biochem exam 2
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
enzymes catalyze reactions under what conditions?
mild conditions - 37 degrees C, 1 atm, PH 7, aqueous environments
where is fumerate reaction located
in the mitochondrial matrix
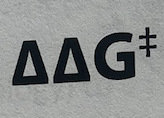
Rate enhancement =
RE, the amount of free energy needed to enhance rate of rxn, change in G uncat - change in G cat
If G reactants > G products…
(-) G for reaction
If G reactants < G products…
(+) G for reaction
what does rate enhancement mean in words
if the catalyst provides x amount of free energy we can enhance the rate by __ times.
Effect of change in G on RE
small changes in change in G can have a large change on the rate
with a fixed amount of enzyme why does the curve flatten off
system becomes saturated because enzyme is fixed and there is way more substrate, enzyme active site becomes saturated
What is the steady state assumption
the amount of ES does not change over the course of the reaction, allows us to set the formation of ES and the breakdown of ES equal. Important for michaelis-menten equation
for michaelis menten what part of the reaction is monitored
only the initial reaction velocity
What does Km stand for
michaelis constant - measures the stability of the ES complex - low Km = forms complex more readily (good)
What does Kcat stand for
Rate constant for conversion of ES to E + product, called the turnover number for the reaction - # of products formed per time interval (sec -1)
Most useful comparison of enzyme reactions
Kcat/Km - specificity constant
enzyme with highest affinity to substrate
lowest Km
enzyme and substrate with greatest turnover # or products formed per time interval
highest Kcat
Catalytically perfect enzyme requirements
K cat needs to be in sec-1
Km has to be in molarity (M)
Kcat/Km = 10^8 - 10^9
what is michaelis menten plot used for
determining kinetic constants from data
Substrate (x) vs velocity (y)
What is the lineweaver burk plot used for
allows visualization of trends in Km and Vmax
1/substrate (x) vs 1/velocity (y)
What does the slope represent in lineweaver plot
slope = km/vmax
what does y intercept represent in lineweaver plot
y int = 1/vmax
what does x intercept represent in lineweaver plot
x int = -1/Km
which points are most reliable in lineweaver plot
x and y int, this are near the high substrate points which are the most accurate
slope is not as accurate because it is highly affected by points to the far right that are low substrate with the highest amount of error
atorvastatin
commonly prescribed to slow biosynthesis of cholesterol in the liver
used to treat high lipid levels in the blood and prevent cardiovascular disease
competitive inhibitor
Reversible inhibition
noncovalent (reversible covalent) - not fixed
noncooperative enzyme
inhibitor can bind to enzyme alone (E), ES complex, or both
Reversible inhibition - inhibitor binding to enzyme alone
Ki - inhibitor dissociation constant for the EI complex
the lower the Ki the tigher the binding between enzyme and inhibitor
Reversible inhibition - inhibitor binding to ES complex
Ki’ - inhibitor dissociation constant for ESI complex
lower the Ki’ the tighter the binding between ES complex and inhibitor
what should inhibitors do to Vmax
Vmax should always decrease with inhibitors
competitive inhibition - from weak to strong
molecules that interact with residues around or in active site
substrate analogs - used to understand interactions between substrate and enzyme
transition state and intermediate analogs - used to understand the catalytic pathway
uncompetitve inhibition
inhibitor binds only ES or EP complex -stops turnover
no way to minimize effect of inhibition
higher substrate = more inhibition
Mixed inhibition
inhibitor binding site is not part of active site - can bind both E and ES leading to mixture of competitive and noncompetitive
binding causes conformational change, inactivates enzyme
How to recognize competitive
v max is the same (y intercept is the same)
Km increases - moves toward positive x axis
How to recognize uncompetitive
parallel
Km and vmax drop proportionally
how to recognize mixed competitive
binds with higher affinity to E than ES
a > a’
V max is smaller (y int)
Km is bigger
how to recognize mixed non competitve
binds with equal affinity to E and ES
a = a’
vmax is smaller
Km = Km control
how to recognize mixed uncompetitive
binds with lower affinity to E than ES
a < a’
vmax is smaller
Km is smaller - not proportional