Chapter 7- Covalent and Metallic Bonding
- When non-metals react with one another, they share electrons instead of transferring them just as in the diatomic elements.
- The bond formed by sharing of electrons between non-metals is the covalent bond.
- Sharing of one pair of electrons, i.e. two electrons, forms one covalent bond. Hydrogen has one covalent bond while nitrogen has three.
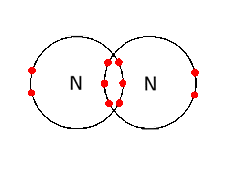
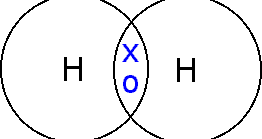
- One covalent bond is represented by one line in the structural formula.
- Compounds linked by covalent bonds are called covalent compounds or molecular compounds. Water, carbon dioxide, and methane are examples.
- Within each molecule of a simple molecular compound, there are weak intermolecular forces of attraction so they have low densities, and melting and boiling points.
- Most simple molecular compounds are soluble in organic solvents but not in water.
- They do not conduct electricity in any state due to lack of mobile electrons/ions.
GIANT MOLECULAR STRUCTURES
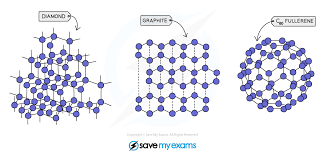
- Diamond and graphite, allotropes of carbon have giant covalent structures.
- Giant molecular structures are usually solids at r.t.p, have high melting and boiling points, and high densities, and do not conduct electricity in any state (except graphite).
- Graphite is slippery so it is used as a lubricant while diamond is hard and shiny so it is used for cutting purposes.
- Sand (silicon dioxide) is another giant molecular structure.
Common prefixes to name covalent compounds are mono, di, tri, tetra, penta and hexa.
METALLIC BONDING
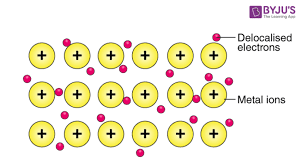
- In metals, electrons break free of the atoms forming cations and become delocalised themselves. The electrons and cations attract each other due to opposite charges on them.
- The force of attraction between positive metal ions and ‘sea’ of delocalised electrons is the metallic bond.
- Since there are a lot of free electrons that move throughout the metallic structure, metals are good conductors of heat and electricity.
- Metals are ductile and malleable because when force is applied, layers of atoms can slide over one another without disrupting the structure.
- Due to strong forces of attraction between electrons and ions, metals have high melting and boiling points, and densities (exception: Group I)