Advanced studies in GPCRs 1
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
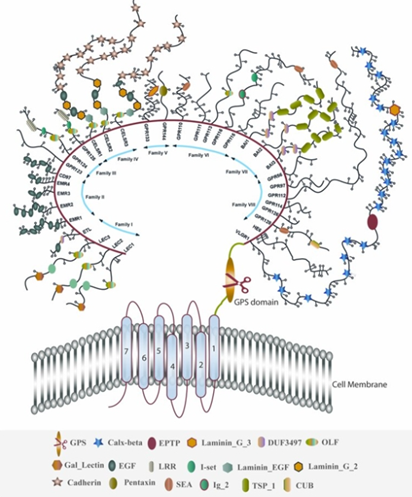
Classification and structure of GPCR families
Organisms like jellyfish, who don’t have nervous systems use monoamines
Previously believed that these organisms didn’t have these molecules
Receptors and their importance
Target sites for drugs
Knowledge of receptor structure and their interaction with ligands are crucial for drug discovery
Helps us develop drugs with higher specificity → we know that GPCRs can activate different signalling pathways and if you favour one over the other then you drive a specific desired (or not desired) effect
Receptors are important components of cell signalling, which is vital for communication between organs for multicellular organisms
Cell signalling requires 2 main components: ligand and receptors, but may involve outher components like secondary messengers and effectors downstream of the signal.
Types of receptors according to structure
Ligand gated ion channels
GPCRs (by far the largest family) → targets of 35-50% of all drugs in the market
Enzymes linked receptors
Intracellular receptors
GPCRs
Approximately 800 in the human genome
For C. Elegans 5-10% of the genome encodes GPCRs
Can be activated by hormones, neurotransmitters, light, AAs, odours, aftty acids, nucleotides etc.
GPCRs are essential for numerous physcioloical processesed and are involved in the regulation of various aspects of human biology
Their ubiquity and importance make them prime targets for pharmecutical targeting
General characteristics of GPCRs
Many families but all share these features
Single polypeptide chain
7 TM domains
3 EC lops and 3 IC loops
Extracellular N terminal and IC C terminal domain
All interact with G proteins and arrestins
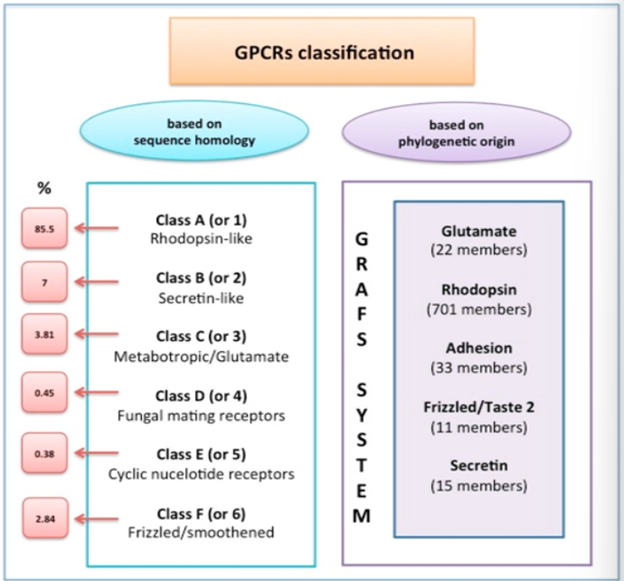
Classification of GPCRs
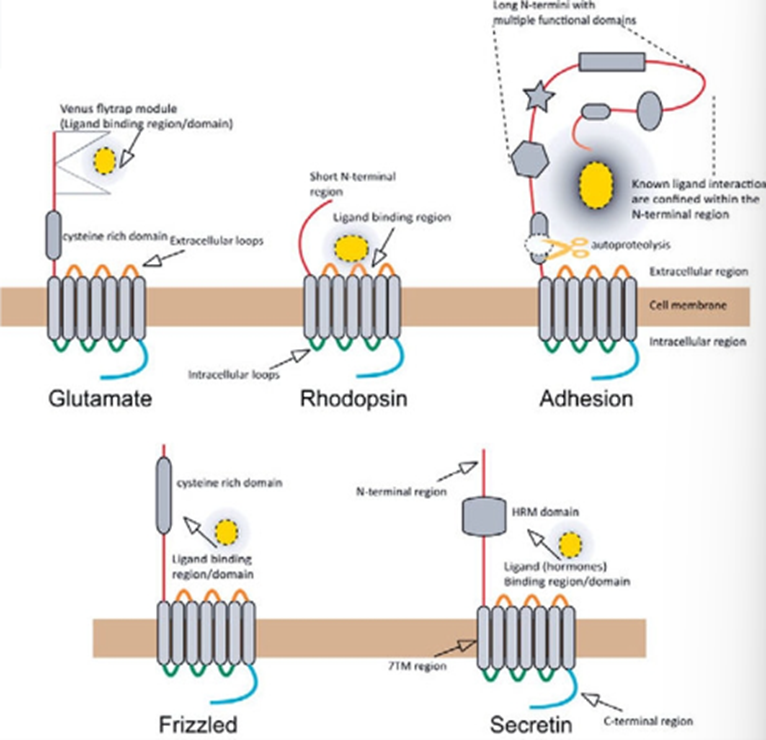
Can be classified by the name of the family, called the GRAFS system, which creates distinct classes of: Glutamate, Rhodopsin, Adhesion, Frizzled/Taste2 and Secretin
Or can be classified based on sequence homology which categorises receptors into classes A, B, C, D, E and F and is used by the International Union of Pharmacology.
This A, B, C etc. nomenclature was later replaced by the 1-6 classification system.
GRAFS system - Glutamate
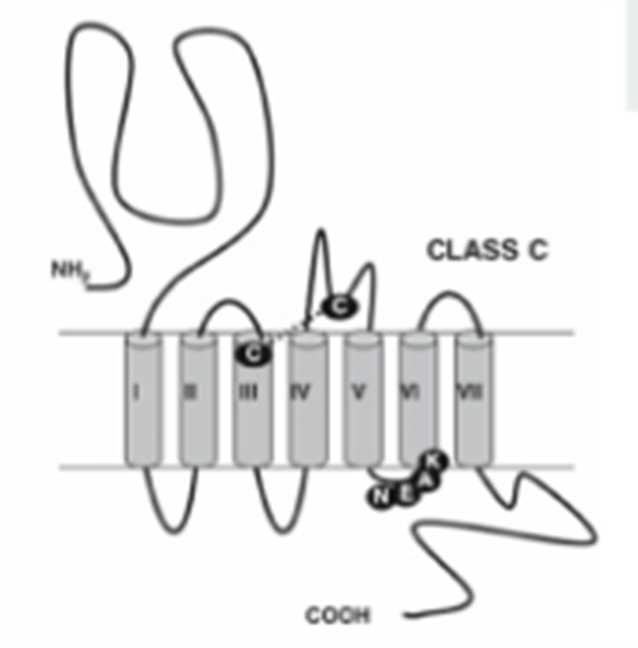
Glutamate family – Class C
majority of the GPCRs in this family are activated by glutamate, but also includes GABA receptors and calcium sensing receptors
Have EC Venus fly trap domains which acts as an intermediate region which when the receptor is activated, causes a conformational change which activates the 7 TM domain part of the receptor
9 cysteine domains present in calcium and glutamate receptors but missing in the GABA receptor
Have a coiled coil domain which is critical for dimerization
Are mandatory dimers and are not functional unless in a dimer
Can be homo or heterodimers → the GABA receptors are obligate heterodimers, glutamate receptor are 5 subunits which can be homo or heterodimers
GRAFS system - Rhodopsin
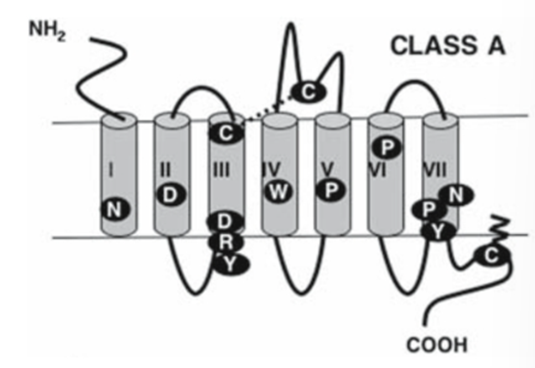
Rhodopsin – Class A
First GPCR in this family was the opsin in the rod cells
Includes dopamine, beta adrenergic receptors, acetylcholine metabotropic receptors
One of the best studied families so lots of crystal structures obtained
E/DRY motif is critical for G protein activation and the ionic lock which keeps the receptor in the inactive form —> mutations in this motif can lead to a constitutively active receptor
NPXXY motif in TM helix 7 plays a role in:
GPCR activation and conformational transitions
facilitates beta arrestin recruitment
structural changes between the active to inactive state.
Short EC domain
GRAFS system - Adhesion
Adhesion family – Class B2
Includes receptors with adhesion component for cell signalling
Have a unique hybrid structure – classic 7TM domain, but have very long N terminal region which contains the GAIN domain
Not only act as receptors, but also working as adhesion molecules to keep the cell joined together, mediating adhesion and transmission of signals
GAIN domain (GPCR Autoproteolysis inducing domain) which is cut and the detached fragment activates the receptor
This is required for changing the function of the receptor or liberating the agonists
Few examples of receptors where the native ligands are themselves, but can be activated by pharmaceuticals
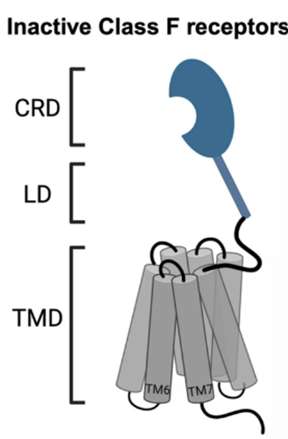
GRAFS system - Frizzled
Frizzled family – Class F and T
A lot of these receptors get activated by transcription factors
Receptors for wnt glycoprotein
Important role in development
Have a large EC cysteine rich domain called CRD which binds the ligand
IC region which couples to a group of proteins called dishevelled
Even though they activate the G protein, activation can also result in the activation of this group of phosphoproteins called dischevelled
CRD is the primary ligand domain for Wnt recognition
Binds in the thumb and hand way – the thumb enters the 7 TM domain and the knuckle enters the CRD which is where activation happens
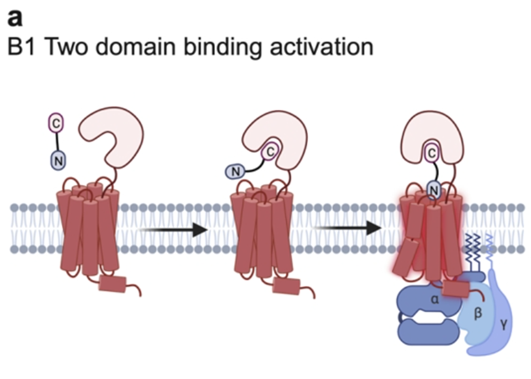
GRAFS system - secretin
Secretin family – Class B
Mostly neuropeptide binding
Hormone receptors with a large N terminal which is crucial for binding
Two domain activation: the peptide will bind to the EC domain first which holds it in the correct position to interact with the receptor
Well characterised by cysteine-rich regions in the EC N terminal, which are important for stability
Notoriously affected by the presence or absence of RAMP proteins (Receptor activity modifying proteins)
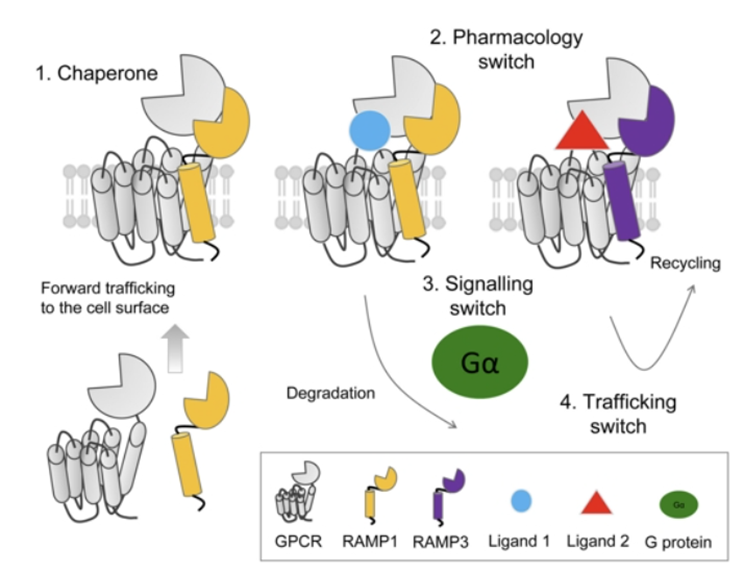
RAMP proteins
RAMP proteins
Act as chaperone proteins to trafficking receptors from the ribosome to the membrane
Or can act as a pharmacological switch to change the downstream pathway which is activated by signalling
Summary of GPCRs
Why are these very different group of signalling proteins linked?
All have 7 TM domains and signal through G proteins
All activated by EC ligands (in some cases opsins not activated by ligand but by light)
All seem to be affected by arrestins
Single GPCRs can also activate multiple G proteins, leading to a strong cellular response
Different functions of the beta-gamma complex
GPCR and G protein interactions
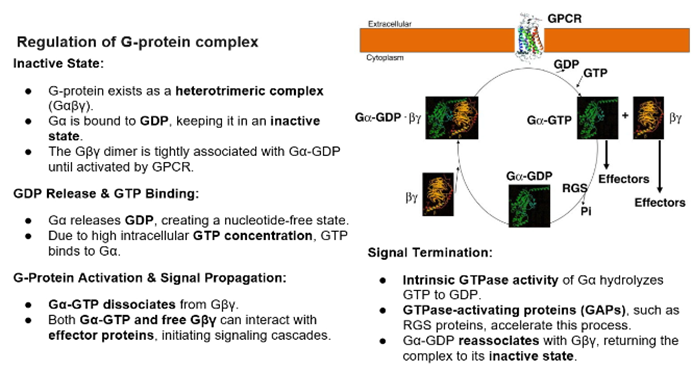
Limited set of G proteins
G protein activation is an intermediate between GPCR activation and intracellular effectors, role is to amplify the signal
Activation of one receptor can lead to the activation to multiple G protein complexes
Eventually, after activation, the GTP will lyse from the alpha subunit either spontaneously or by the help of other proteins, which will convert into the inactive state which can be activated by GPCRs
Ligand binding triggers a conformational change of the receptor in the EC and IC regions. The IC change repositions the protein to favour interaction with the G protein
G alpha binding regions formed by Tm domains 3,5,6 and 7 which come together to form the binding site
Number of subunits
Alpha: 21 types
Beta : 5 types
Gamma: 12 types
Different combinations of these allows for specificity, enabling cell-specific responses
Alpha proteins
4 major groups: s, I, q, 12
Each family has some conserved features but plays a distinct role in the activation of various downstream signalling pathways
Main difference is where they’re expressed:
E.g: s and olf are expressed in the olfactory neurons, while s is ubiquitous throughout
Beta proteins
We know that beta and gamma have important roles but we don’t know exactly what combinations change the transduction pathways or what roles they may have
We know that there is variation in expression though
Gamma proteins
Always bound to the beta
Important for the proper folding and functional activity of beta à acts as a chaperone protein of beta
Know that the dimers can regulate ion channels (like GIRK) when coupled to G alpha proteins
Phsopholipase C which is activated by beta gamma can influence cellular excitability and second messenger pathways
Gamma subunits are prenylated (farnesylated or geranylgeranylated) which anchors the Gby complex to the plasma membrane, ensuring proper spatial organisation of GPCR signalling
G alpha subunit function
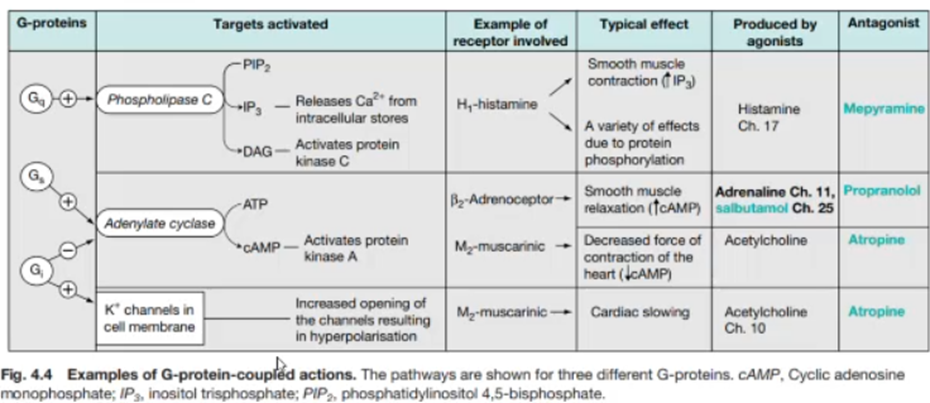
Galphas: stimulates adenylyl cyclase to increase cAMP, which activates the phosphorylation of proteins via PKA. Phosphorylation of these proteins results in activation of other pathways which can mediate behavioural responses
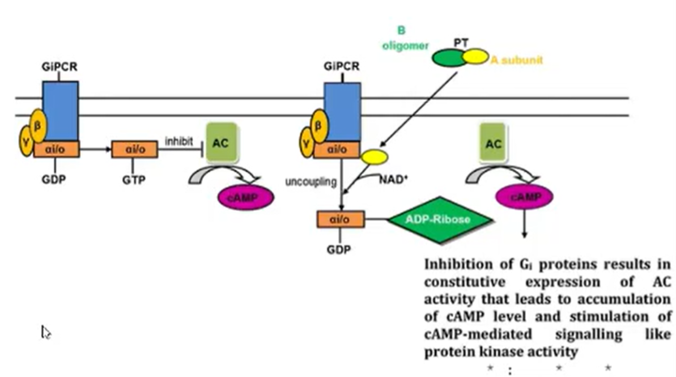
Galphai: inhibits adenylyl cyclase which decreases cAMP levels and opposes Galpas effects
Opposing activation to stimulation is important for returning to basal levels of cAMP
When the Gi activated: the beta gamma subunit can activate GIRK (ion channels). Some subtypes of receptors (M2 muscarinic Ach receptors) will not only decrease cAMP, but will also open postassium channels on Galphai activation, which is responsible for the cardiac slowing
Most of the adenylyl cyclase resides in the cell membrane but the reproductive organs also have a soluble form
cAMP acts as a mediator for a number of growth and metabolic functions in the cell
Galphai is inhibited by the pertussis toxin
has residues (cys) which are prone to targeting by the pertussis toxin
toxin covalently inhibits the Galpha I subunit and makes you unable to decrease the levels of cAMP, causing it to accumulate to toxic levels
only protein which is a target for pertussis toxin
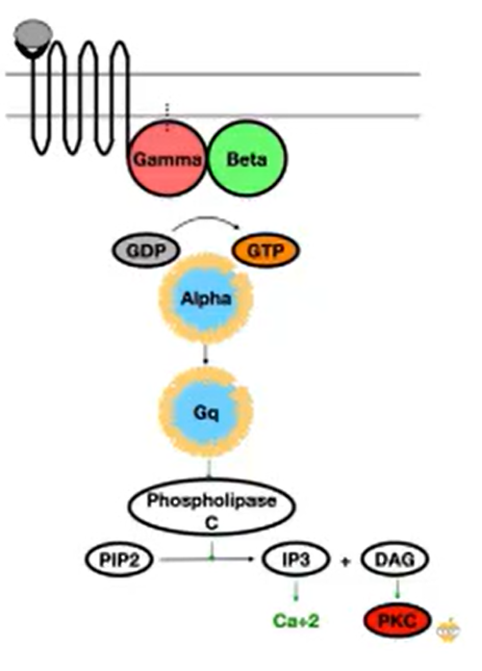
G alpha q
Activates PLC which is a protein which cleaves PIP2 into IP3 and DAG
IP3 is received by receptors in the ER, causing it to become activated and release Ca2+ ions à leads to many IC effects (e.g: depolarisation of neurons)
DAG leads to the activation of PKC
G alpha 12/13
Most recently G alpha subunit and the primary downstream effector is rho guanine nucleotide exchange factors à activate small rho GTPases which are important for regulating the cytoskeleton and cell motility
Tightly bound to adhesion receptors, some of the main mediators for adhesion receptors
Summary of G protein interactions
Chimeric G proteins
What allows the G proteins to only bind to one type of receptor and have this specificity
Engineer G alpha subunits which can reroute a receptors signalling into a different pathway
Example: Galpha qi5 – the original backbone of the protein was a Gq, but the last 5 residues were mutated to make it an I.
Some receptors are very specific in the type of G protein that they bind, while others are not so specific:
Adeonsine A1 and D2 preferentially bind to Gi proteins, whereas the adrenoreceptors which is able to bind to different pathways.
If you take the C terminal region of a G apah q and replace it with the C terminal region of a G alpha I protein
Did this sequentially – first changed one amino acid LàF and observed no activation by the D2 or A1 receptor
Then changed another AA NàG and start to see that the D2 receptor produced a small amount of signal
Did this for a series of residues and found that when they got to the fourth residue, the receptor was not interacting with the Gq proteins and were instead interacting with the I proteins
The activation of the receptors was measured by levels of IP3 (as a result of Gq activation)
However, if you change too many amino acids, you will lose function of the receptor à at 13 AA mutation, the D2 receptor loses function but the adenosine receptor still works
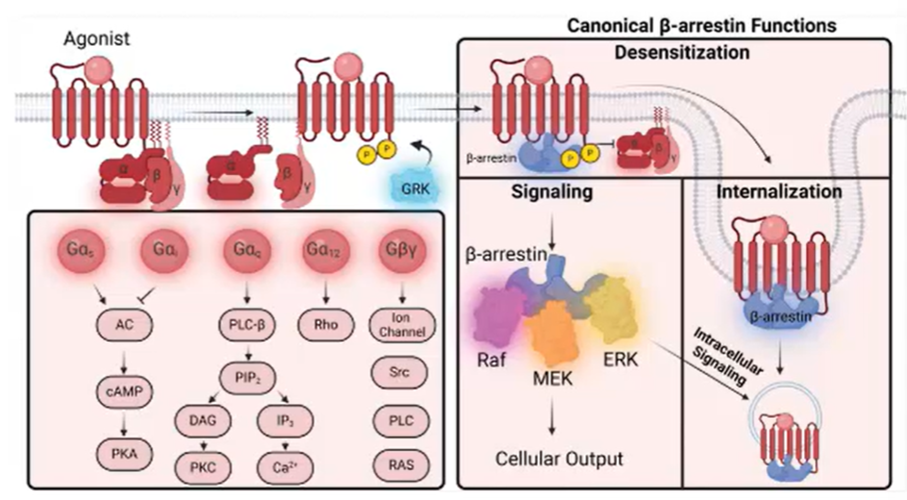
Non-cannonical signalling pathways
Proteins GRKs and arrestins were originally thought to be the determination of the signalling
When you need to return to basal levels the signal is terminated
3 competing molecules
G protein
GRKs (G protein coupled receptor phosphorylases) which are a family of serine threonine kinases that try and phosphorylate the receptor to terminate signalling
Arrestins – try and internalise the receptor back into the cytoplasm
GRKs
Serine threonine residues in the C-terminal of the receptor that get phosphorylated
Once phosphorylates can signal to the arrestins for internalisation
Phosphorylation is determined by many factors, such as:
Which cell type is it expressed in
Is it expressing more arrestins or G proteins (G protein>>arrestin results in lots of activation, whereas arrestin>>G protein results in rapid inactivation)
Once the signal has been terminated and the receptor internalised, can act as a signalling molecule
GRKs not only act on GPCRs but also have many other substrates
Arrestins
Arrestins thought only to be the signal terminators, but later found that receptors can also be compartmentalised and can travel to other organelles to have other functions à internalisation not always the ending of the signal but can be used to modify the ongoing signal
This could be why some drugs have secondary effects and if we can regulate this, can be used to mitigate secondary effects
Arrestins can also work as signalling molecules:
Phosphorylate other proteins
Activate MAPK pathways
Limited set of arrestins: 3 different families, only one that works with opsins
Majority of arrestins work with any type of receptor, which is useful for measuring arrestin interactions in assays
Summary of this section