Chemistry Unit 2 Exam
1/47
Earn XP
Description and Tags
Chapters 4 & 5
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
Synthesis Chem. Rxn
Type of chem. rxn
2 components → one product
A + B → AB
Decomposition Chem. Rxn
Type of chem. rxn
1 compound → 2 products
AB → A + B
Single Replacement Chem. Rxn
Type of chem. rxn
Nonmetals switch
AB + C → B + AC
Double Replacement Chem. Rxn
Type of chem. rxn
Metals switch partners
AB + CD → AD + CB
Solutions
Homogenous mixtures of 2+ pure substances
Solvent
Present in greatest abundance in a solution
All other substances are solutes
Aqueous Solution
When water is the solvent
Dissolving Solutes
There are attractive forces between solute particles holding them together
There are attractive forces between solvent molecules
When mixing solute w/ solvent, there are also attractive forces between solute particles and solvent molecules
If the attractions between solute and solvent are greater than the other two, the solute will dissolve
Aqueous Solutions and Dissolving Solutes
All substances dissolve by solvation (surrounding of solute by solvent)
Ionic compounds dissolve by dissociation, where water surrounds separated ions
Molecular compounds disperse in water, but mostly remain intact
Some molecular substances form ions in water when they dissolve
Don’t break into individual atoms but stay as a single unit
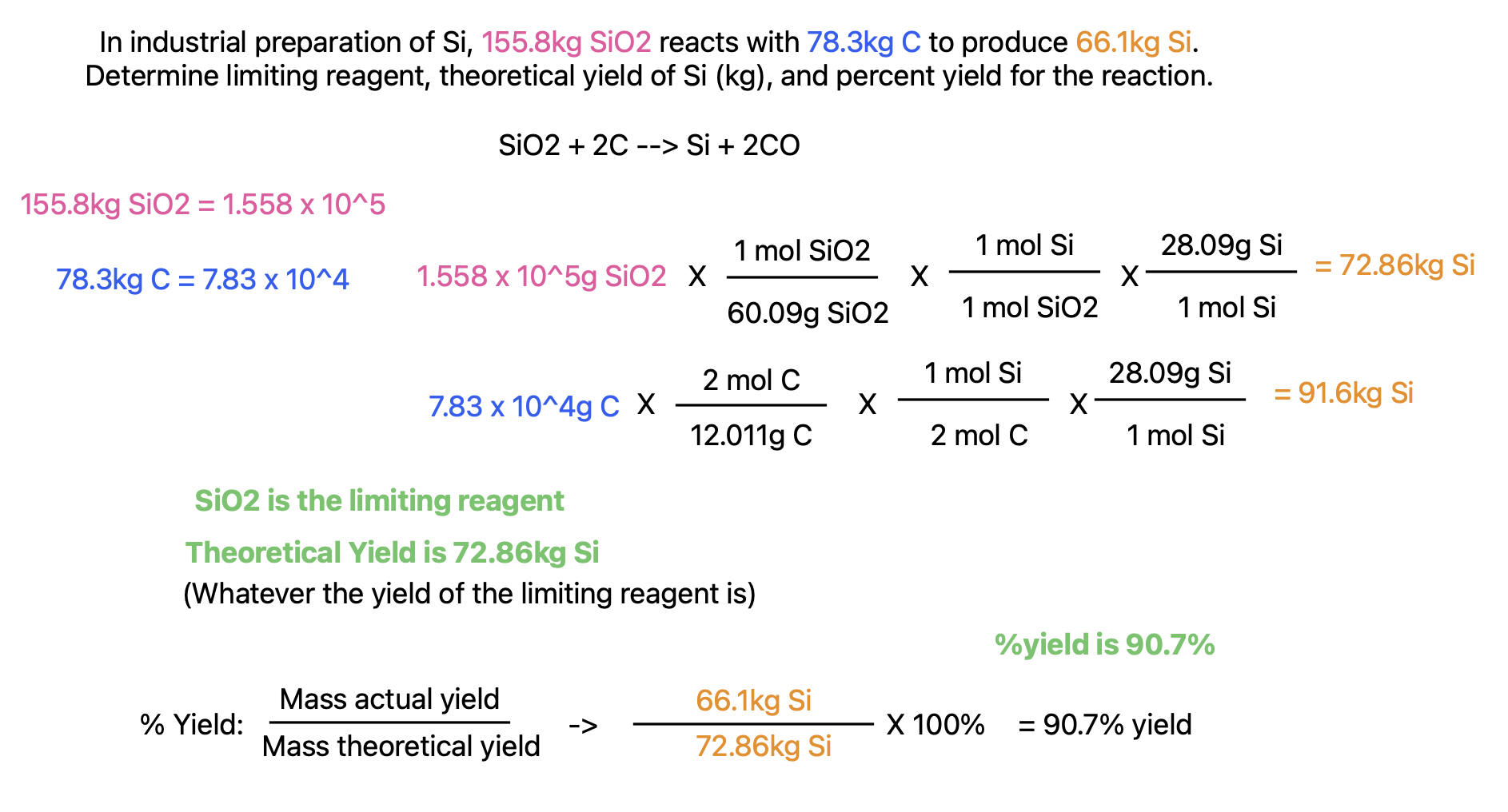
Limiting Reagent
In most chemical rxns, one substance will be limiting to the reaction.
Idea: if you have 3 bags of flour but 2 eggs, the eggs will limit how many pancakes you can make, while the flour is in excess
Typical Process:
Find mol of product produced with reactant 1
Find mol of product produced with reactant 2
etc….
Which produces a small #mol of product is the limiting reagent.
To find how much excess is actually used, perform stoichiometry from limiting reagent, molar mass, molar ratio, to find actual mol or g of other reactant that was used.

Theoretical, Actual, and & Percent Yield
Theoretical yield is how much of a product you would theoretically have after a chem. rxn
Actual yield is how much of a product you actually produce
Percent yield is the percent of the theoretical yield that was actually produced: (mass actual yield) / (theoretical yield) x 100%
Charge Distribution in Water Molecules
Uneven distribution is e- within molecule
Causes oxygen to be partially neg. and hydrogens partially pos.
Makes water molecules polar
LIKE DISSOLVES LIKE
Non-polar solvents dissolve non-polar solutes
Polar solvents dissolve polar solutes
Electrolytes
Dissociate: break off into smaller ions or atoms
Electricity: flow of free e-
Strong electrolytes: dissociate completely into ions when dissolved in H2O; conducts electricity
ex. NaCl
equation has single arrow
Weak electrolytes: dissociates partially when dissolved in H2O; cannot conduct (not enough charged particles)
equation represents chem. equilibrium w/ equilibrium arrow
Non-electrolytes: DOES NOT DISSOCIATE in H2O (may dissolve, not dissociate); does NOT conduct
Sugar
Acids ionize in H2O to varying degrees
Completely ionize: strong acids
Don’t completely ionize: weak acids
Electrolytes and Solubility
Ionic compounds that are SOLUBLE are STRONG ELECTROLYTES
Ionic compounds that are INSOLUBLE are WEAK ELECTROLYTES
When determining strength of electrolyte, look at solubility rules
Weak and Strong Acids and Bases
Strong Acids:
HCl
HBr
HI
HNO3
H2SO4
HClO3
HClO4
(SO I BRought NO CLean CLOthes)
SO4, I, Br, NO3, Cl, ClO3, ClO4
H2SO4, HI, HBr, HNO3, HCl, HClO3, HClO4
Strong Bases:
LiOH
NaOH
KOH
RbOH
CsOH
Mg(OH)2
Ca(OH)2
Sr(OH)2
Ba(OH)2
(Like NAthan Knows, CAlifornia is SupeR BAsic)
Li, Na, K, Ca, Sr, Ba
LiOH, NaOH, KOH, Ca(OH)2, Sr(OH)2, Ba(OH)2
+ RbOH, CsOH, Mg(OH)2
(Main idea: most group 1A and 2A cations)
Weak Acids:
Not strong acids
Weak Bases:
No OH group
Empirical Method
Conducting experiments and creating rules based on observations
Solubility of Ionic Compounds
When an ionic compound dissolves in water, the resulting solution contains component ions in solution, not intact ionic compound
Some ionic compounds are insoluble, and do not dissolve in water
Ionic compound in solution:
Precipitate is an insoluble ionic compound, which does not dissolve in water
Can be in suspension (floating) or collected at bottom
Supernate is liquid above collected precipitate at bottom
Solution (when no insoluble products present) can be colored, but always clear
Precipitation Reactions
Reaction that yields an insoluble product (precipitate) when 2 solutions are mixed
Reactions between aqueous solutions of ionic compounds produce insoluble ionic compounds
No precipitation means no reaction
Predicting Precipitation Reactions
Determine which ions constitute each aqueous reactant
Determine formulas of possible products
exchange ions and balance charges of combined ions to get formula of each product
Determine solubility of each product in water
If neither products will precipitate, write no reaction after arrow ( → NR)
If any possible products are insoluble, write formulas
Balance equation
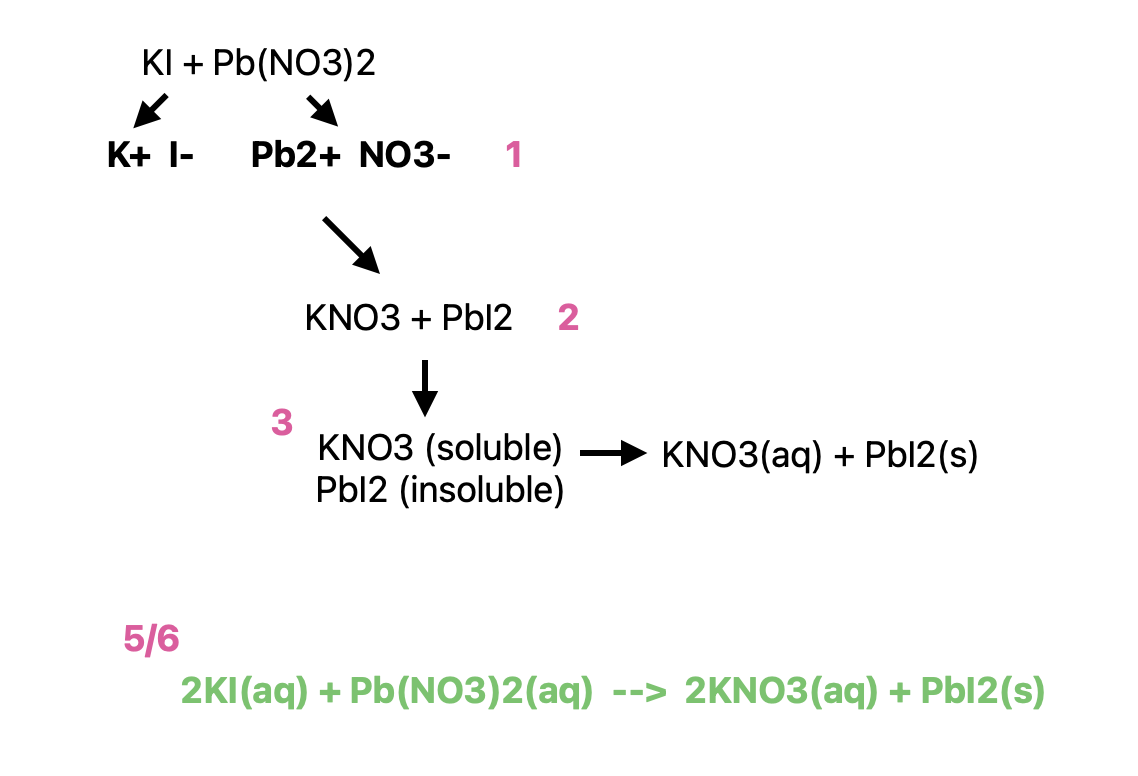
Molecular Equations, CIE, & NIE of Precipitation Reactions
Molecular Equations: the original equation after determining solubility and balancing equation
Complete Ionic Equation: separate all parts into their ions w/ solubility & charges (ex. KI(aq) (ME) = K+(aq) + I-(aq) (CIE))
Net Ionic Equation: only include ions from CIE that are directly involved in producing the precipitate product
Remove spectator ions (those not involved in formation of the precipitate)
NOTE**: precipitate never written out into its ions since the ions don’t dissociate at all
Aqueous Reactions
Precipitation Reactions
Acid-Base Reactions
Gas Evolution Reactions
Anion from one reactant combines with cation of another
Acid-Base Reactions
Also “neutralization reaction”
Acid reacts with base and they neutralize each other, producing water (or sometimes a weak electrolyte)
In rxn of an acid & base:
H+ from acid combines w/ OH- from base to make water
Cation from base combines w/ anion from acid to make a salt
NIE: H+(aq) + OH-(aq) → H2O(l)
(as long as salt that forms is soluble)
Hydronium Ions in Solution
Acids ionize in water to form H+ ions
More precisely, the H+ from the acid molecule is donated to a water molecule to form hydronium ion (H3O)+
Most chemists use H+ and H3O+ interchangeably
Polyprotic Acids
More than 1 ionizable proton and release them sequentially
First ionizable proton strong, and subsequent ones are weaker
Ex. H2CO3, H3PO4, H2SO4, etc
more than one H in compound
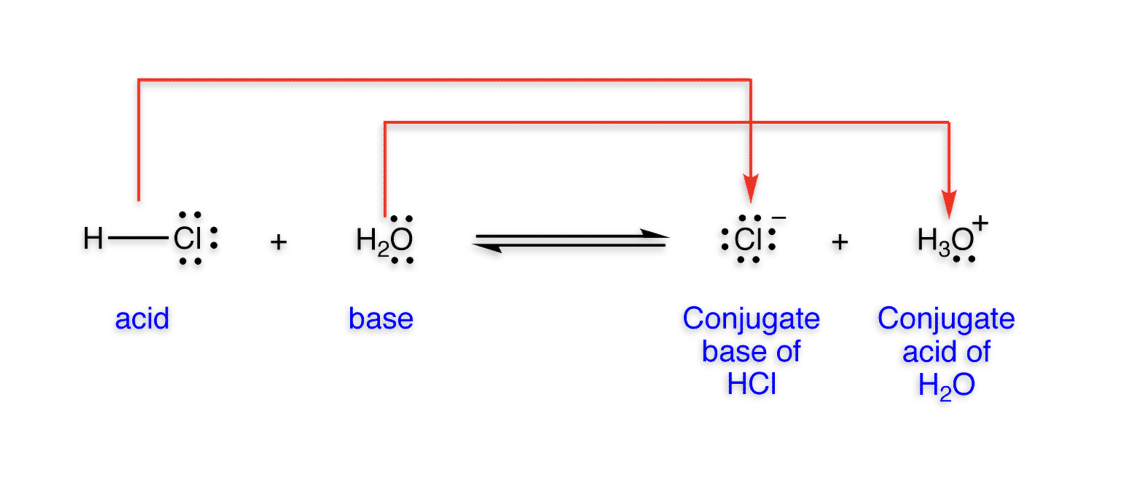
Arrhenius Definition
Acid: substance that produces H+ in aq solution
Base: substance that produces OH- in aq solution
Bronsted-Lowry Definition
Acid: substance that can donate a proton
Base: substance that can accept a proton
Water is amphoteric; it acts either as an acid or base
Solution Concentration
Since solutions are mixtures, composition can vary one sample to another
We quantify the amount of solute relevant to the solvent, or concentration of solution
Common way to express solution concentration is with Molarity
M= (mol solute)/(L ENTIRE solution)
Can be used as a conversion factor
Solution Dilution Equation
Used when stock solutions (concentrated) need to be diluted, meaning more solvent is added
Amount of solute doesn’t change, just volume
M1V1 = M2V2
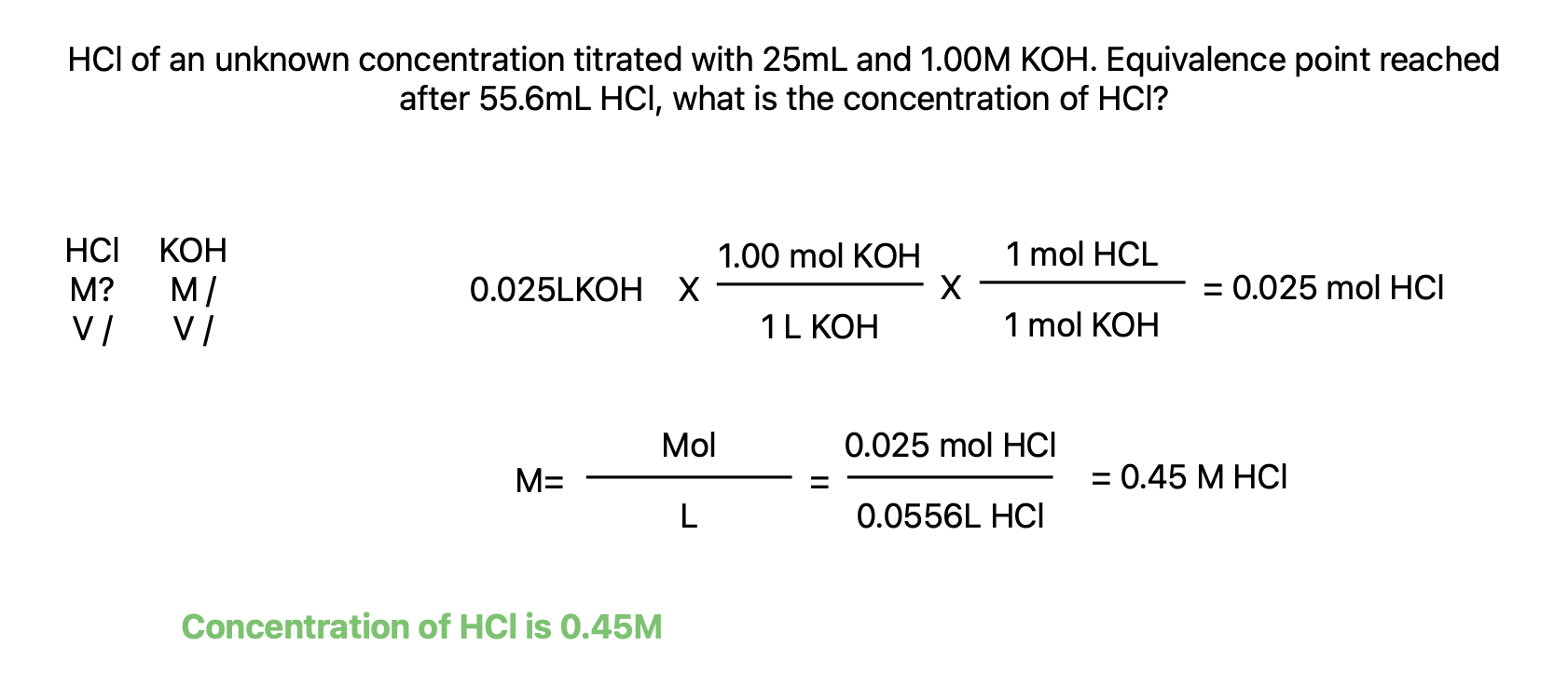
Acid-Base Titrations
Titration: lab procedure where substance in solution of known concentration (titrate) is reacted with another substance of unknown concentration (analyte)
Equivalence point is point in titration where H+ and OH- from reactants are in their stoichiometric ratio and are completely reacted
An indicator is a dye whose color depends on acidity or basicity of solution
Gas Evolution Reaction
Gas is produced, results in bubbling
Many GE rxns are AB rxns
Some rxns form a gas directly from ion exchange
Others form gas through subsequent decomp of one of the ion exchange products into a gas and water
The product that undergoes decomp is the intermediate
Types of Compounds That Undergo GE Reactions
Sulfides
No intermediate product
H2S gas eveolved
Carbonates/Bicarbonates
H2CO3 intermediate
CO2 gas evolved
Sulfites/Bisulfites
H2SO3 intermediate
SO2 gas evolved
Ammonium
NH4OH intermediate
NH3 gas evolved
Redox Reactions
Electron transferred from one reactant to another
Many involve the reaction of a substance with oxygen
Electron transfer does not need to be complete transfer to qualify as a redox rxn
Can be unevenly shared
Ex. Lower e- density in H and higher e- density in Cl
Oxidation: loses e-
Reduction: gains e-
(OILRIG)
Oxidized component loses e- and reduced component gains e-
Oxidized component is reducing agent; reduced component is the oxidizing agent
Redox rxns are:
Any species that changes its oxidation state
Any metal + non-metal reacting
Sing-replacement rxns
MOST synthesis/decomposition rxns
NOT double replacement or A-B rxns
Oxidation States
Imaginary # assigned to elements to determine loss/gain of e-
Oxidation # of an atom in a compound is the “charge” it would have if all e- were assigned to the atom with the greatest attraction for those e-
Oxidation states are imaginary charges assigned w/ rules, and written magnification then charge (ex. +1, -1, etc)
Note: ion charges are real, measurable charges, and written charge before magnitude (ex. 1+, 1-, etc)
Rules:
Free elements have oxidation state of 0 (ex. Cu=0, Cl2=0)
Monatomic ions have oxidation state equal to charge (ex. Ca2+=+2, Cl-=-1, etc)
3a. Sum of oxidation states of all atoms in a compound is 0 (ex. H2O, H=+1, O=-2, together=0)
3b. Sum of oxidation states of all atoms in a polyatomic ion equals charge of ion (ex. NO3-, N=+5, O=-2, together=-1)
4a. Group 1 metals have oxidation state +1 in all compounds (ex. Na=+1 in NaCl)
4b. Group 2 metals have oxidation state +2 in all compounds (ex. Mg=+2 in MgCl2)
In their compounds, non-metals have oxidation states according to table (higher on top, higher priority):
Nonmetal | Oxidation State | Example |
Fluorine | −1 | M g F 2 −1 o x state |
Hydrogen | +1 | H 2 O +1 o x state |
Oxygen | −2 | C O 2 −2 o x state |
Group 7A | −1 | C C l 4 −1 o x state |
Group 6A | −2 | H 2 S −2 o x state |
Group 5A | −3 | N H 3 −3 ox state |
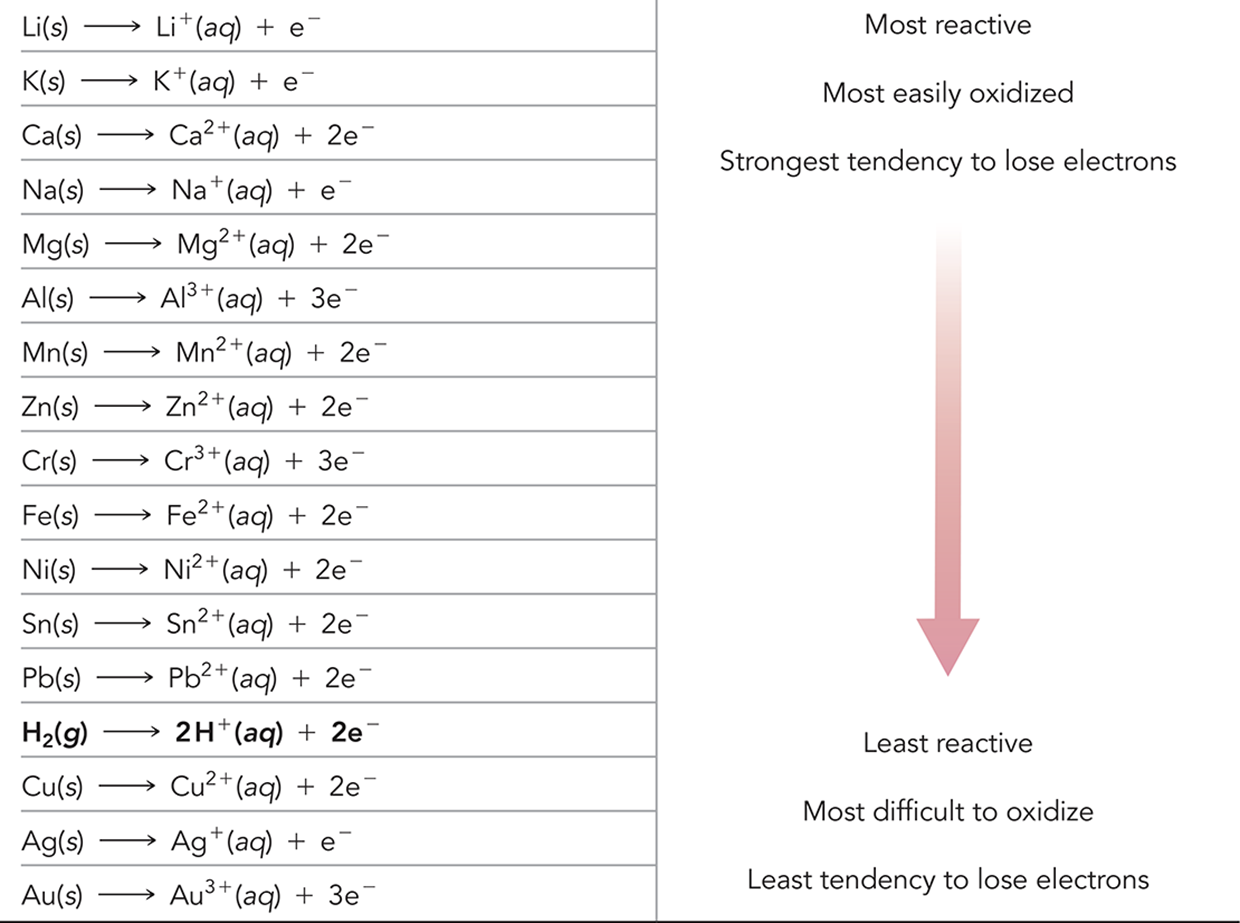
Activity Series
Top is most reactive and has highest tendency to undergo oxidation
Decreasing position on list, less reactive
Half-rxns at top most likely to occur in forward direction
Half-rxns at bottom most likely to occur in reverse direction
Kinetic Molecular Theory (KMT)
Simplest Model For Behavior of Gases
Postulates of Ideal Gases:
Gas particles spread out and so small relative to overall volume that their size and volume and negligible
Gas particles are in constant random motion
When gas particles hit one another or the walls, collisions are elastic (no KE lost)
Assumer there are no attractions between gas particles (just bounce off one another)
Average KE (speed) of gas particles is directly proportional to Kelvin temperature
Not all particles moving at same speed (due to different masses), but avg KE increases as temperature increases
Negligible
RM
Elastic
Proportional
Attraction
KMT-PropANERM
Pressure
Pressure: force exerted per unit area by gas molecules as they strike surfaces around them
Result of many gas molecules exerting forces on surfaces around them is a constant pressure
Pressure is (Force)/(Area), F/A
Gas pressure is result of constant movement of gas molecules and their collisions w/ surfaces around them
Pressure of gas depends on:
# gas particles in given volume
volume of container
average speed of gas particles
Total pressure exerted by gas depends on several factors including concentration of gas molecules
Higher the concentration, higher the pressure
As volume increases, concentration decreases (#particles is same, but concentration is decreasing), this results in less collisions and therefore lower pressure
Fewer gas particles, lower force per unit area, and so lower pressure too
Higher density of particles, higher pressure; lower density, lower pressure
Gas pressure and temp. impact how gases behave
Common Pressure Units
Atmospheres - - - - - - - - - - atm - - - - - 1atm (avg air pressure at sea level)
Pounds per square inch - - - psi - - - - - - 14.7psi
Torr - - - - - - - - - - - - - - - mmHg/torr - - 760torr or 760mmHg
Inches of mercury - - - - - - - inHg - - - - - 29.92inHg
Pascal (1N/m²) - - - - - - - - - (Pa) - - - - - - 101,325Pa
Ideal Gas Law
PV=nRT
P= pressure (atm)
V= volume (L)
n= # moles (mol)
R= gas constant; 0.08206 (L x atm)/(K x mol)
OR 8.314 (J)/(mol x K)
OR 8.314 (L x kPa)/(mol x K)
T= temperature (K)
Can derive simple gas laws (which dictate behavior of gas if 2 variables change and others remain constant)
Simple Gas Laws
Describe relationships between pairs of properties involved with gases
Properties interrelated
Boyle’s Law: P1V1 = P2V2
Charles’s Law: V1/T1 = V2/T2
Gay-Lussac’s Law: P1/T1 = P2/T2
Combined Gas Law: (P1V1)/T1 = (P2V2)/T2
Avogadro’s Law: V1/n1 = V2/n2
Standard Temperature and Pressure (STP)
1 atm
273K
ANY gas at STP, molar volume is 22.4L (1mol=22.4L)
Molar volume: V=(nRT)/P; and w/ STP, n=1mol, T=273, and P=1atm, meaning V=273
Density of a gas
Tells you how “heavy” a gas is
d=(PM)/(RT)
M=(dRT)/P
Dalton’s Law of Partial Pressure
Ptotal = Pa + Pb + Pc +…
Pa = X(mole fraction)a + Ptotal
Pn = nn(RT/V)
Average Kinetic Energy
KE=(1/2)mv²
Avg KE = (3/2)RT
R= 8.314 (J/Kxmol)
Average KE is only reliant upon temperature, nothing else
Since there are different masses depending on the type of gas, the only way to keep the same avg KE is if lighter particles move faster while slower particles move slower. This allows gases to have the same avg KE. This can be seen through examples of the equation KE=(1/2)mv², because as m grows, v is smaller.
Root-Mean-Square Velocity
Avg speed of gas particles; depends on molar mass
At same T, all gases have same avg KE, but this is because they have different speeds.
Lighter molecules will move faster while slower particles move slower
Urms= sqrt(3RT/M)
Mean Free Path
Molecules in a gas move in a straight line until colliding
MFP is the average distance a molecule travels between collisions
So, as P increases, meaning the V decreases, MFP also decreases
Graham’s Law of Effusion
Diffusion: movement of gas molecules from an area of high concentration to an area of low concentration
Effusion: when a collection of molecules escape through a small hole in a vacuum, meaning the molecules are still traveling to areas of lower concentrations, but the process to getting there takes longer since molecules must escape through a small hole or a membrane first (meaning smaller particles escape quicker since they’re faster and smaller)
Law states that lighter particles move faster, and so they will escape quicker than heavier particles during effusion
r1/r2 = sqrt(M2/M1)
Real Gases and Ideal Behaviors
Real gases do not behave ideally under low T and high P
Smaller volume, so particles are closer together (KMT states particles so small relative to overall volume that they’re size and volume is negligible; this breaks down when particles are in small spaces (they take up more space))
Under low T, avg KE and Urms decrease, meaning particles move much slower. Since real gases can have polar molecules with intermolecular forces, this situation gives particles a better chance of attracting to one another since they move slower and will be close by for a longer period of time (KMT states that we should assume molecules do not have any intermolecular forces between them, but this breaks down if polar gas molecules are nearby and attract each other)