CH30 Amino acids, proteins and DNA
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
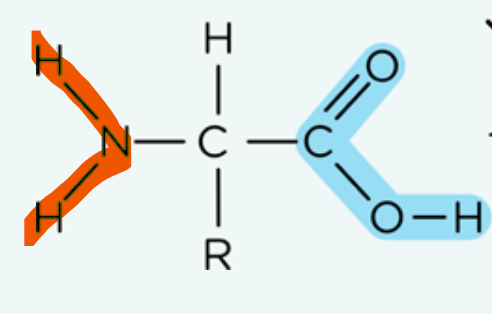
Describe the structure of amino acid:
Contains a carboxylic acid group
Contains a amine group (primary amine)
The chiral carbon is bonded to 4 different groups, so optical isomerism can be present, where they can exist as an enantiomer.
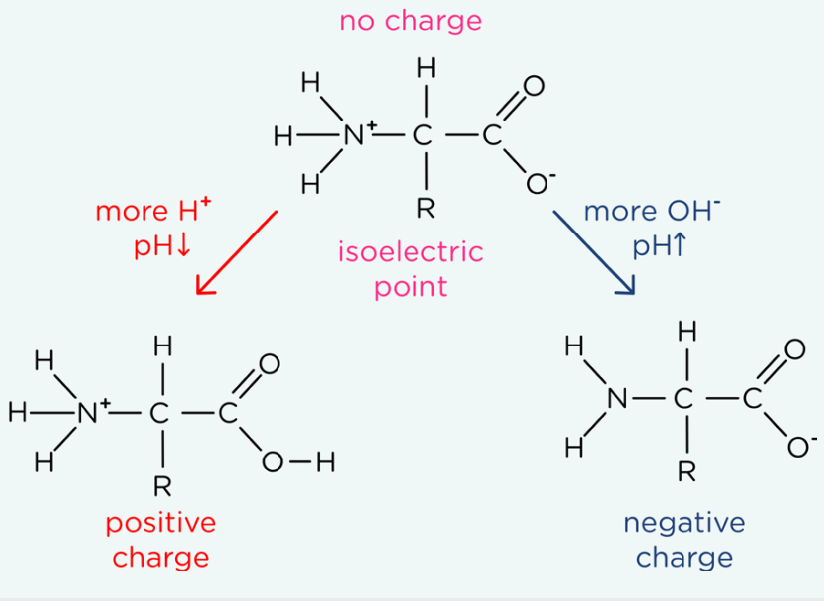
The acidic and basic properties of amino acids:
So, a Zwittern Ion can be formed:
The carboxylic group has a tendency of loosing a proton: Basic property, gaining a negtive charge
The amine group has a tendency of gaining a proton: Acidic property, gaining a positive charge
Since both groups are present then the amino acid has an overall neutral charge
Because they are ionic, amino acids have a high melting point and dissolve well in water, but poorly in non-polar solvents. A typical amino acid is white solid at room temperature, and behaves as an ionic salt.
When there is strong acidic conditions:
The amine group ONLY gains a proton, PROTONATED, forming overall positive charge
When there is strong alkaline conditions:
The carboxylic group ONLY looses a proton, DEPROTONATED, forming overall negative charge
When there is two amine groups, make sure that both of them gain a proton (amine in the R groups sometimes)
Everything about how two amino acids join:
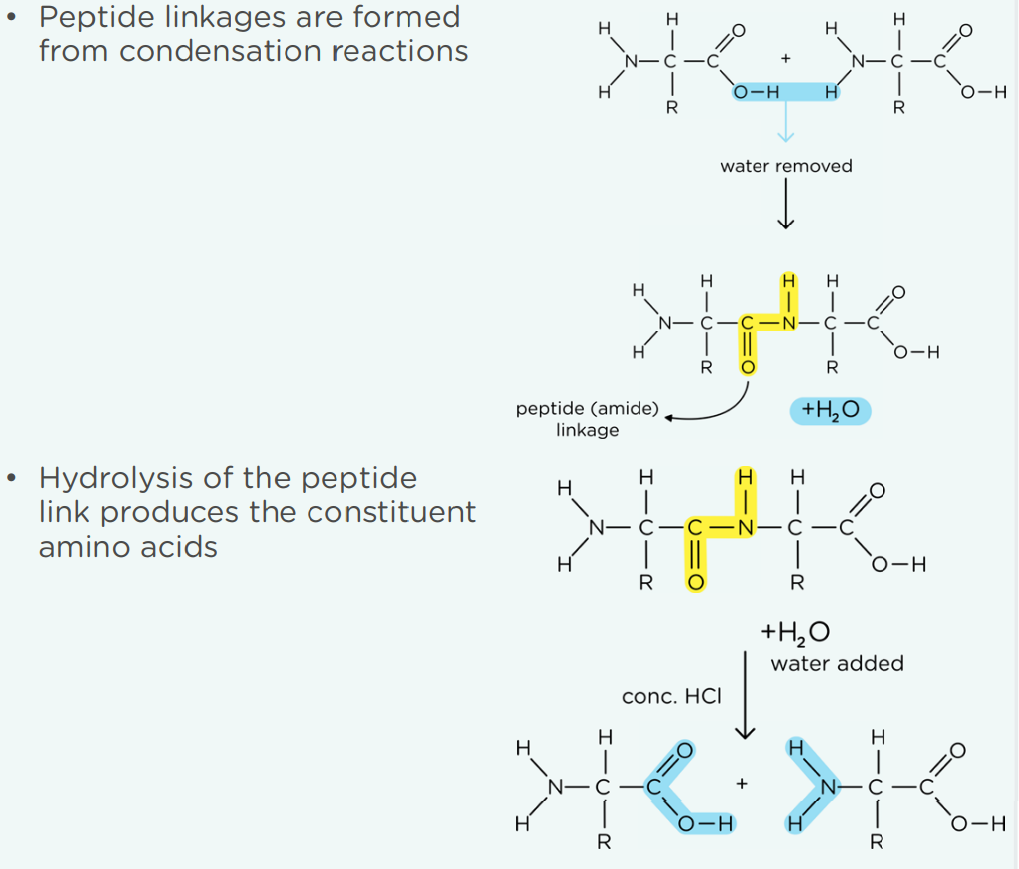
(Hydrolysis: when boiled with conc HCl, all peptide links are hydrolised)
Define: Primary structure, secondary structure, tertiary structure and quaternary structure:
Primary structure: The sequence of amino acids, held together by peptide bonds
Secondary structure: Folding of polypeptide chain into local structures, mostly alpha helix or beta pleated sheets, held together by hydrogen bonding between the C=O of one peptide and N-H of another
Tertiary structure: The overall 3-dimensional shape of a single polypeptide, maintained by interactions between R-group (hydrogen, ionic, sulfur-sulfur, (van der vaals))
Quaternary Strucuture: Multiple polypeptide chains to form complete functional protein, may also contain some prosthetic groups (non-protein groups), bonding that holds the chain together is the same as the tertiary structure.
How to draw the bonding between R-groups, the hydrogen// ionic// sulfur-sulfur
Hydrogen bonding: Between the C=O and the N-H
Ionic interactions: COO- and -NH3+
Disulfide bridges: When amino acid cystenine has the group -CH2SH, where under suitable oxidising conditions, two cysteine molecules react to form a double amino acid cystine and water. They are the strongest because they from a covalent bond (1 mark in mark scheme)
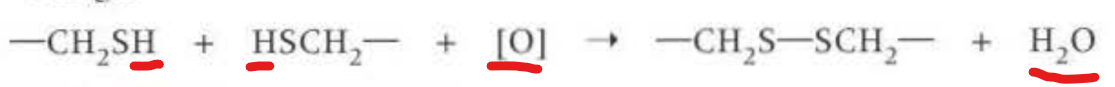
Everything about enzymes in chemistry:
Enzymes are proteins, they are biological catalysts that speed up the rate of reaction without being used up.
Enzymes’ have an active site which reacting molecules fit the active site, AND must bond to the active site temporarily by intermolecular forces. While being bonded, these forces promote the movement of electrons within the substrate - which lowers the activation energy for the reaction.
Enzymes are so specific to particular molecules, where the enzymes can only bind to one or other of a pair of enantiomers - which are called sterioisomers. So, enzymes are called stereospecific.
Ways to stop enzyme’s reaction:
Inhibiting blocking the active site
Denaturing the enzymes: changing pH and temperature
Everything about DNA from Chemistry POV
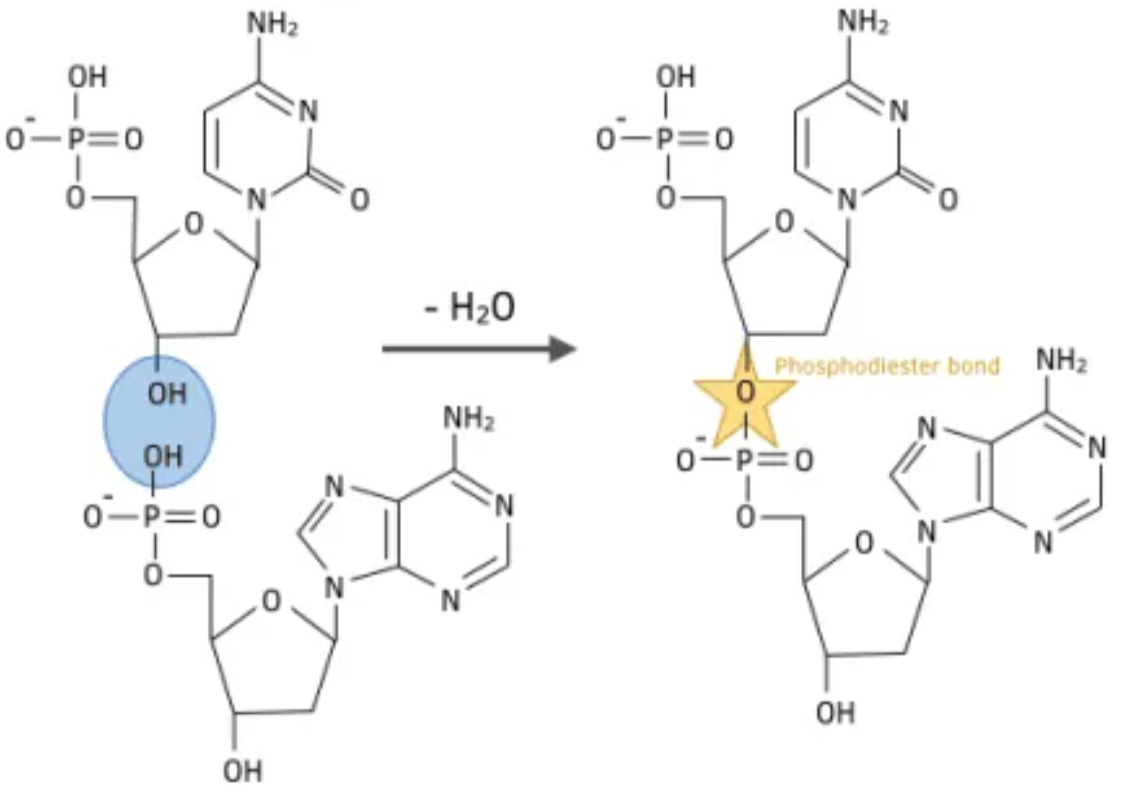
DNA is a polymer of nuculotides. Double helix, held together by hydrogen bonding between the bases. A-T // C-G
Nucleotide: Phosphate ion, deoxyribose sugar and a base.
How nueclotides join to each other? Via condensation reaction
H from the -OH of Phosphate
OH from the OH of the sugar
This forms water
The action of Anti-cancer drug:
When DNA replication goes wrong, results in a cancerous cell - leading to uncontrollable cell division.
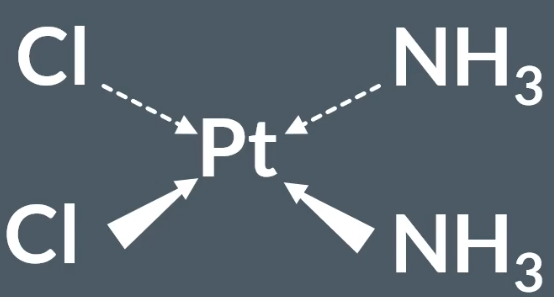
Chemotherapy: contains the drug Cisplatin - transition metal complex.
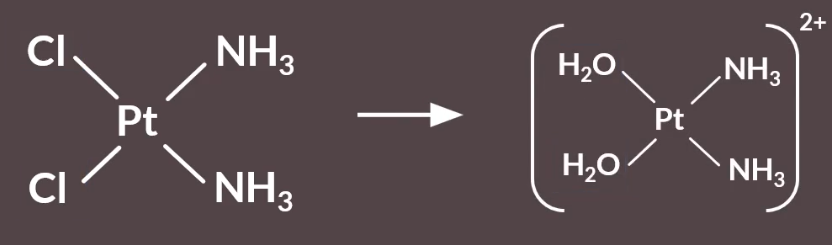
When Cisplatin first enters the cell, the chloride ligands are replaced by water ligands, via a ligand substitution reaction.
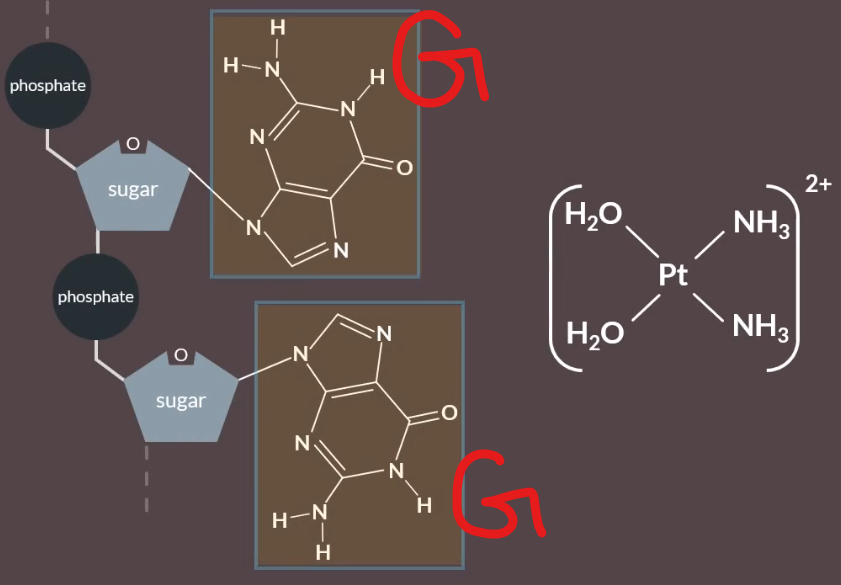
Then where there is two guamines next to each other, the nitrogen atoms are replaced by the water ligands.
This causes a kink or a distorted shape of the DNA, that stops cell division.
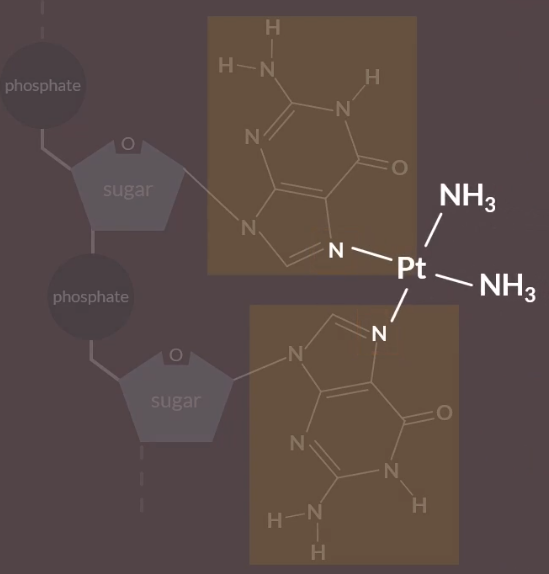
However, the action of these drugs can also harm healthy DNA cells, which can cause side effects, such as weight-loss, vomitting and hairloss.
The side effects can be reduced, by only targetting the drug to a localised area, and using smaller dosage.