RACC 1
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
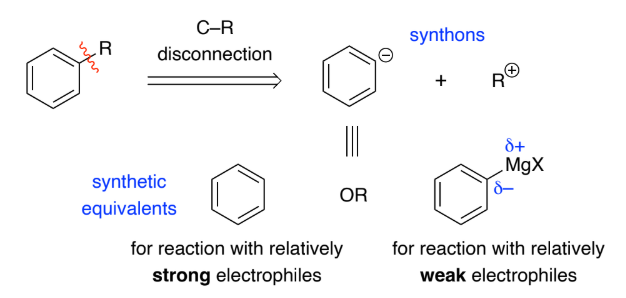
Normally the bond to the aromatic ring is broken to give a synthon with a _________ charge on the aromatic ring as they react with __________ in ____________substitutions
negative, electrophiles, electrophilic
OH & OR groups are difficult to add to a benzene ring, so don’t disconnect them but use _______ as a starting material
phenol
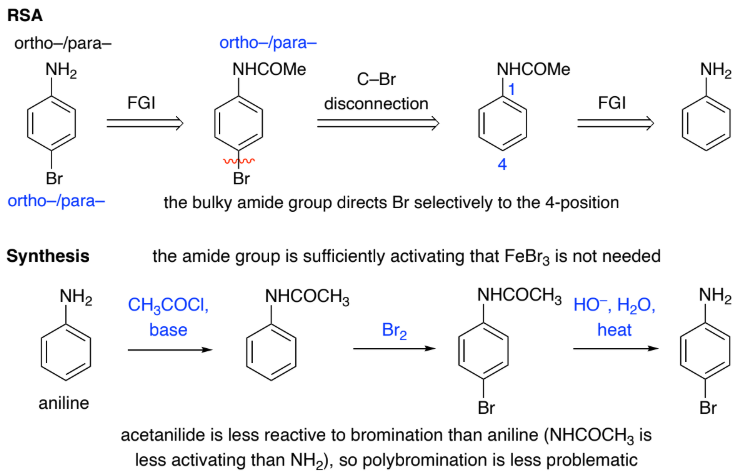
To improve regioselectivity, introduce a temporary _______ group e.g.
convert an NH2 group into a larger NHCOCH3 group
blocking
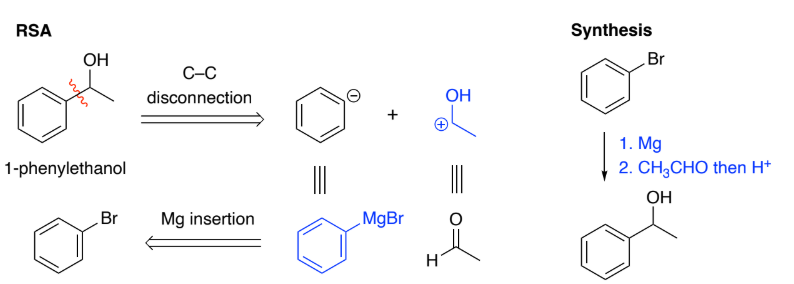
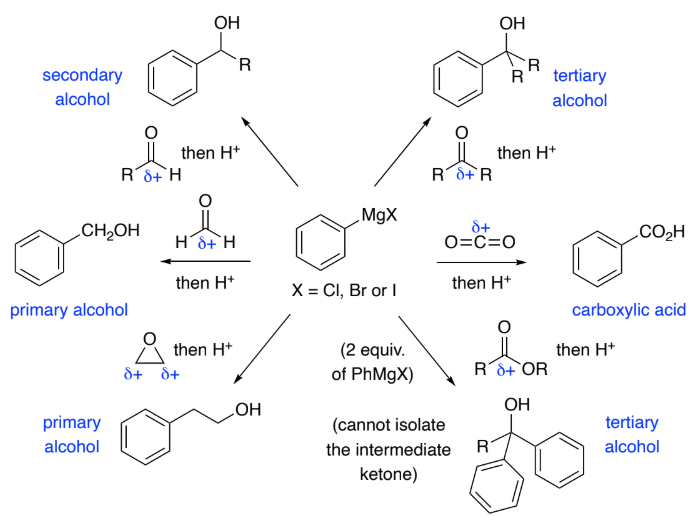
Grignards are formed by Mg insertion into an aryl halide.
They are strong nucleophiles & react with a range of electrophiles to form C–C bonds.
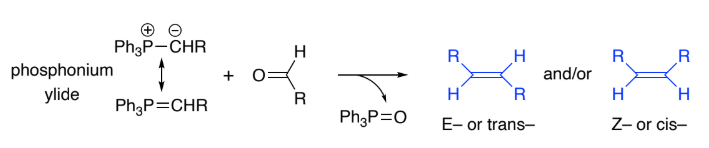
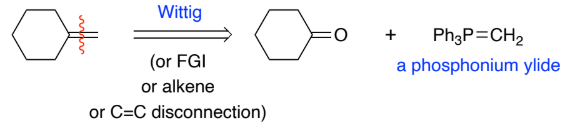
Wittig reaction is the general method for making ______ bonds.
C=C
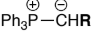
Stabilised ylides are prepared by deprotonation of a phosphonium salt, that has an EWG, Ph3P+CH2EWG w/ weak base. The EWG stabilises the carboanion formed
They:
are unreactive to ________
react with aldehydes to give mainly ___ - alkenes, why?
ketones, E
E is more thermodynamically stable
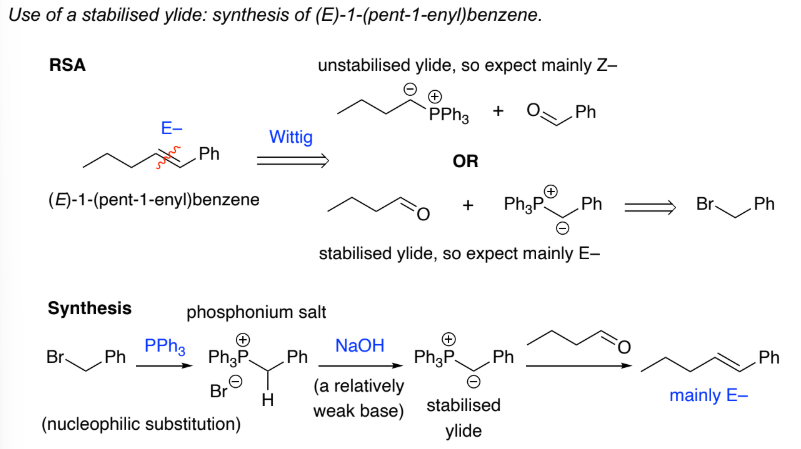
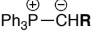
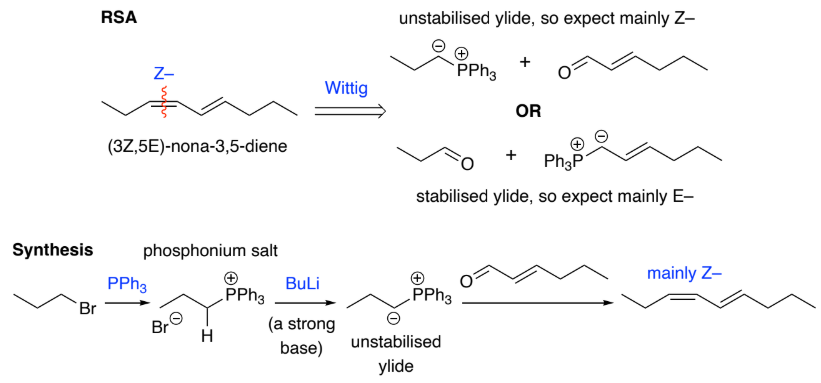
Stabilised ylides are prepared by deprotonation of a phosphonium salt, that has an EDG, Ph3P+CH2EDG w/ strong base. The EDG further destabilises the carboanion formed
They:
react with ketones & aldehydes to give mainly ___ - alkenes
Z
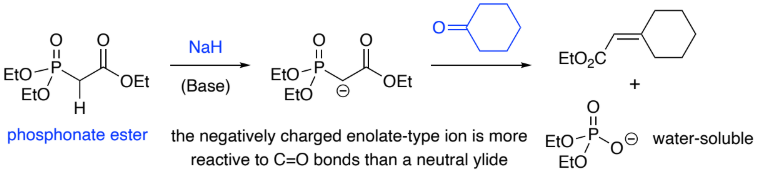
A deprotonated phosphonate ester (________ charge) is more reactive to a ____ bond than a phosphonium ylide (neutral).
Reactions of stabilised (EWG) phosphonate ester anions w/ aldehydes/ketones
gives mainly ____ - alkenes.
negative, C=O, E
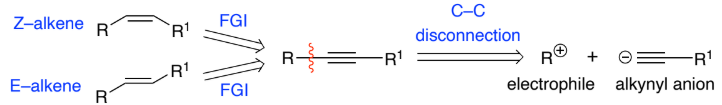
Alkynyl anions are formed by deprotonation of _____________ w/ base
They’re strong nucleophiles & react with a range of electrophiles to form C–C bonds.
C≡C bond can be reduced to form ________ stereoselectively using:
_____________ to form a Z–alkene
________ to form an E–alkene
Terminal alkynes, alkenes
H2/Lindlar catalyst
Na/NH3
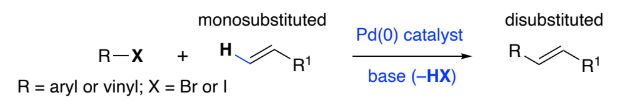
Heck reaction:
Palladium(0)-Catalyst can be used to form a disubstituted alkene from a terminal alkene, base &:
aryl halide (___)
or
vinyl halide (____) – the C bonded to X must be ___ hybridised
Reaction is highly selective:
• R group is introduced at the _______________ end of the C=C bond
• More stable _________ is formed.
ArX
C=CX, sp2
least hindered, E-alkene
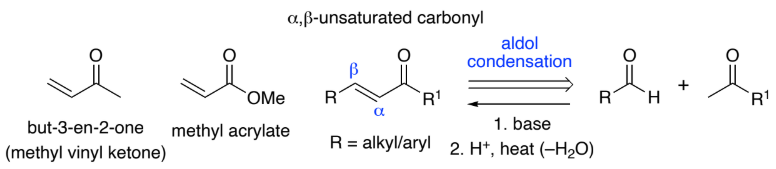
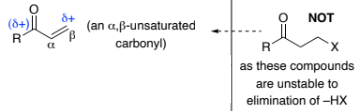
___-Unsaturated carbonyl compounds are RASM’s that are prepared via aldol condensation reactions of aldehydes, ketones & esters
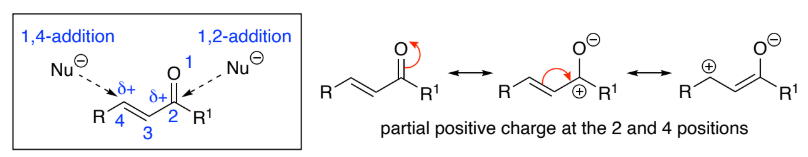
Conjugation of the C=O bond with the C=C bond means that α,β-unsaturated carbonyls are susceptible to attack by ___________ at both the 2- & 4-positions
1,4-addition, is called Michael addition
Addition of carbon nucleophiles results in _____ bond formation
α,β , nucleophiles, C–C
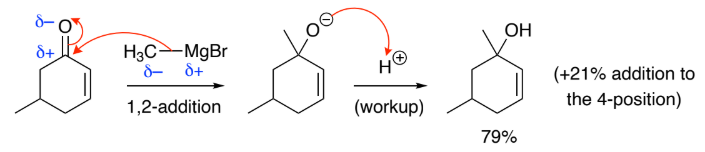
1,2-Addition (C=O addition):
Grignard reagents (____) attack directly at the ___ bond
Under ________ control because the stronger C=O bond is broken & the weaker _____ is retained
Grignard reagent is a _________ & the C on C=O is a _____________ because
C on C=O is a _____________ because it is directly attached to O which has a high δ+ density
Grignard reagent is a _______________ because the R in RMgX has a high δ– density as it’s adjacent to an electropositive Mg
Hard reacts preferably w/ hard so Grignard reagent prefers 1,2 (C=O) addition
RMgX, C=O, kinetic, C=C
hard electrophile
hard nucleophile
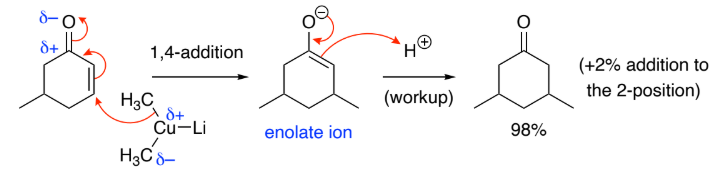
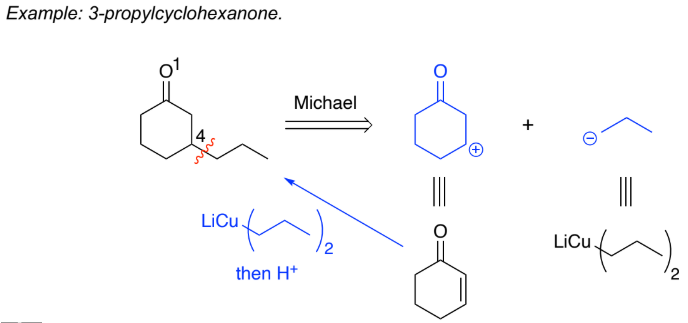
1,4-Addition (Michael addition):
Organocopper reagents (RCu) & cuprates (R2CuLi) attack the ___ bond
Under ___________ control because the stronger ___ bond is retained & the weaker C=C bond is broken (more stable product is formed)
C at position 4 is a ______________ because it has low d+ density
A cuprate is a ______________ because the R groups in R2CuLi have low d– density because they’re adjacent to a weakly electropositive Cu
Soft reacts preferably w/ soft so organocopper reagents/cuprates prefer 1,4 (Micheal) addition
C=C, thermodynamic, C=O
soft electrophile
soft nucleophile
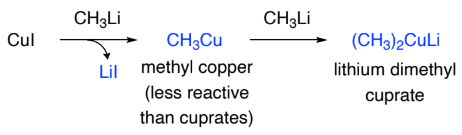
Organometallics containing copper can be prepared from ________ compounds:
organolithium
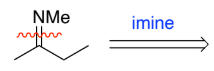
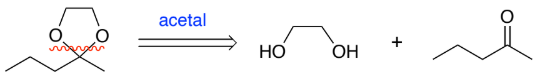
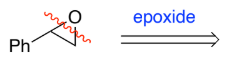
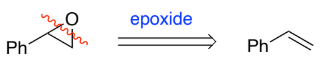
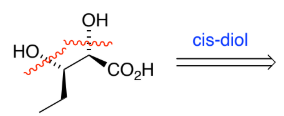
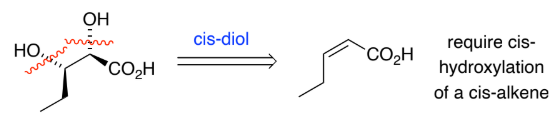
RC(=O)R → RC(-OH)R (ketone to secondary alcohol)
LiAlH4
H+
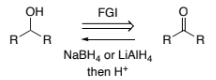
RC(=O)OH → RC(-OH)H (carboxylic acid to primary alcohol)
LiAlH4
H+
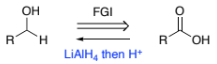
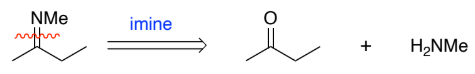
RC(=NH)R → RC(-NH2) (imine to amine)
NaBH4
H+
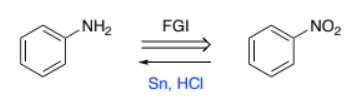
Ph-NO2 → Ph-NH2 (benzene w/ nitro to benzene w/ amine
Sn, HCl
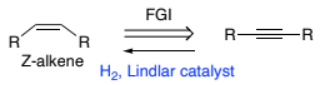
R-C☰C-R → Z- RHC=CHR (alkyne to Z-alkene)
H2, Lindlar catalyst
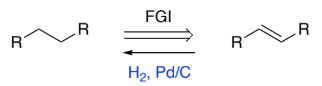
RHC=CHR → RH2C-CH2R (alkene to alkyl)
H2, Pd/C
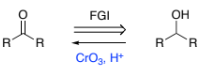
RC(-OH)R → RC(=O)R (secondary alcohol to ketone)
CrO3, H+ reflux
RC(-OH)R → RC(=O)R (primary alcohol to ketone)
CrO3, H+ distillation (or milder oxidant e.g. PCC)
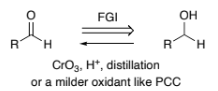
RHC-OH → RCO2H (primary alcohol to carboxylic acid)
CrO3, H+ reflux
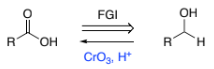
Ph-CH3 → Ph-COOH
KMnO4 (potassium permanganate)
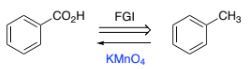
RH2C=CH3 → peroxide
RCO3H (peracid)
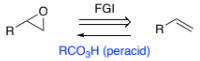
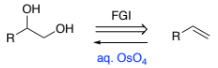
RH2C=CH3 → RH(HO-)C-C(-OH)H2 (alkene to diol)
aq. OsO4
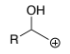
R+
R-I
R-Br
R-OSO2Me (R-OMs)
R-OTs

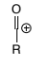
HO-C+
Ketone/aldehyde

HO-C-C+
Epoxide
O=C+-R
Carboxylic acid derivatives
e.g. X=
Cl
OCOR
OR
NR2
CO2
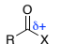
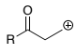
R(O=)C-CH2-C+
α,β-unsaturated carbonyl
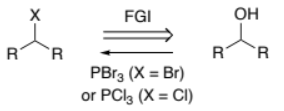
RHC(-OH)R → RHC(-X)R (alcohol to halogen)
PBr3 (when X = Br)
or
PCl3 (when X = Cl)
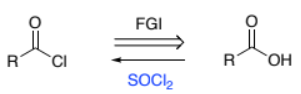
RC(=O)OH → RC(=O)Cl (carboxylic acid to acyl chloride)
SOCl2
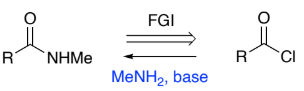
RC(=O)Cl → RC(=O)NHMe (acyl chloride to secondary amide)
MeNH2, base
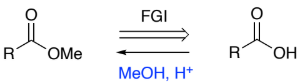
RC(=O)OH → RC(=O)OMe (carboxylic acid to ester)
MeOH, H+
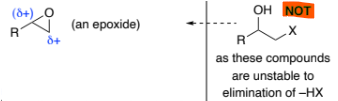
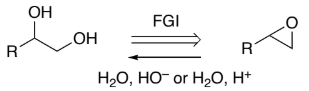
epoxide → R(HO)C-C(OH)H2 (epoxide to diol)
H2O, HO–
or
H2O, H+
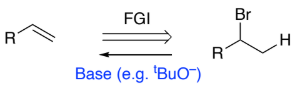
RH(Br)C-CH3 → RHC=CH2 (halogenoalkane to alkene)
Base (e.g. tBuO–)
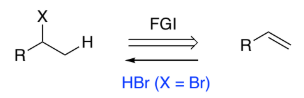
RHC=CH2 → RH(X)C-CH3 (alkene to halogenoalkane)
HBr (when X = Br)
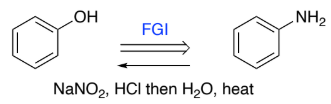
Ph-NH2 → Ph-OH (aniline to phenol)
NaNO2, HCl
H2O, heat
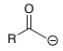
R-
Organometallics
δ–R–MgX
δ–R–Li
δ–R2CuLi
Ar-
ArH
δ–Ar–MgX
δ–Ar–Li
δ–Ar2CuLi
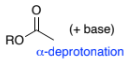
RC(=O)C-H2 (ketone)
RC(=O)CH3 + base for α-deprotonation

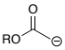
ROC(=O)C-H2 (ester)
ROC(=O)CH3 + base for α-deprotonation