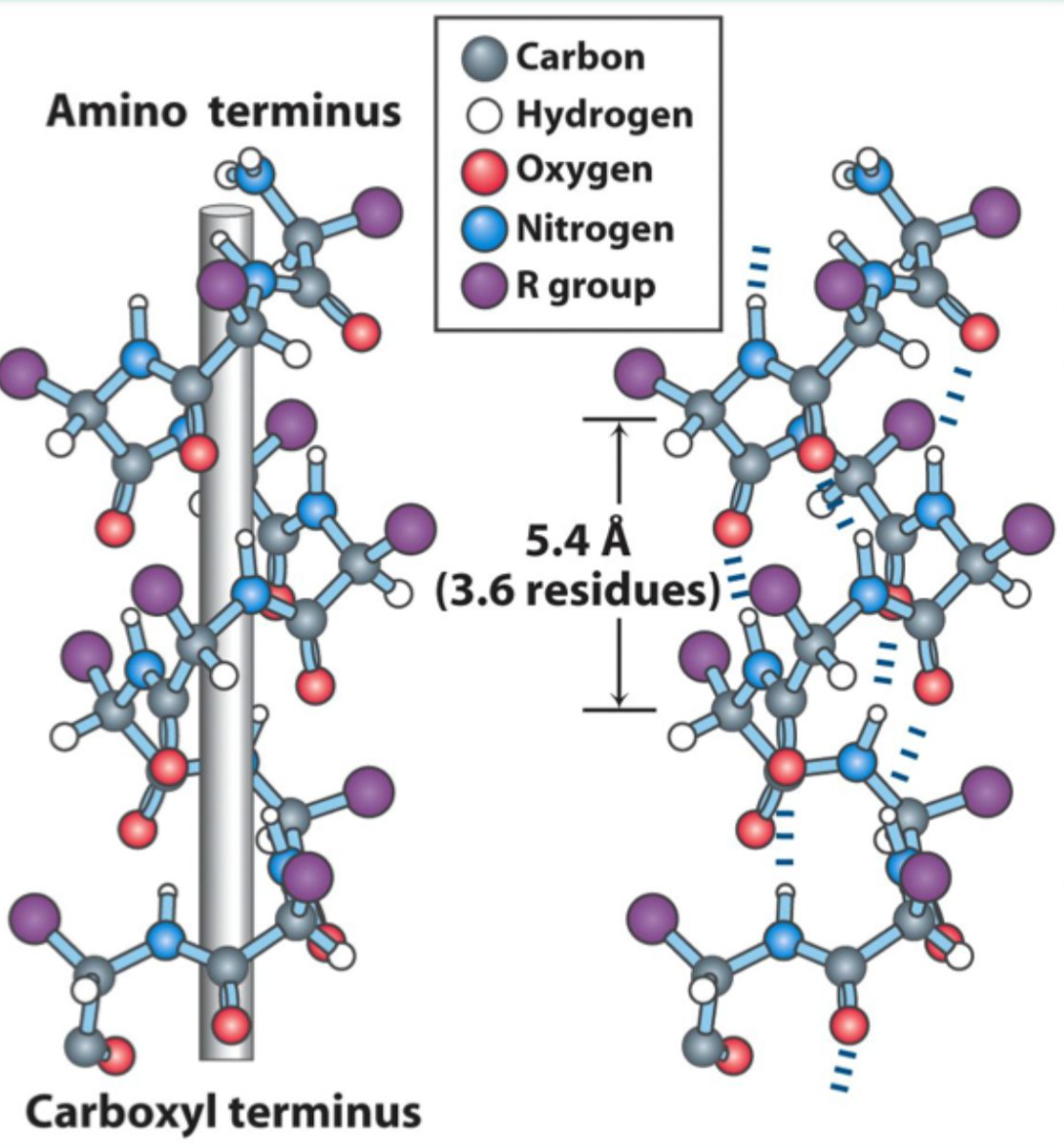
Is the α-helix right-handed or left-handed?
Right-handed
Which amino acids destabilize α-helices?
Proline (kink due to rigid φ = -60°) and Glycine (too flexible)
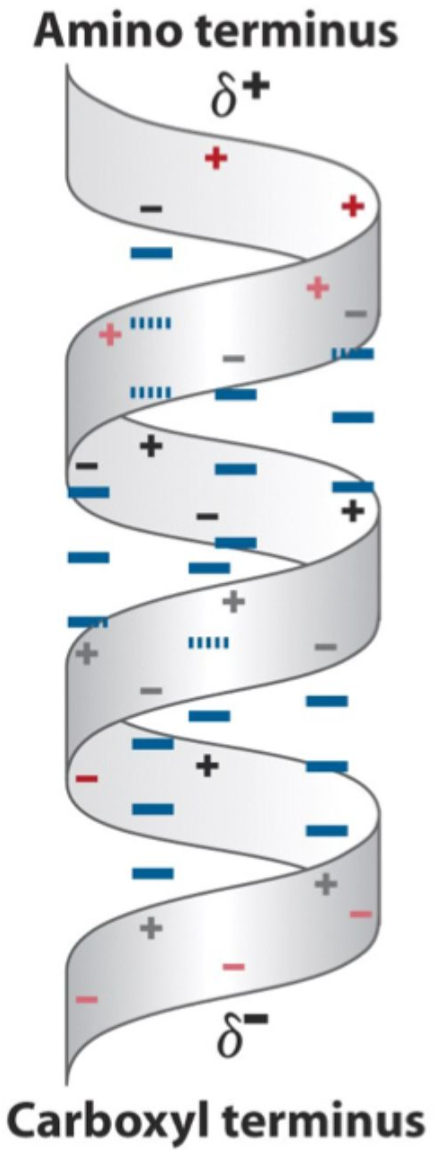
Why does an α-helix have a dipole moment?
All C=O groups point toward the C-terminus and all N–H groups point toward the N-terminus, creating a dipole.
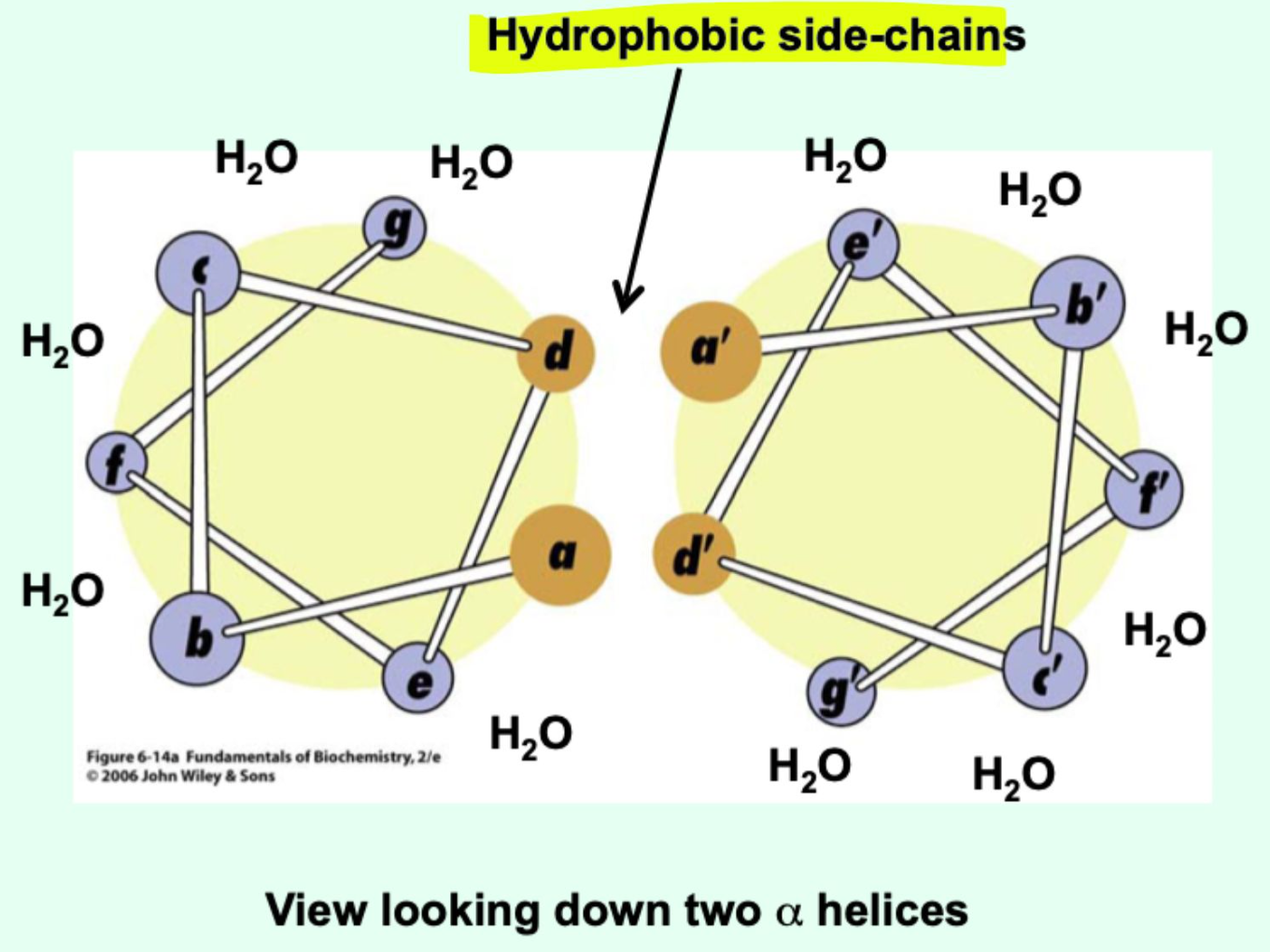
What does “amphipathic α-helix” mean?
An α-helix with one polar (hydrophilic) face and one non-polar (hydrophobic) face.
In a soluble protein, how does an amphipathic α-helix orient?
Polar face interacts with water
Non-polar face interacts with other parts of the protein
In a membrane protein, how does an amphipathic α-helix orient?
Non-polar face interacts with lipid
Polar face interacts with other protein regions
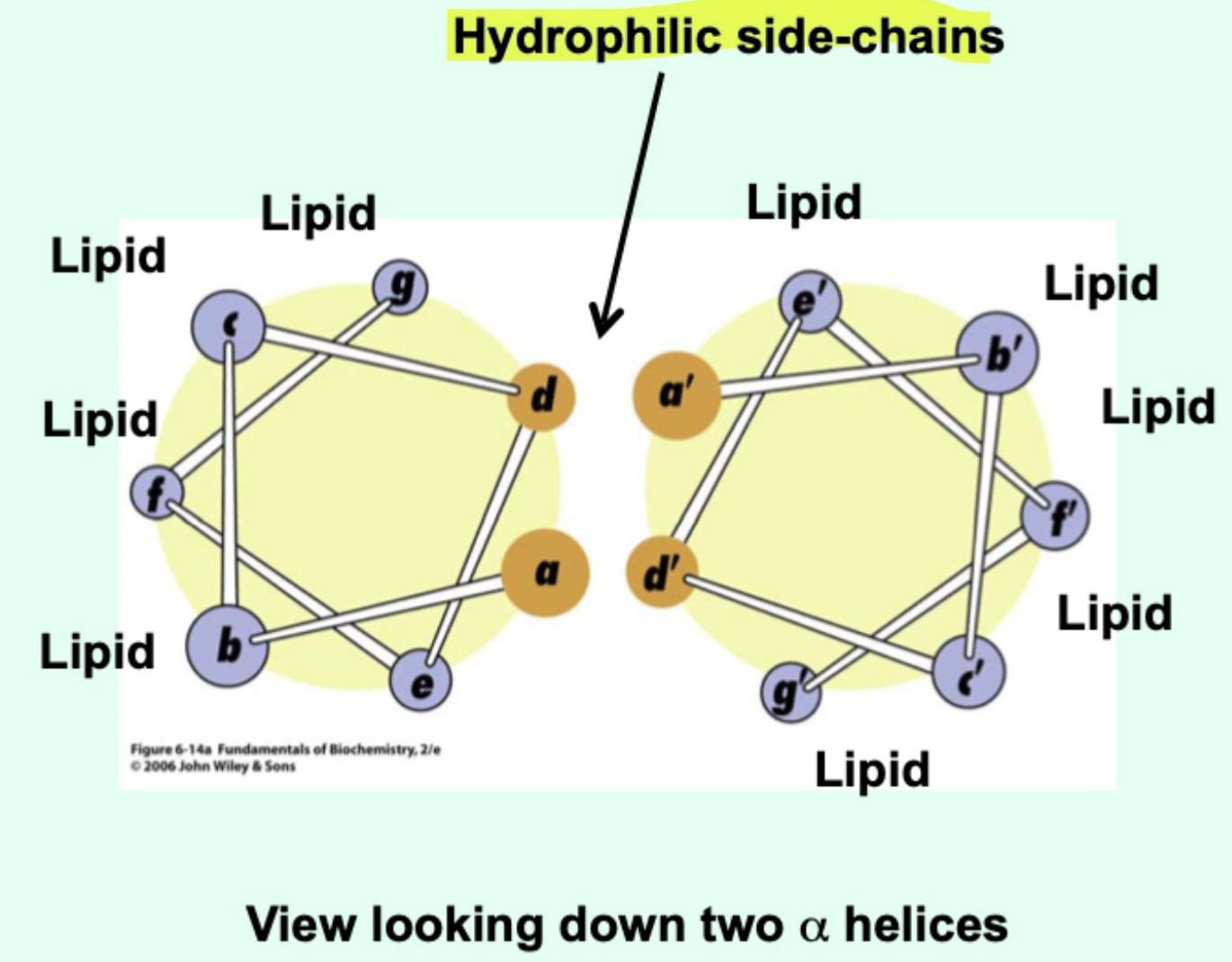
What do amphipathic helices promote?
They promote helix-helix interactions.
Which β-sheet type has stronger hydrogen bonding?
Antiparallel β-sheets (more linear and optimally aligned, so stronger and more stable)
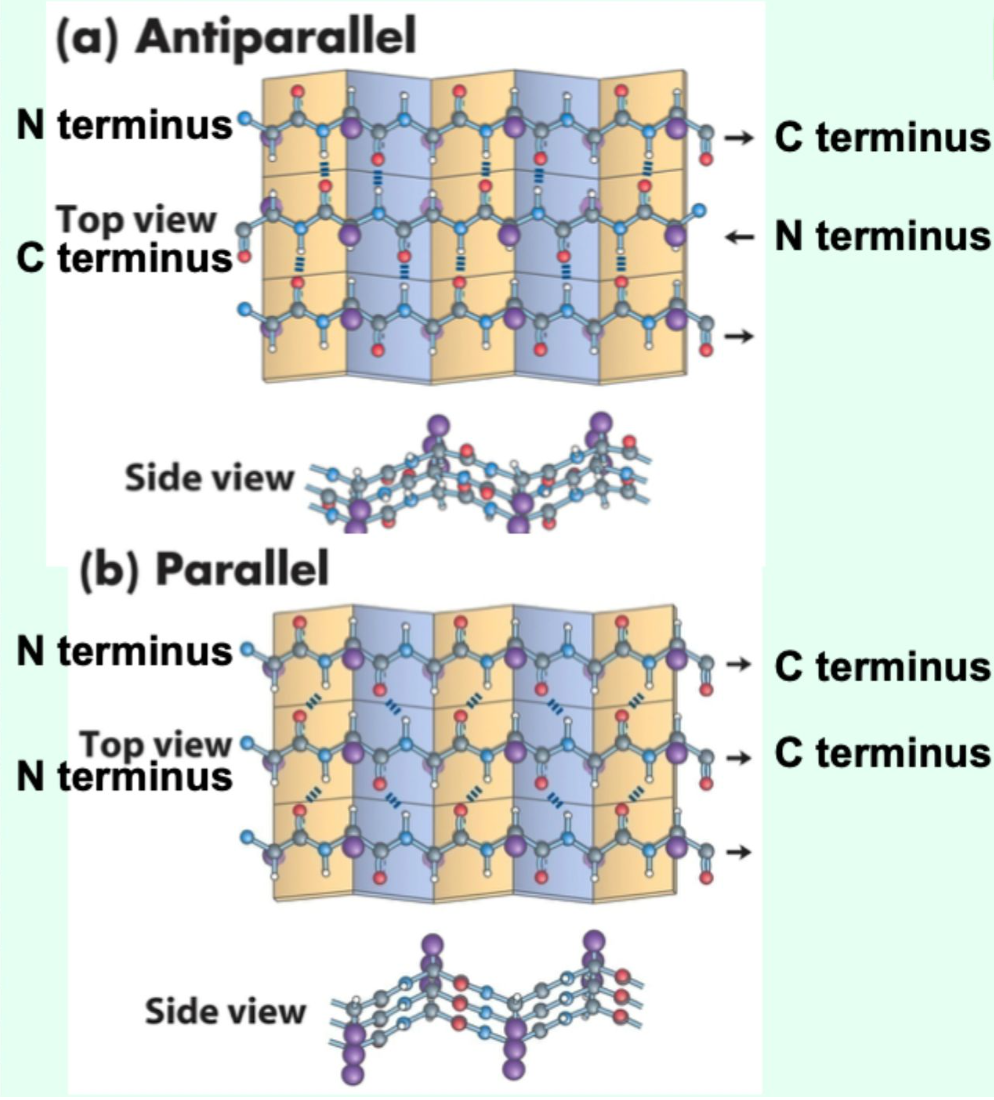
What β-sheet arrangements are possible?
Antiparallel, parallel, or mixed.
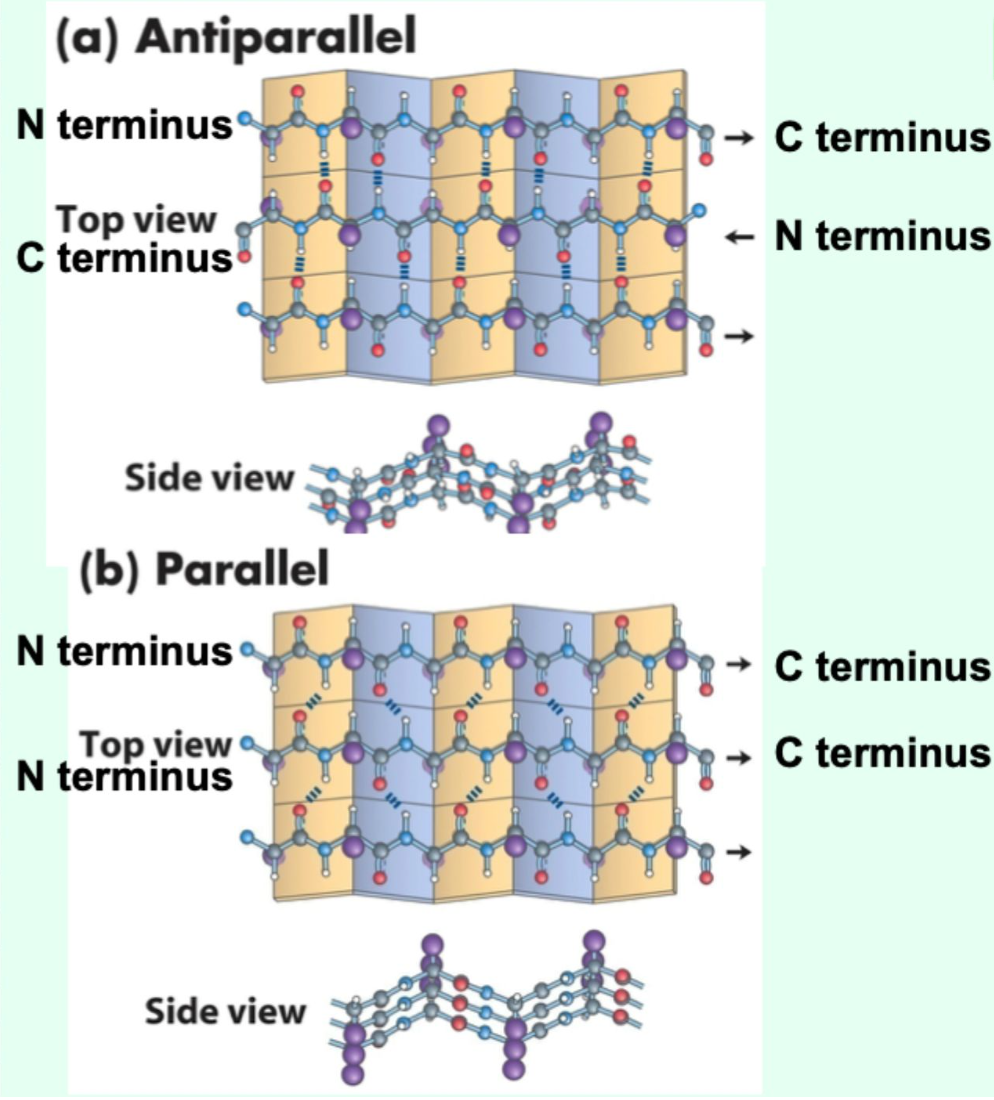
What twist do β-sheets exhibit?
Right-handed twist
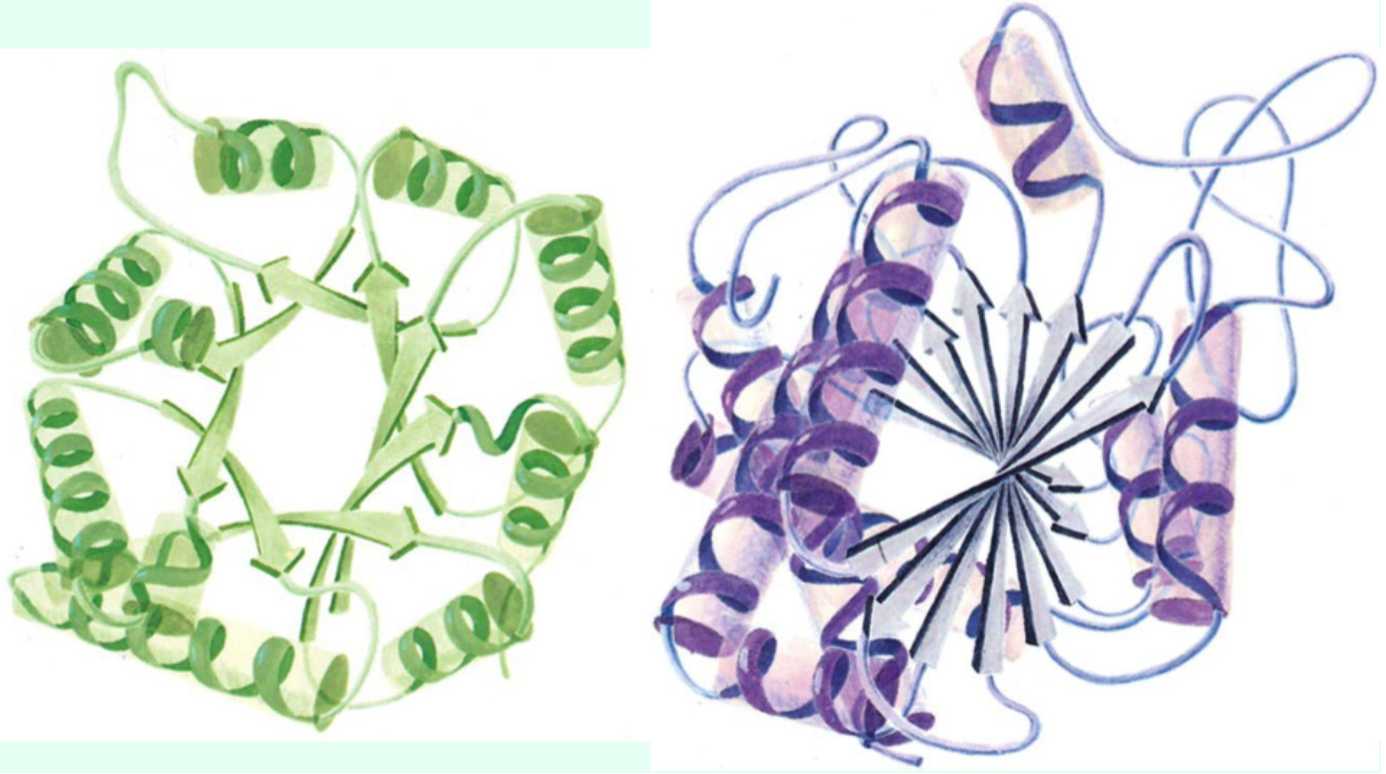
What is the function of turns and loops?
They connect α-helices and β-sheets.
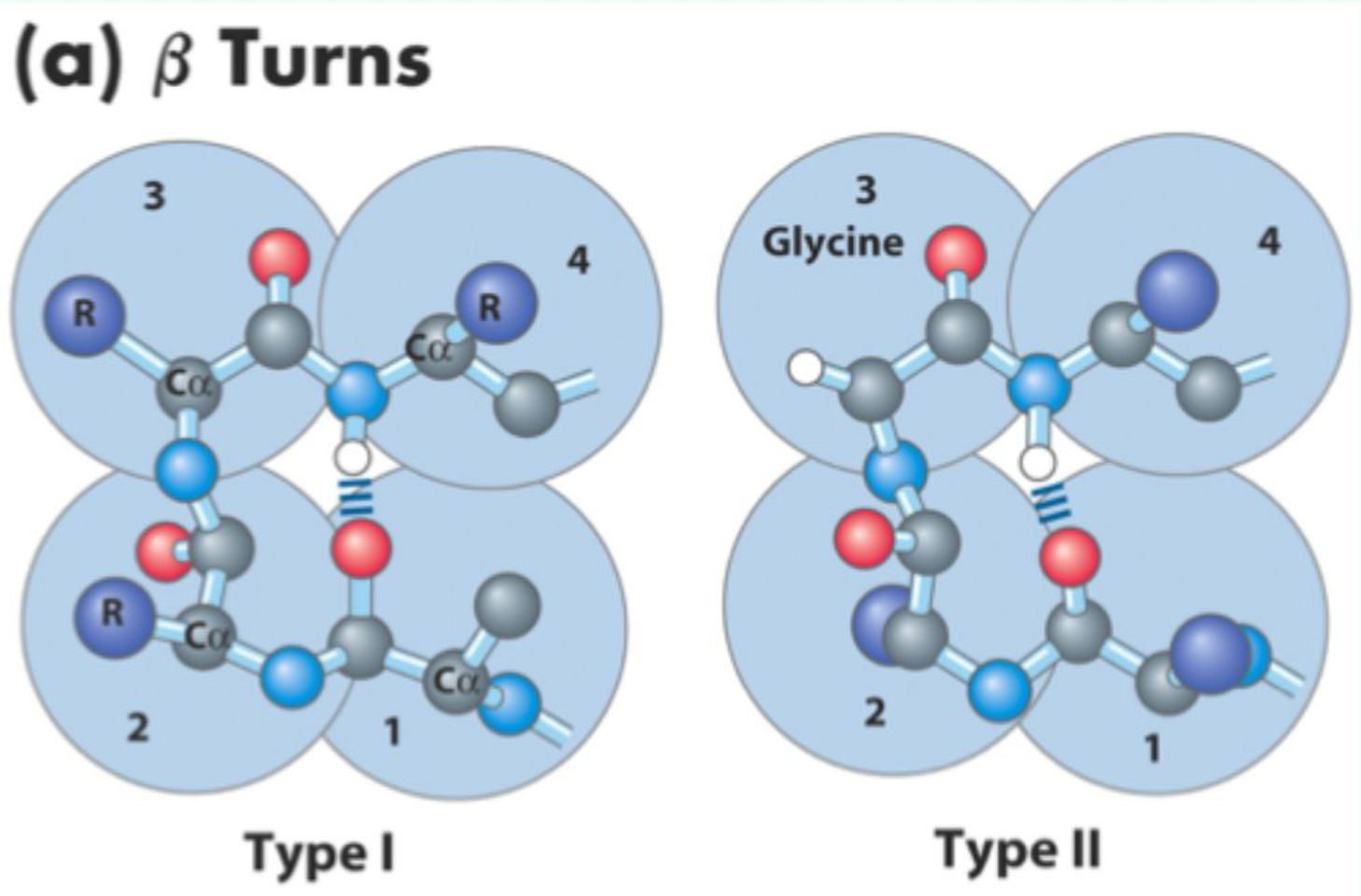
Which amino acids are commonly found in turns?
Glycine and Proline
Which β-turn types should you know?
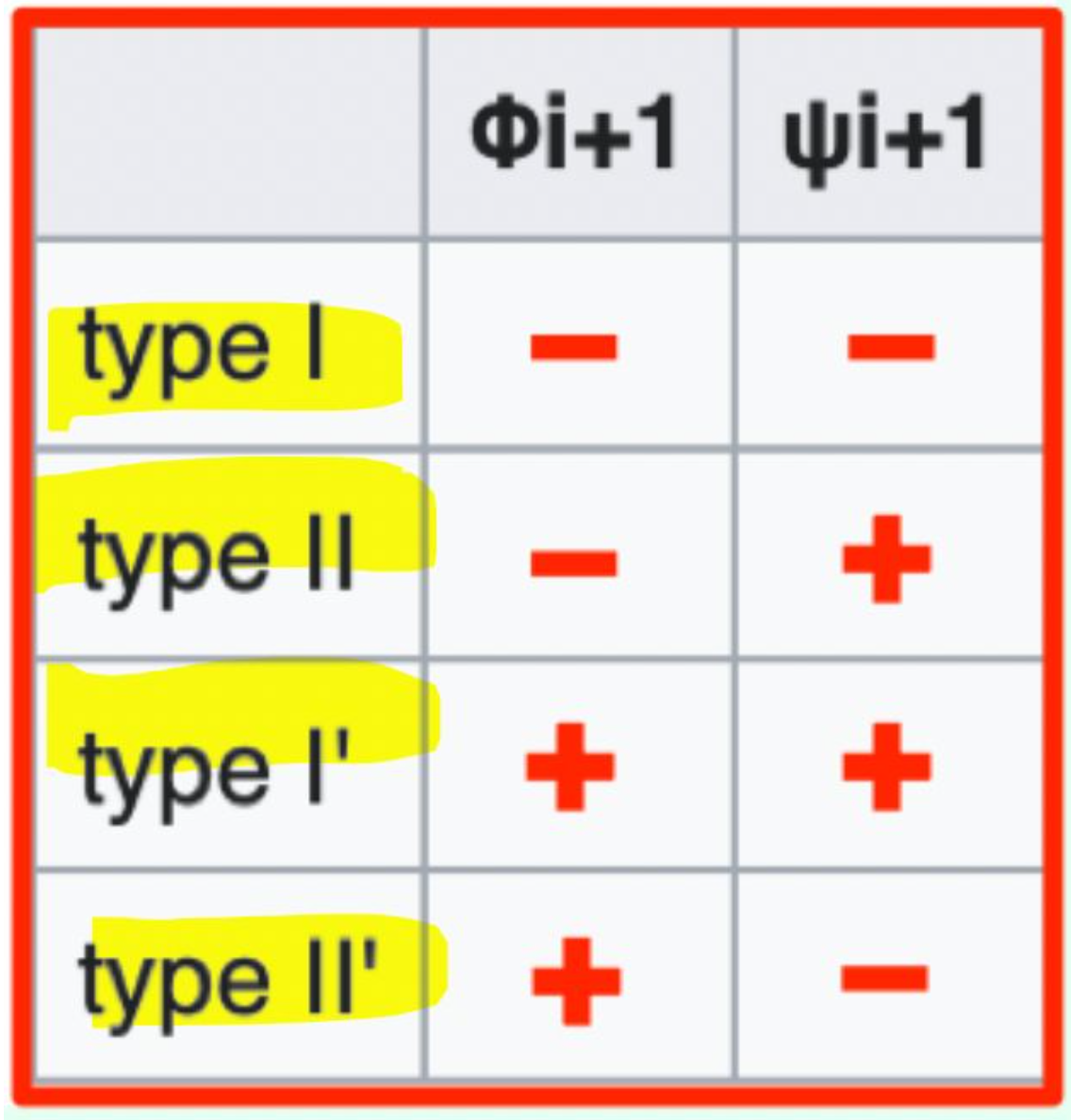
Type I, Type II, Type I', Type II'
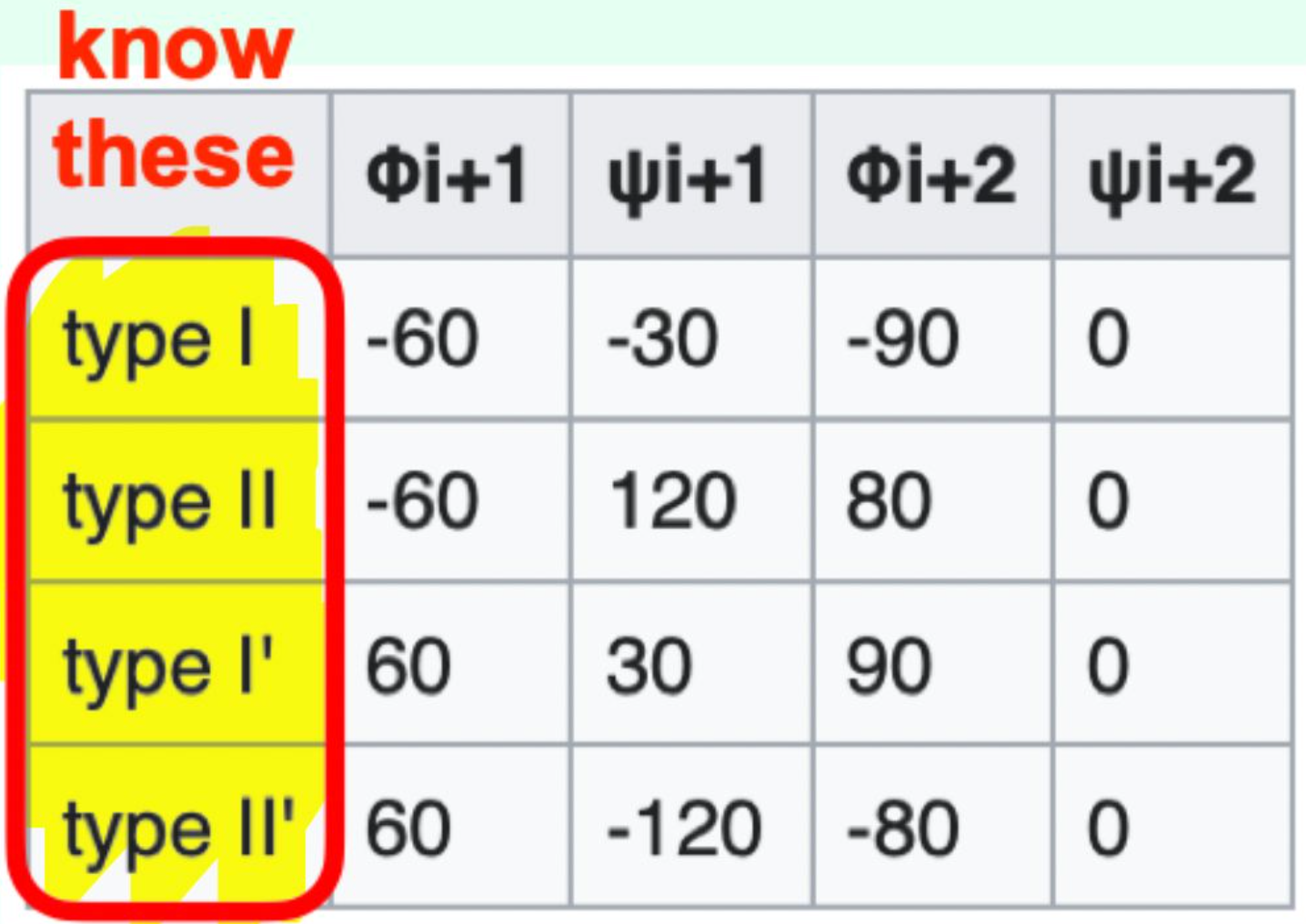
Which β-turn type has φi+1 = (−) and ψi+1 = (−)?
Type I β-turn (− / −)
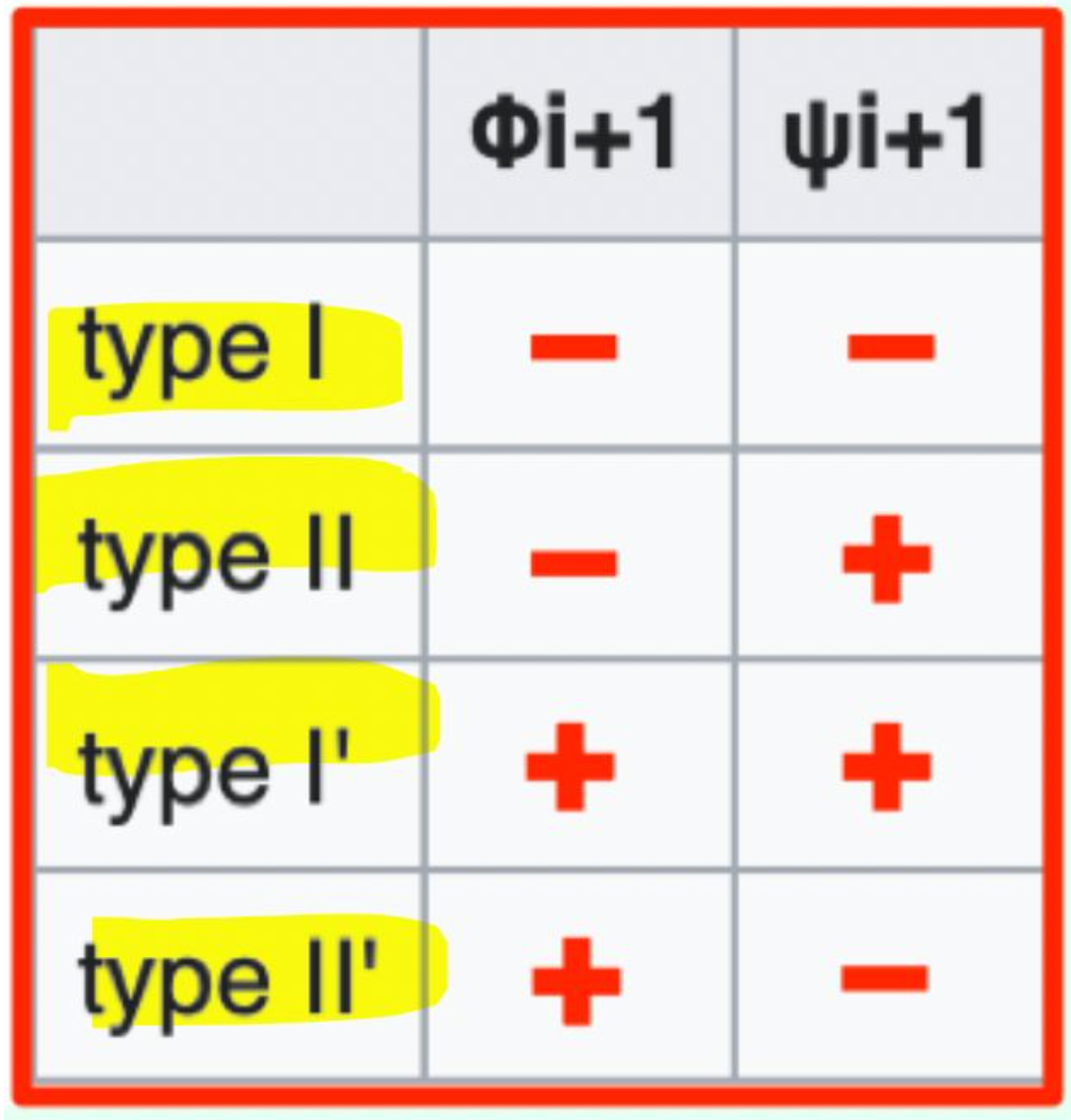
Which β-turn type has φi+1 = (−) and ψi+1 = (+)?
Type II β-turn (− / +)
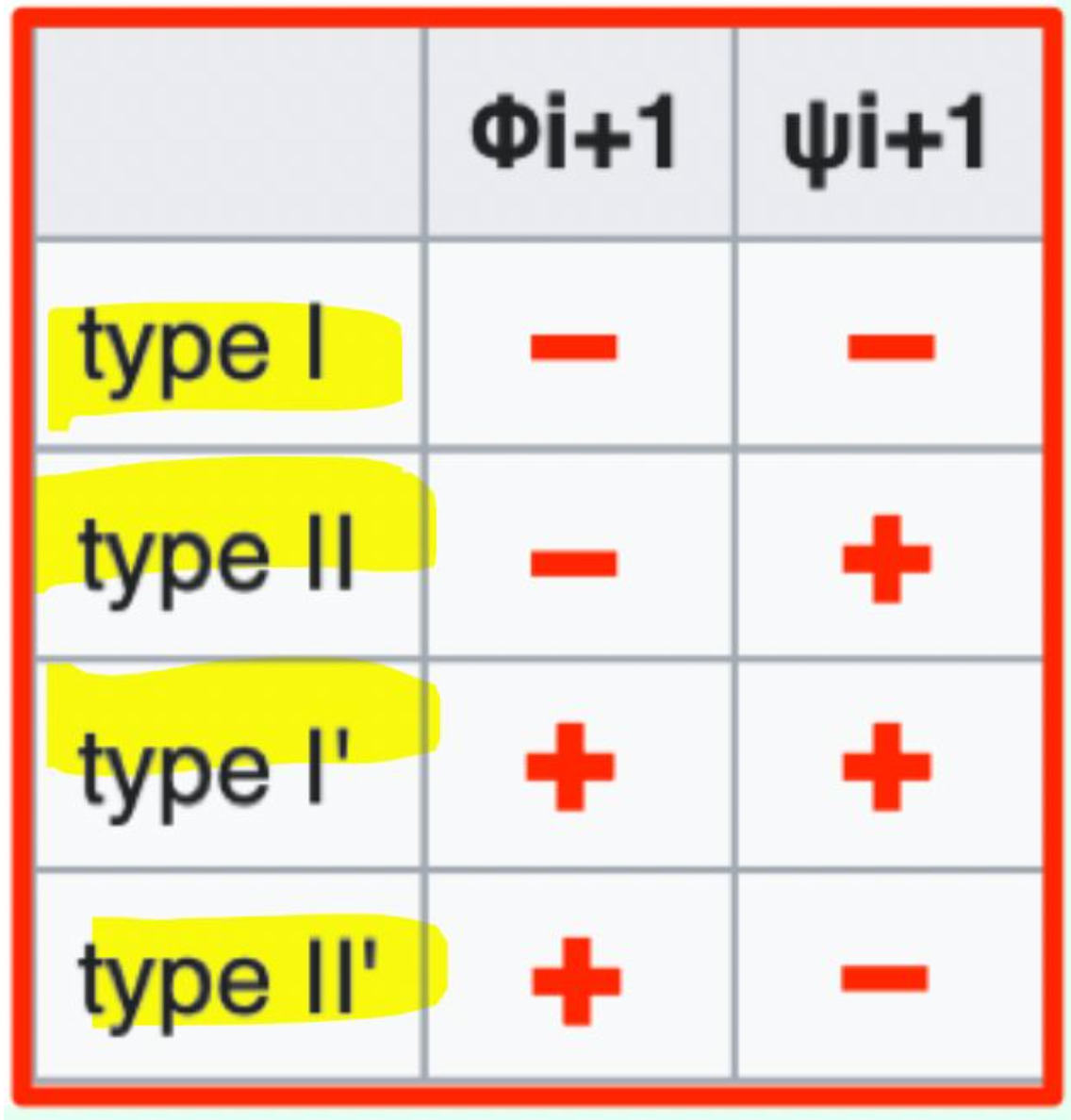
Which β-turn type has φi+1 = (+) and ψi+1 = (+)?
Type I′ β-turn (+ / +)
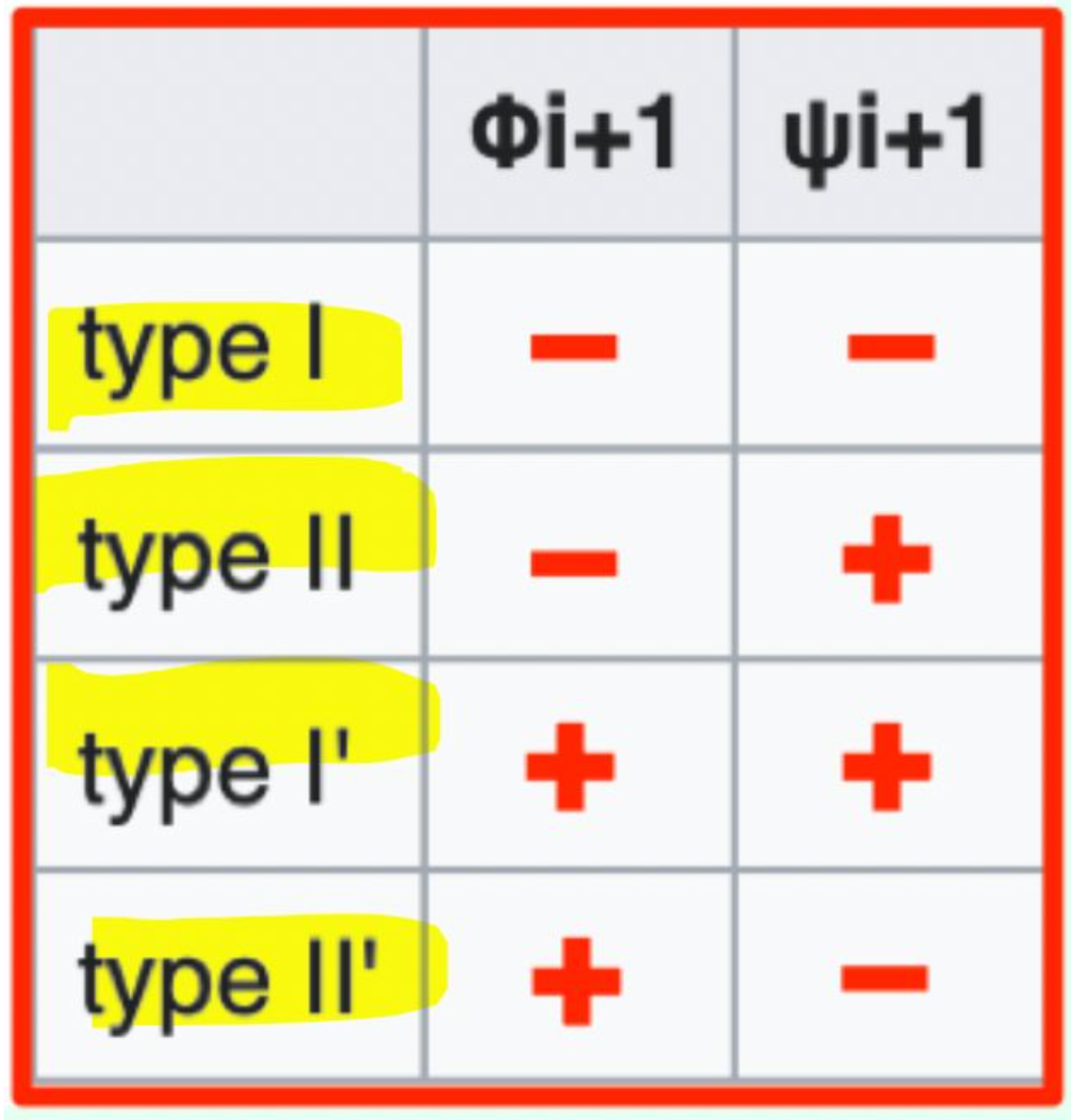
Which β-turn type has φi+1 = (+) and ψi+1 = (-)?
Type II′ β-turn (+ / −)