Pharmacology of Adrenal Corticosteroids (Adrenal Gland
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
24 Terms
What is the adrenal glands?
- Two glands and pyramidal in shape, which lie on the anterior of the kidney
- Average size: 2-3 cm wide, 4-6 cm long and 1 cm thick with a mean weight of
4g irrespective of age, sex or weight
- At autopsy, the adrenal gland may weight up to 22g which appears to be a
result of the stress
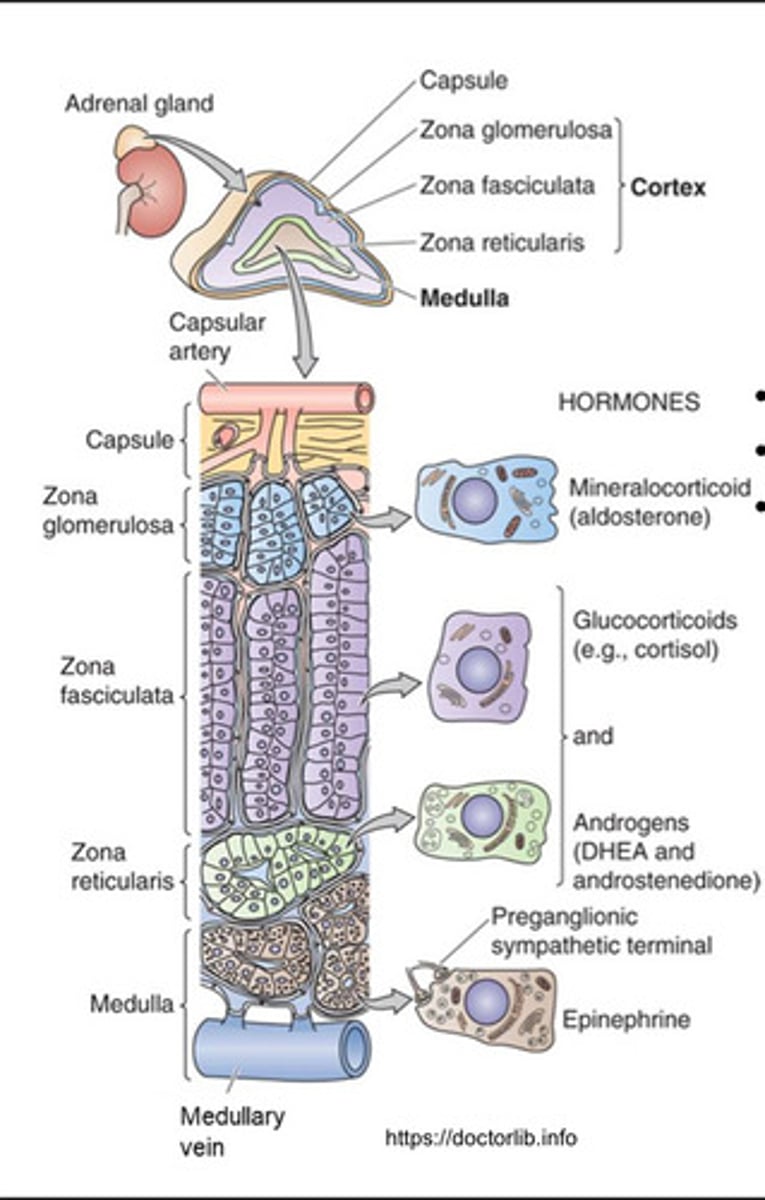
What is the structure of the adrenal gland?
- The adrenal gland sits on top of the kidney
- It consists of an outer cortex ~90% and a central medulla ~10%
- The cortex consists of three layers: zona glomerulosa, (zG), zona fasciculata
(zF), zona reticularis (zR)
- Mineralocorticoids are made in the zG
- Glucocorticoids are made in the zF
- Androgens are made in the zR
- A number of steroid hormone biosynthetic reactions require enzymes
localised in the mitochondria or the smooth endoplasmic reticulum
- Steroid hormone synthesis requires cholesterol which is stored in lipid
droplets
- Catecholamines are stored (primarily epinephrine i.e. adrenaline) in chromoffin granules (large vesicles in the adrenal gland that store and release hormones and other compounds) are secreted through
exocytosis
- Different cells in each layer of adrenal glands have different amount of
organelles depending on demands e.g., more mitochondria are found in the zG to make more
Mineralocorticoids because more enzymes involved in the synthesis of Mineralocorticoids are present in the mitochondria therefore you need more mitochondria to make the mineralocorticoids.
What are glucocorticoids?
A group of hormones that are similar to cortisol in structure and function. Glucocorticoids have anti-inflammatory, immunosuppressive, and vaso-constrictive effects. They are also used as drugs to treat a variety of conditions
What is the biosynthesis of steroid hormones?
- Main building block is cholesterol
- First step of synthesis of steroid hormones: conversion
of cholesterol to pregnenolone via catalysis of
Desmolase - remember this enzyme as it is responsible for the rate limiting step
-Remember one or 2 enzymes if you can but main focus is the steps and the hormones
- Each cortex layer is able to make 1 hormone and not another determined by
- Primarily involves CYPs and HSDs (enzymes)
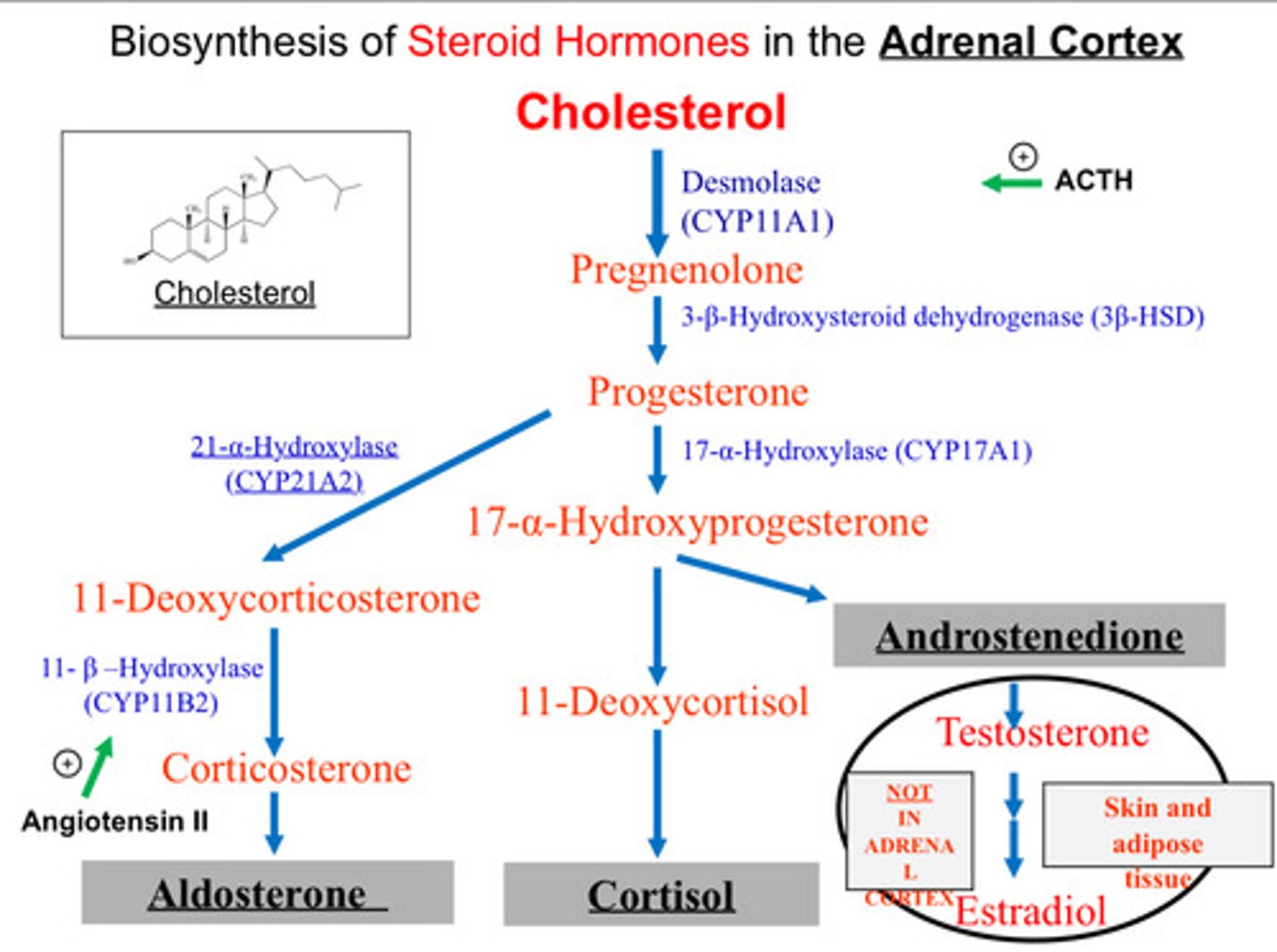
Slightly more detailed biosynthesis flow diagram of synthesis of steroid hormones
First step and its enzyme is very important !!!
- First step of synthesis of steroid hormones: conversion
of cholesterol to pregnenolone via catalysis of
Desmolase - remember this enzyme as it is responsible for the rate limiting step
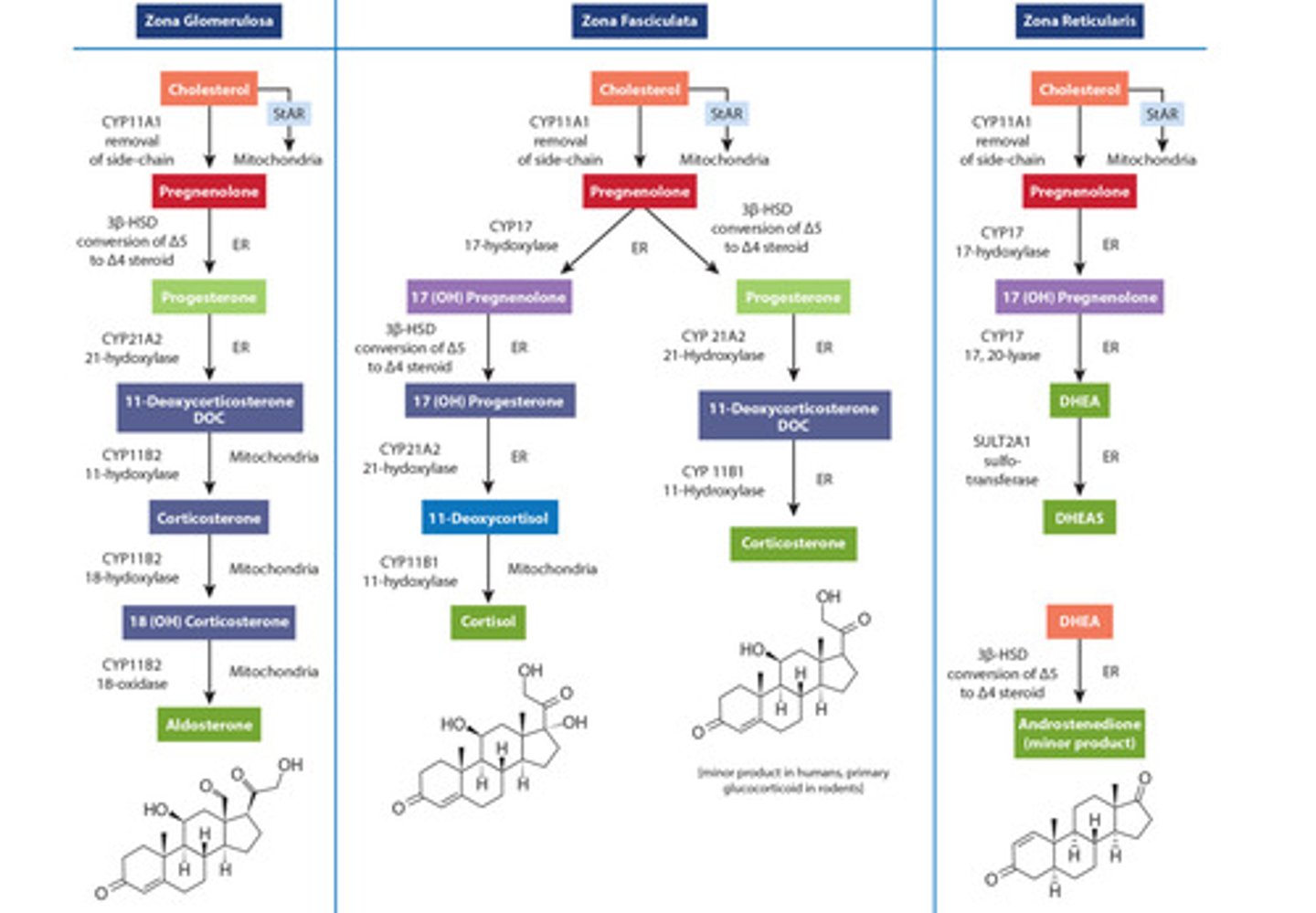
- Steroids in different colours align with different layers - represent availability of some of these enzymes of these cell types e.g. zG has green and no purple because In zG, it is missing 17 OH Pregnenolone- enzyme to convert is not present here
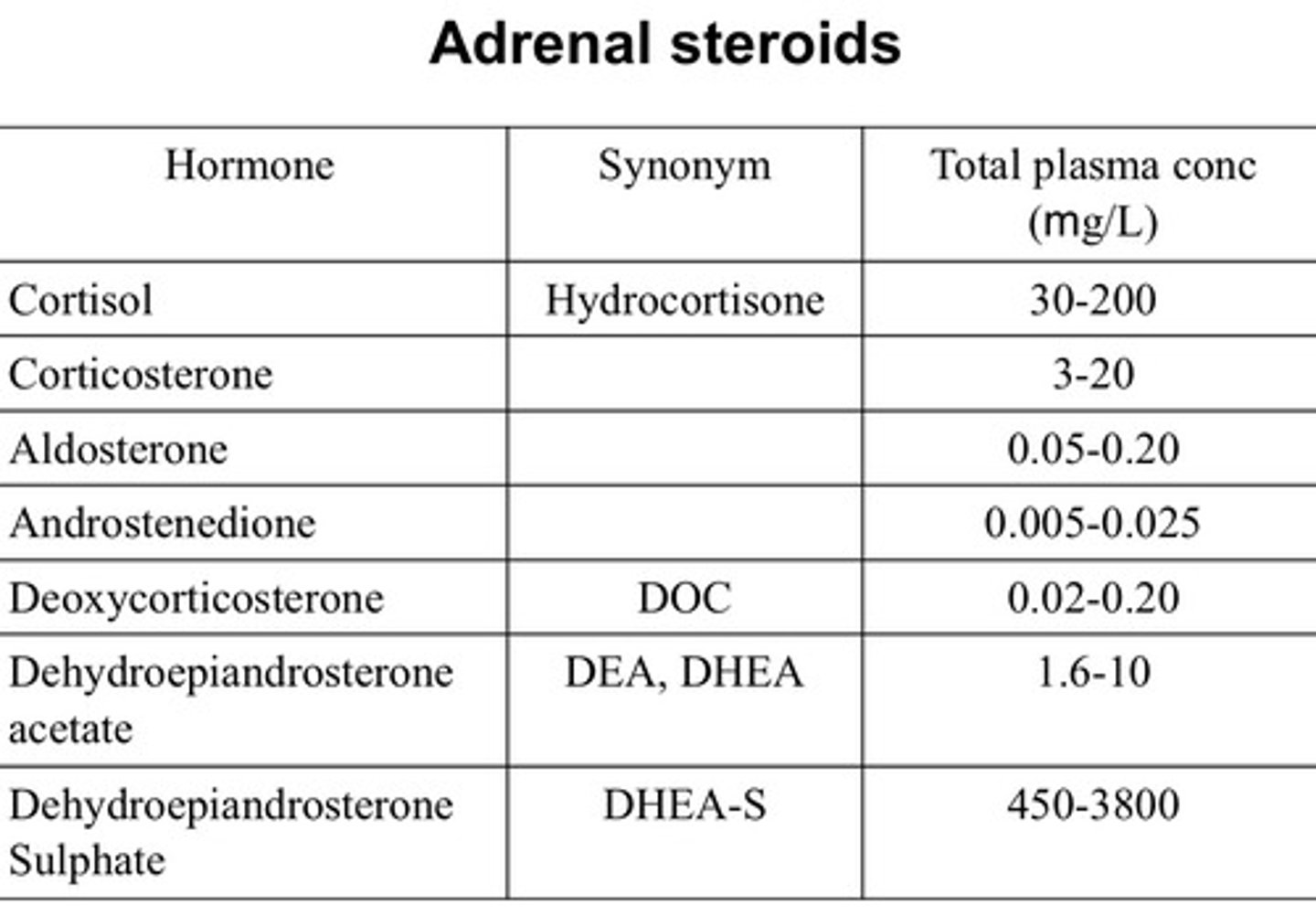
How much of each adrenal steroid circulates in the body?
- Cortisol one of highest active hormones
Note: DHEA-S is inactive so doesnt do anything but it is also in high levels
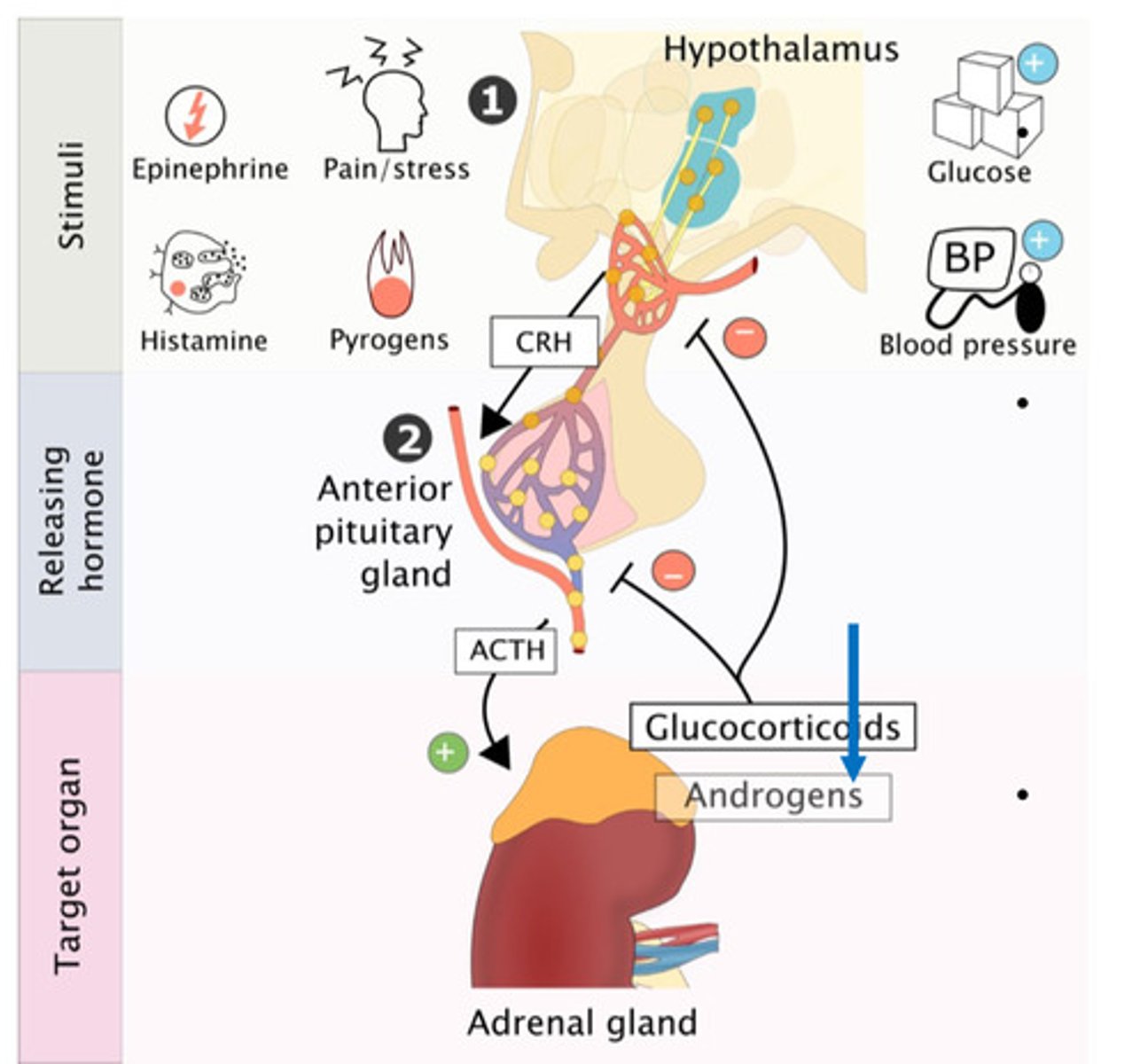
How is the synthesis of cortisol regulated?
-Stimuli stimulates the hypothalamus
- Hypothalamus releases corticotropin releasing hormone CRH which acts on
CRHR1
- CRH stimulates corticotrophs (neurones) in anterior pituitary to produce ACTH; ACTH
stimulates adrenal cortex to synthesise and release cortisol
Negative feedback mechanism:
- Cortisol feeds back on the corticotrophs of anterior pituitary to decrease
ACTH release and on the hypothalamus to inhibit CRH release
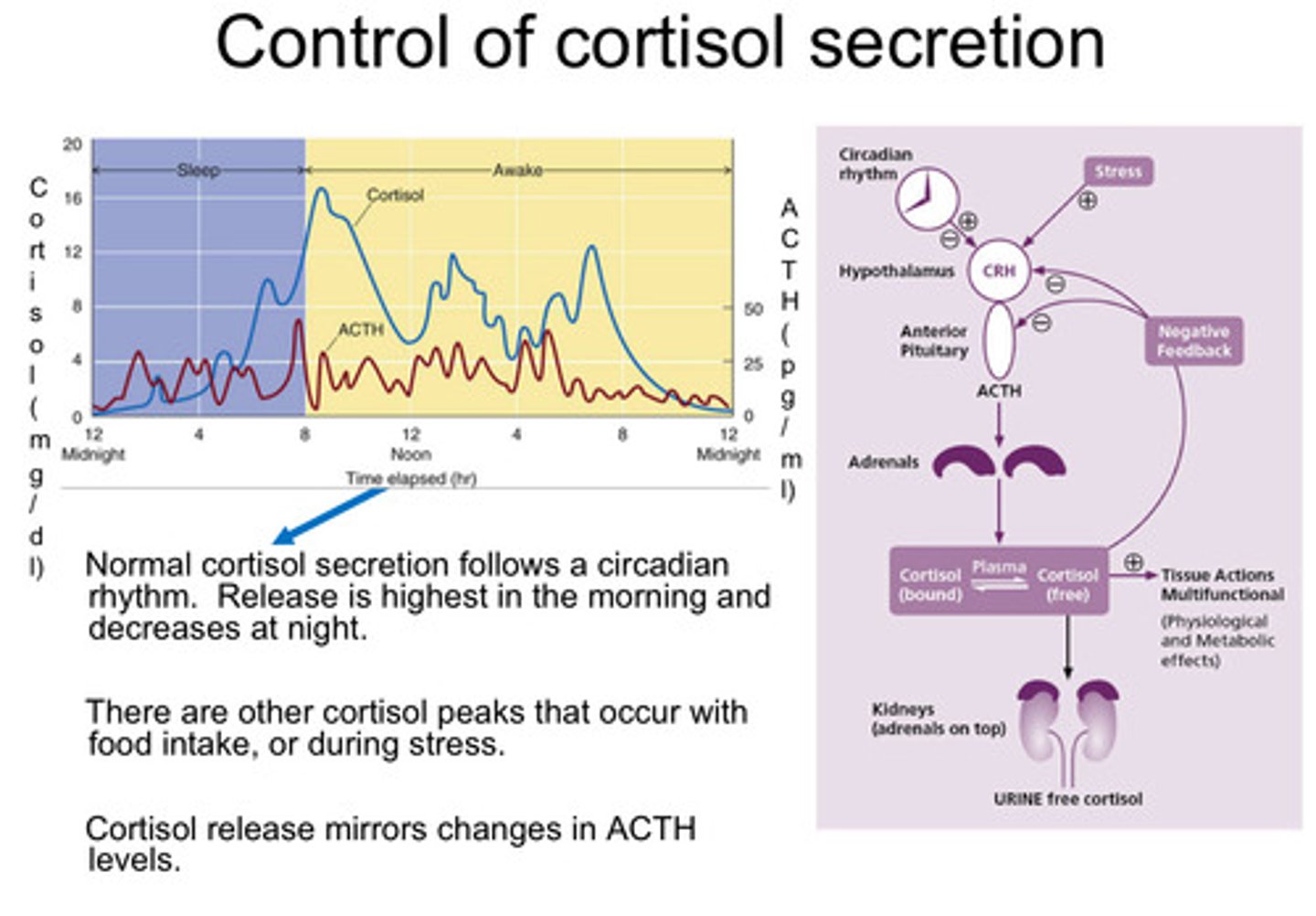
How is cortisol secretion controlled?
- Normal cortisol secretion follows a circadian rhythm
- Release is highest in morning and decreases at night
- Other cortisol peaks with food intake, stress
- Cortisol release mirrors changes in ACTH levels
- ACTH peaks with cortisol peaks- ACTH made first, stimulating
adrenals to make and release cortisol (therefore ACTH peaks will be shifted slightly to the left)
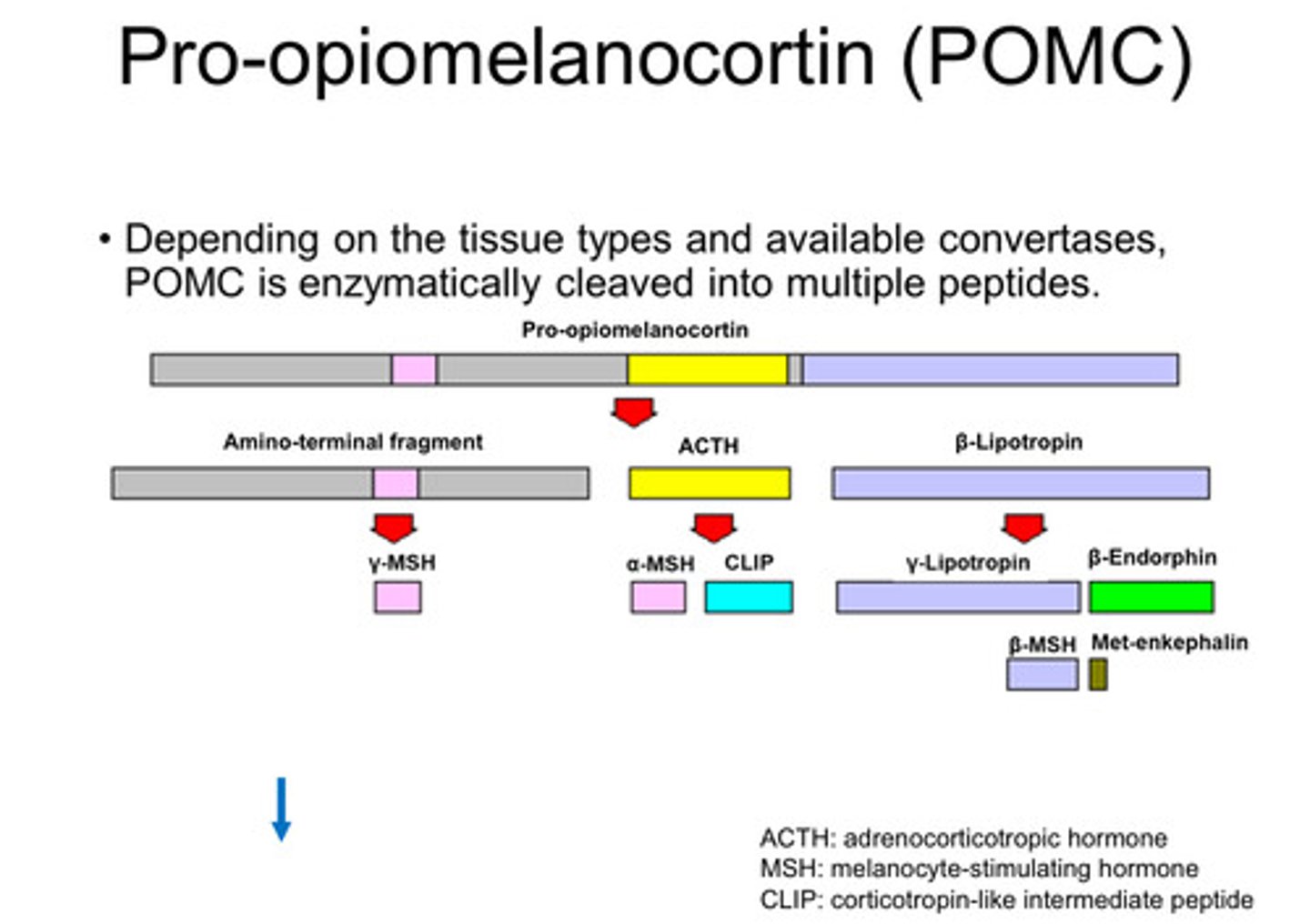
What is Pro-opiomelanocortin? POMC
- POMC is where ACTH comes from
-ACTH is not synthesised as ACTH but synthesised in a pro form (POMC) that contains several neuropeptides
-This allows the cell to save some energy and allows another level of regulation as the only places ACTH is synthesised is in the cells that contain the proteases that can cleave the pro form POMC.
- Depending on tissue types and available convertases, POMC is enzymatically
cleaved into multiple peptides
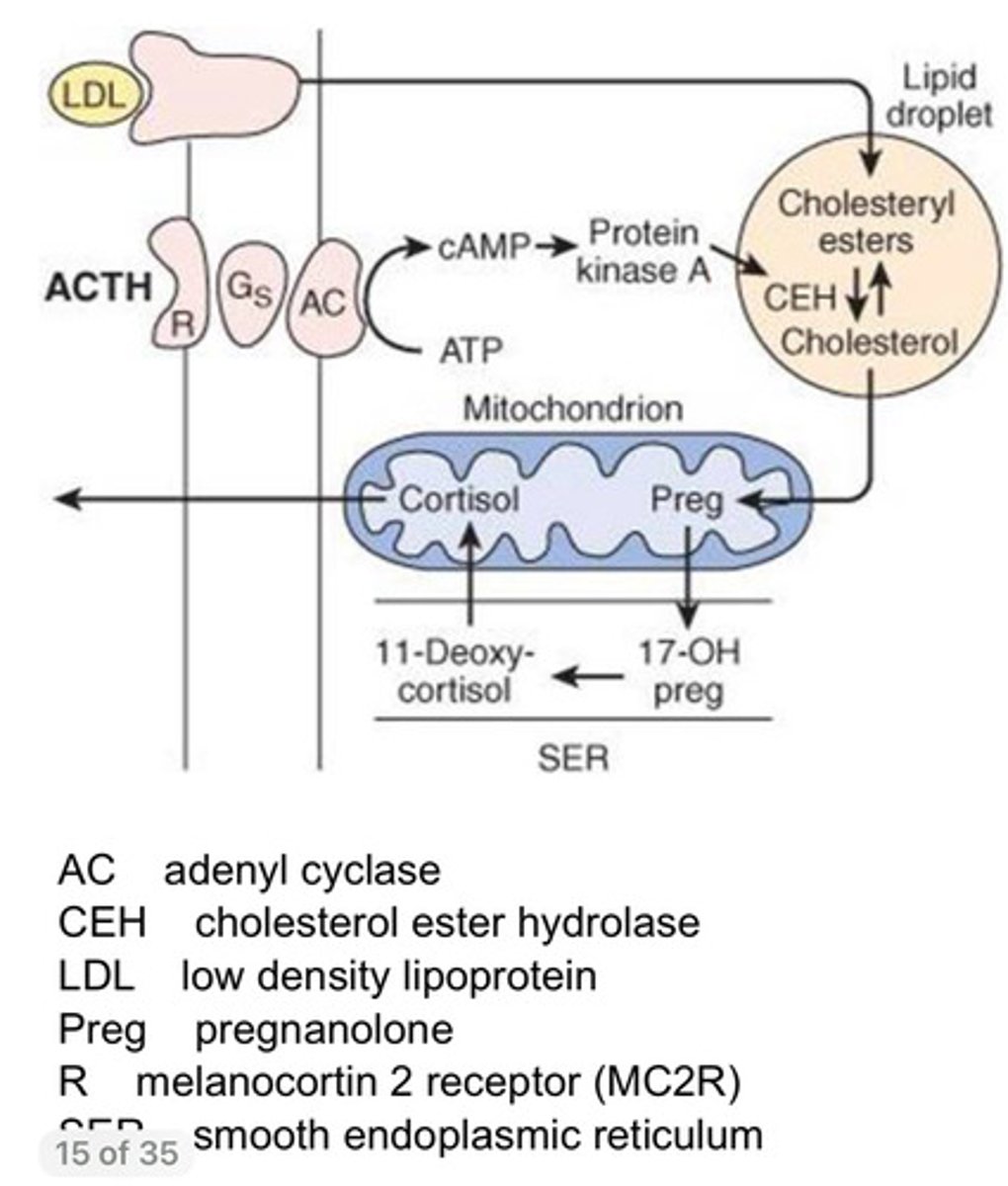
How is Cholesterol taken into the cells and how do we get them to stay in cells until we need them?
-Cholesterol is the basic building blocks of all of these hormones
-Cholesterol in all cells are taken up in LDL receptors (they take up Cholesterol bound LDL)
-Cholesterol in the cell can disassociate from LDL but due to it being a fatty molecule it wants to leave the cell through its lipid membrane
-To keep the cholesterol in the cell, we use esterification to be stored in that form in lipid droplets (Charged cholesterol can't leave)
Summary:
- Harder to keep cholesterol in lipids due to lipid-solubility, so is esterified by
enzyme which changes cholesterol into a cholesterol ester- making it charged
so it cannot leave the lipid droplets- when its needed to make steroid
hormones, ACTH causes activation of CEH enzyme which converts esterified
cholesterol back to normal cholesterol to make it leave droplets easily
How are steroid hormones synthesised (in this case cortisol)?
- Lipid droplets release cholesterol and (-PKA allows cholesterol to go into mitochondria, where it converted into pregnenolone ) it goes into
mitochondria
- In mitochondria, cholesterol is converted to Pregnenolone
- Pregnenolone leaves mitochondria into smooth ER, undergoes modifications
and leaves SER back into mitochondria to be converted to cortisol
- Cortisol is lipid-soluble diffusing out of mitochondria to target tissues
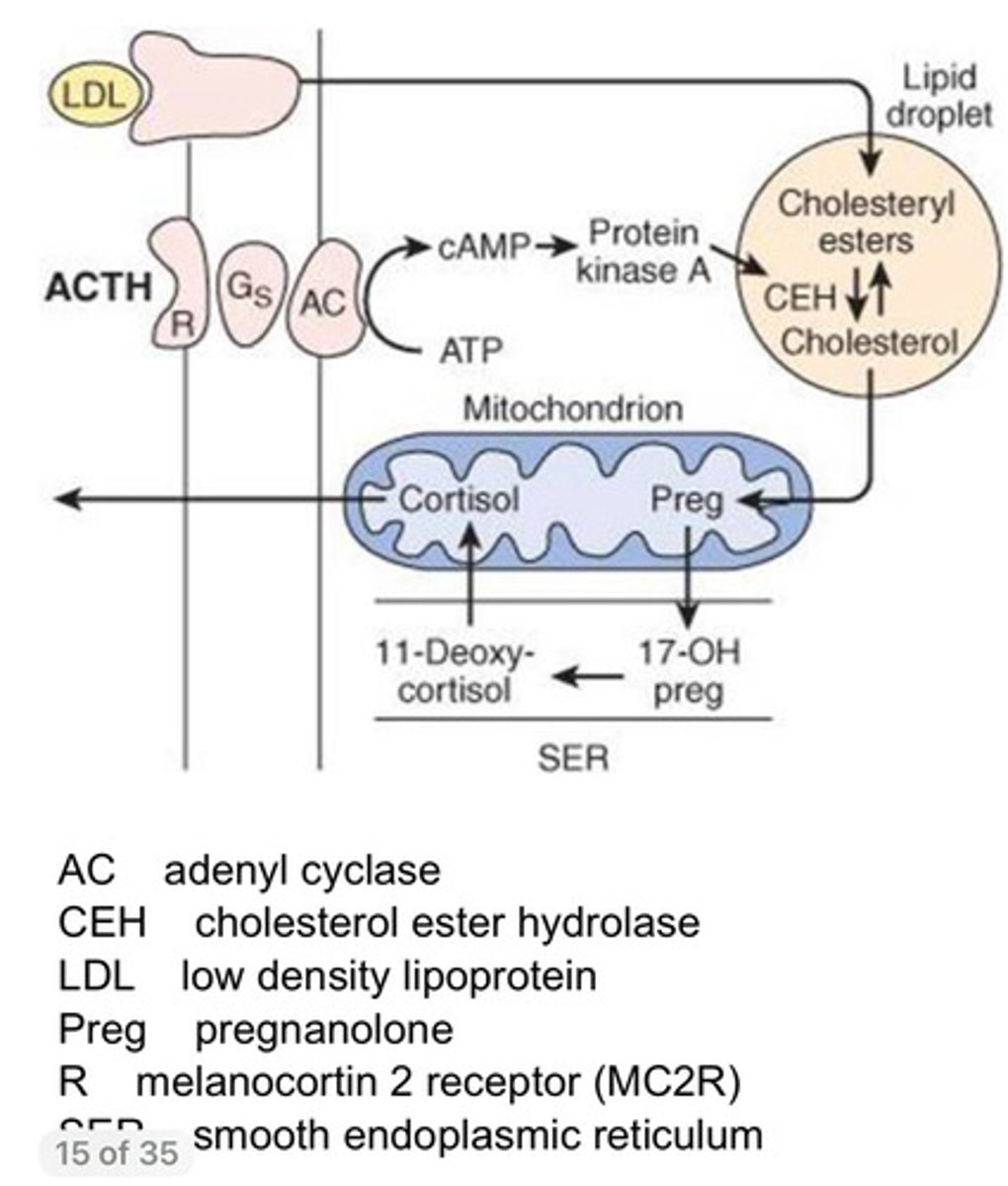
What is the action of ACTH on cortical cell?
- When ACTH binds to its receptor (A G protein couple receptor - Melanocortin Receptor, MC2R), it activates adenyl cyclase whicterm-16h stimulates cAMP (a 2nd messenger) synthesis
- This causes activation of Protein Kinase A (PKA); PKA has protein
substrates it phosphorylates = regulation of various
signalling pathways and metabolic rafts
PKA activation leads to:
•Activation of cholesteryl ester hydrolase (CEH) which liberates cholesterol from lipid droplets.
•Uptake of cholesterol into the mitochondria.
•Stimulation of cholesterol desmolase/CYP11A1 (rate limiting step in steroid hormone biosynthesis) which results in increased synthesis of cortisol.
ACTH has little effect on aldosterone synthesis, which is driven primarily by angiotensin II
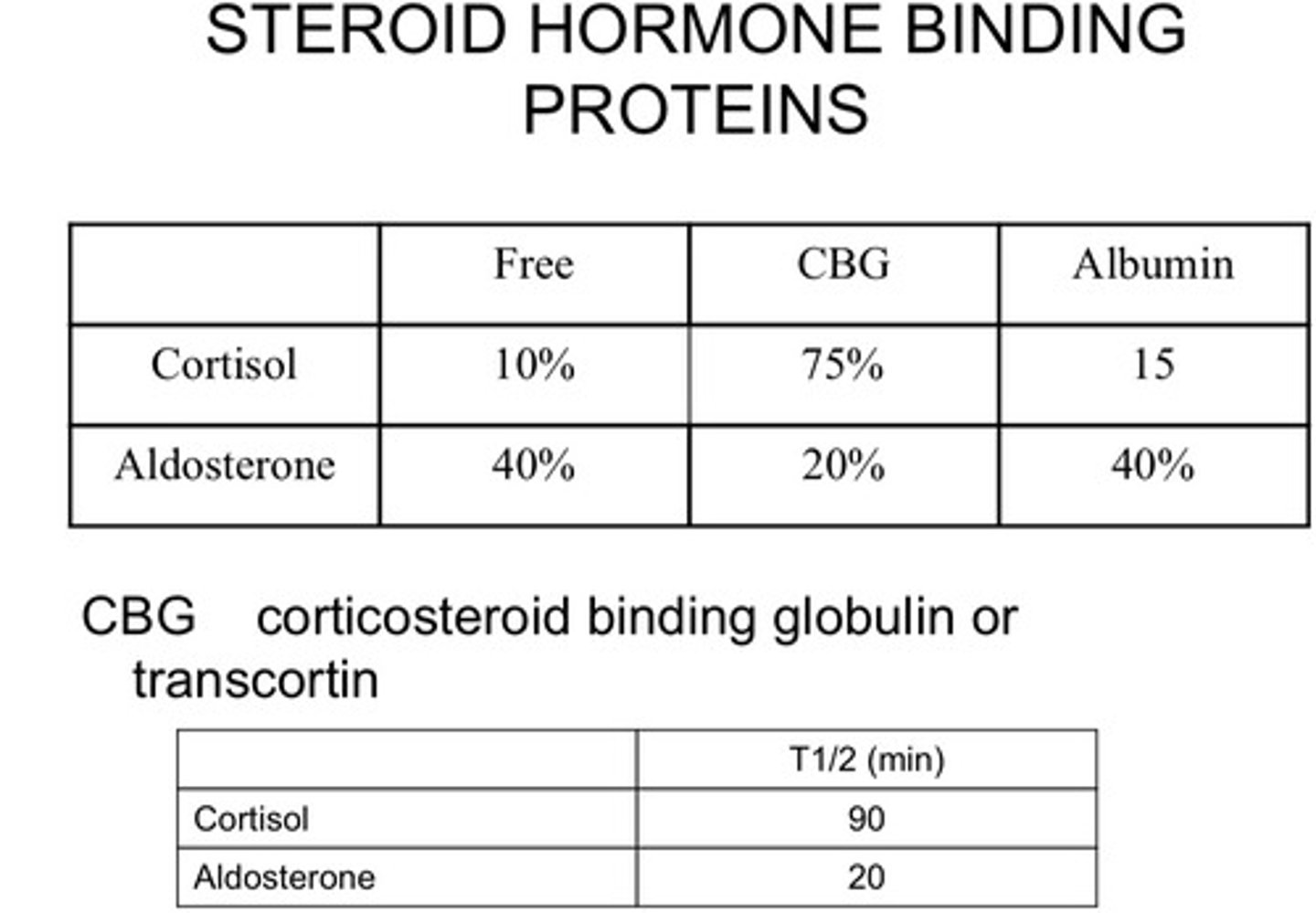
What determines how much steroid hormone is present in circulation?
- How much hormone in circulation is dependent on its
half-life
- When bound to these steroid hormone binding proteins they are harder to degrade
- Although aldosterone exists in free form 40%, its half-life is 5x shorter than
that of cortisol = Aldosterone is more unstable
-Less cortisol but it stays in circulation for longer
What is the mechanism for steroid receptor activation?
- Glucocorticoid Receptor made up of 3 domains: at end terminus there is ligand-binding
domain (where glucocorticoid binds) -
- In middle: DNA binding domain - where DNA binds
- At beginning: transcription activating domain- causes transcriptional
machinery to assemble target genes
- Inactive version of receptor kept in inactive form out of nucleus (in the cytosol) so unable to do transcription and also bound to group of proteins called Heat shock proteins HSP (90 and 70
keep them in cytosol)
- When Cortisol binds to one receptor = causes major conformational change leading to:
1) HSP proteins released= Allows the receptor to go from the cytosol into the nucleus which will then allow it to find these Hormone Response Elements (HRE - the DNA sequences that these receptors recognise.).
-When these transcription factors are activated by hormone they recognise these sequences and they recruit the transcription machinery and promote the expression of these genes. (How all these ligand transcription factors function)
-These transcription factors can also repress the expression of genes that are consistently expressed. Glucocorticoid regulated genes activates half of the genes it regulates it will activate and half it will repress.
-When DNA sits on receptor if Glucocorticoid regulated genes want to activate it, it will recruit transcription coactivators (these bring in mRNA synthetic machinery). For genes it wants to repress, it will recruit transcriptional corepressors (keep the machinery away so transcription doesn't occur).
Summary of What is the mechanism for steroid receptor activation?
Summary:
The *Glucocorticoid Receptor (GR)* has three key parts:
1. *Ligand-binding domain (at the end)*: This is where glucocorticoids like cortisol bind.
2. *DNA-binding domain (in the middle)*: This part connects to specific DNA sequences.
3. *Transcription activating domain (at the beginning)*: This area recruits the machinery needed for gene expression.
When inactive, the receptor stays in the cytosol (outside the nucleus) and is bound to proteins called *Heat Shock Proteins (HSPs), which keep it inactive. When cortisol binds*, the receptor changes shape, releasing the HSPs and moving into the nucleus.
Once in the nucleus, the receptor binds to *Hormone Response Elements (HREs)* on the DNA. This can either:
- *Activate gene expression* by recruiting coactivators (promoting mRNA production),
- Or *repress gene expression* by recruiting corepressors (blocking the transcription machinery).
So, the glucocorticoid receptor can both turn on and turn off different genes based on its interaction with DNA.
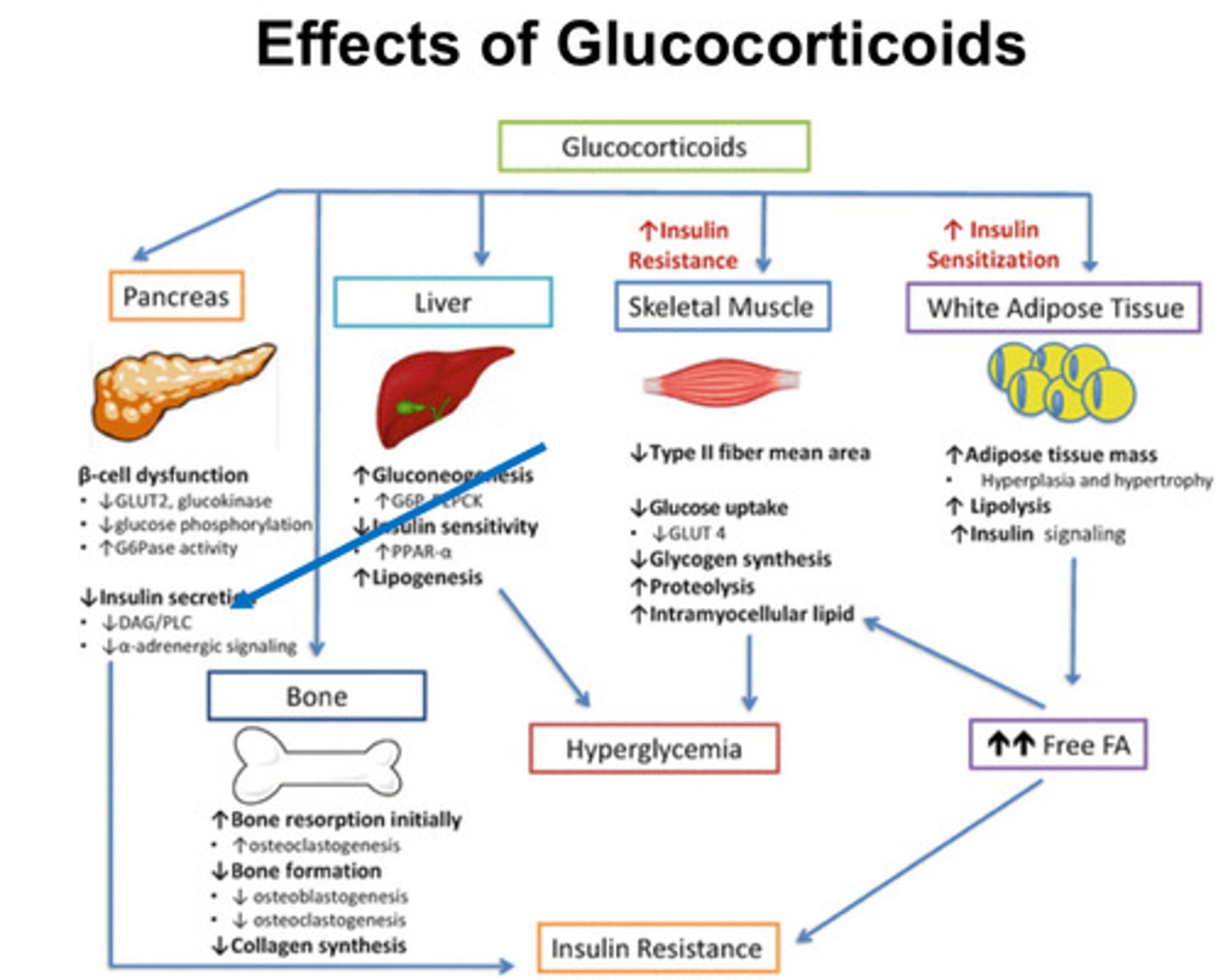
What are the effects of glucocorticoids e.g. cortisol?
- Metabolic effects: cortical stimulation = decrease in glucose uptake and use
& increase in gluconeogenesis; effect is hyperglycaemia
- Decrease in protein synthesis and increase in protein break up = muscle
wasting
- Decrease calcium absorption in gut, its excretion in kidneys
- Decrease activity of osteoblast and increase in osteoclasts = bone destruction
= osteoporosis
- Anti-inflammatory effects - Basically stops swelling: in early inflammation phase: reduce redness, heat , swelling
- In late inflammatory phase: reduced wound healing, repair and proliferation.
- How do they do this? By regulating expression of things which regulate
inflammatory responses e.g., decrease COX-2 expression, decrease
cytokines production, decrease complements in blood, decrease production of
Nitric oxide, Histamine release and IgG production
- Glucocorticoids (GC) also increase Lipocortin-1 which inhibits phospholipase A2 (Phospholipase A2, is also responsible for the production of arachidonic acid which also causes inflammation)
How can glucocorticoids act as mineralocorticoids?
- MCS involved in water regulation and electrolyte balance
- They increase Na, K and water retention
How does cortisol activation of mineralocorticoid receptor prevented?
- Cortisol has higher affinity for mineralocorticoid receptor than glucocorticoid
receptor (But can activate both)
- Glucocorticoids e.g. can exist as cortisol (active form) or cortisone (inactive)
- Converted by 11bHSDs enzyme- both forms catalyse either reaction
- 11bHSD2 isoform expressed in aldosterone sensitive tissues and converts cortisol to
cortisone - tissues that are responsive to mineralocorticoids, you don't want them responsive to cortisol hence why cortisol is converted to cortisone, so mineralocorticoids can carry out its function without glucocorticoids interfering (inactivates Glucocorticoids)
- 11bHSD1 isoform expressed in liver, adipose and muscle tissue and converts
cortisone to cortisol as cortisol is needed to act as a glucocorticoid (active form)
What are the adverse effects of glucocorticoids in chronic exposure?
- Can be precipitated by taking steroids on a regular basis
- Suppresses response to infection = more susceptible to infections
- Suppresses endogenous glucocorticoid production - Negative feedback, tells Hypothalamus and Anterior pituitary to stop producing ACTH = Stop producing Glucocorticoids
- Metabolic effects (Discussed above somewhere)
- Osteoporosis
- Iatrogenic Cushing’s syndrome
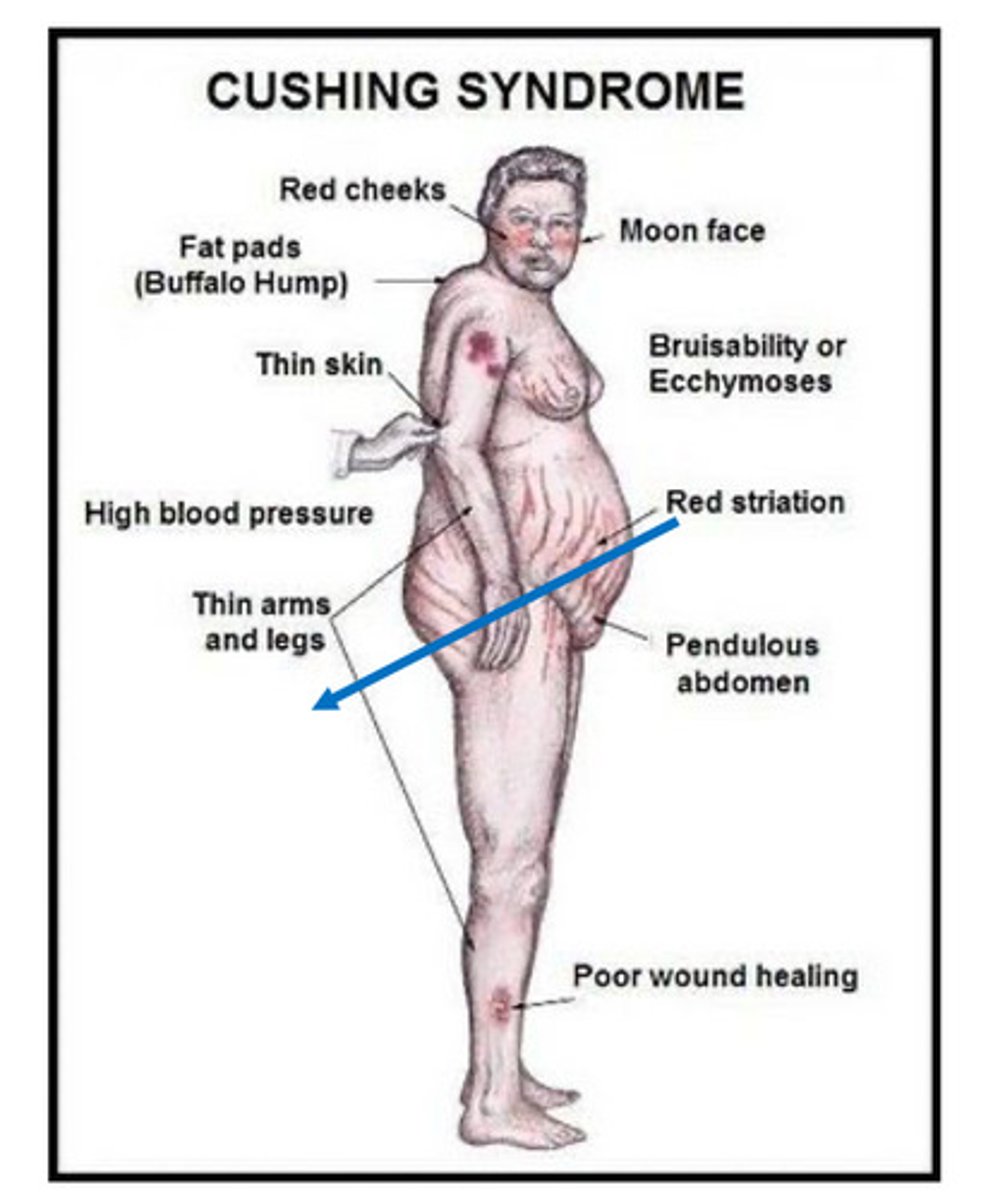
What is Cushing's Syndrome, what causes it and what does it cause?
- Caused by prolonged exposure to elevated levels of cortisol of exogenous
glucocorticoid drugs
- Also cause: osteoporosis, negative Nitrogen balance, increased appetite,
obesity, increased infection susceptibility
- Cushing’s disease can also be due to a pituitary tumour producing excess
ACTH
- ACTH-dependent make up 80% of cases
- ACTH-pit tumour Cushing’s disease is ~70% of the 80%
- Ectopic production of ACTH
(when a hormone is produced by a cell type or tissue that doesn't normally produce it, or produces it at low levels) ~10%
- ACTH-independent = adrenal adenoma, adrenal hyperplasia or cortisol-
producing adrenal carcinoma
- Clinical features: obesity, hypertension, hirsutism, striae, acne, bruising,
menstrual disorders, neuropsychiatric, impotence, glucose intolerance,
diabetes
What is the drug treatment for Cushing's syndrome?
- Inhibiting steroid biosynthesis: Metyrapone, Ketoconazone- these inhibit
enzymes
- Inhibiting ACTH release: Pasireotide, Cabergoline
- Inhibiting glucocorticoid receptor: Mifeprestone (also progesterone-receptor
antagonist)
Choice of drug depends on where the defect has taken place

What is Addison's disease?
- Chronic adrenal insufficiency - too little cortisol
- Primary insufficiency due to destruction of adrenal cortex
- Secondary insufficiency due to lack of ACTH
- Clinical features: weakness, fatigue, anorexia, weight loss,
hyperpigmentation, hypotension, GI disturbances, salt craving, postural
symptoms
- Treatment: replacement therapy with Fludrocortisone
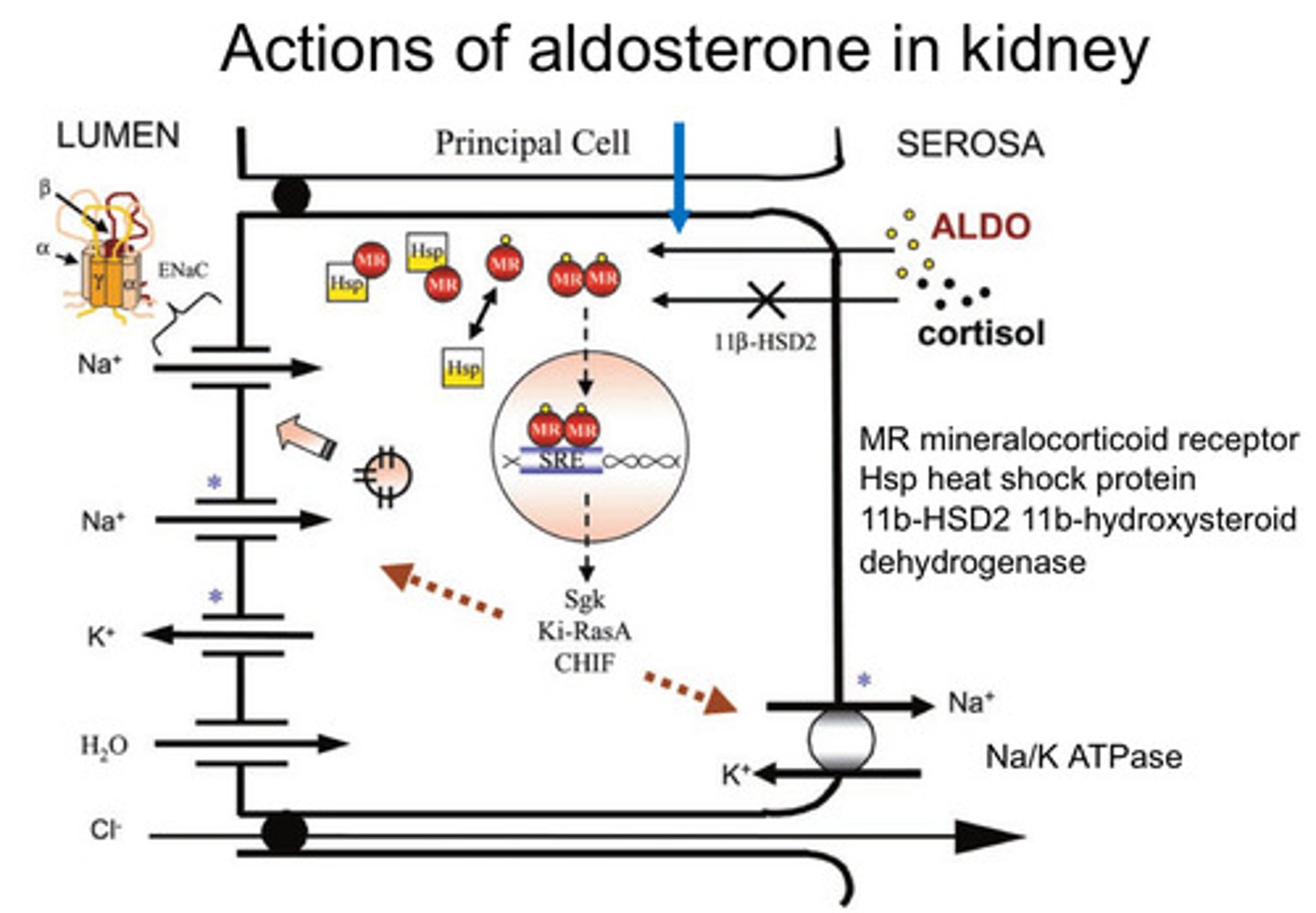
What are mineralocorticoids effects? - Rewatch this bit
- Aldosterone effects in the kidney: increases number of sodium channels in
apical membrane (ENaC transporter) and increases Na/K/ATPase symporter in basolateral
membrane of the collecting duct of the kidney
- Spironolactone: aldosterone antagonist which is used as K+ sparing diuretic
What are androgens and oestrogens?k
Adrenal gland secretes weak androgens:
-Dehydroepiandrosterone (DHEA)
-DHEA sulphate
-Androstenedione
Pre-menopausal women:
50% of androstenedione comes from the adrenal gland.
Androstenedione converts to oestrone and testosterone in peripheral tissues.
Excess secretion leads to hirsutism (excess hair) and virilisation (male characteristics).
Post-menopausal women:
-Main estrogen is oestrone (after ovary regression).
-Oestrone production occurs in fat (adipose) tissue.
-Obese women produce more oestrone than thin women.
Males:
-Only 5% of testosterone comes from adrenal androgens.
-Most testosterone is produced by the testes