Organic Chemistry
IUPAC rules: prefix (substituents) - parent (# of carbons) - suffix (what family)
Alkanes
steps for naming alkanes:
- name the main chain (the longest continuous chain of carbons)
- number the carbon atoms in the main chain
- start at the end closest to the first substituent
- identify and number the branching substituent
- write the name as a single word
- use hyphens to separate different prefixes and commas to separate numbers
- list substituents in alphabetical order
properties of alkanes
- called paraffins since they don’t react as most chemicals
- will burn in a flame producing carbon dioxide, water, and heat
- react with Cl2 in the presence of light to replace H’s with Cl’s
- boiling points and melting points increase as size of alkane increases
- forces between molecules are weak
reactions of alkanes
cracking: large alkane + hydrogen gas → smaller alkane
reforming: small alkane → larger alkane + hydrogen gas
substitution (halogenation): replace H with a halogen atom
- initiated by addition of energy in the form of heat or ultraviolet light
combustion: hydrocarbon + oxygen → carbon dioxide + water
- all hydrocarbons undergo combustion
cycloalkanes - alkanes that have carbon atoms forming a ring
steps for naming cycloalkanes:
- count the number of carbon atoms in the ring and the number in the largest substituent chain
- if the number of carbon atoms in the ring is equal or greater than the number in the substituent the compound is named as an alkyl-substituted cycloalkane
properties of cycloalkanes
- melting points are affected by the shapes and way the crystals pack
- don’t change uniformly
isomers: different molecules with the same molecular formula
structural isomers: different pattern of atom attachment
Alkenes and Alkynes
alkenes: contain a double bond (C=C)
- one double bond = CnH2n
- have straight chains and are unsaturated
- polyunsaturated = many double bonds
- much more reactive than alkanes
steps for naming alkenes:
- find the longest continuous carbon chain containing the double bond
- identity the substituents
- number the chain from the end closest to the double bond
- write the name in the following order:
- substituents in alphabetical order
- number of first carbon double bond
- name of main chain
- end with -ene
alkynes: contain a triple bond
- one triple bond = CnH2n-2
- have straight chains and are unsaturated
- more reactive than alkenes
steps for naming alkynes:
- find the longest continuous carbon chain that contains the triple bond
- identify the substituents
- number the chain from the end closest to the triple bond
- write the name in the following order:
- substituents in alphabetical order
- number of first carbon triple bond
- name of main chain
- end with -yne
geometric isomerism: result of rotations around the double bond being highly restricted
- different molecules of groups have different spatial orientation about the double bond
- cis isomerism: groups are bonded on the same side
- trans isomerism: groups are bonded on opposite sides
Aromatic Hydrocarbons
aromatic hydrocarbons: contain a ring structure with a series of alternating single and double bonds in a delocalized arrangement
- in simple aromatic compounds, the benzene ring is the parent chain
- if the attached benzene group is not easily named the benzene ring is the attached branch, called the phenyl group
numbering carbons on the benzene ring:
- 1,2 - ortho (o)
- 1,3 - meta (m)
- 1,4 - para (p)
Reactions of Hydrocarbons
substitution reaction: the hydrogen atoms in an alkane may be substituted by a halogen such as F2, Cl2, Br2
- F2 reacts vigorously with alkanes
- Cl2 and Br2 require heat or ultraviolet light to react
- Product is a halogenated alkane of an alkyl halide
addition reaction: adding a molecule across the multiple bond (for alkenes and alkynes)
hydrogenation: adding H2
- converts unsaturated molecule → saturated
- alkene/alkyne + H2 → alkane
halogenation: adding X2 where X = F, Cl, Br, I
hydrohalogenation: adding HX where HX is polar
- when adding a polar reagent to a double or triple bond the positive part attaches to the carbon with the most H’s
hydration: adding water
- converts unsaturated hydrocarbon → alcohol
Markovnikov’s rule: in the addition of HX to alkene, the H attaches to the carbon with the most H’s, and the X attached to the end with the most alkyl substituents
aromatic compounds: have chemical reactions between those of alkanes and alkenes
- undergo substitution reactions
- substitution reactions with cyclohexane and bromine occur much slower and requires addition of heat
Organic Halides
organic halides: a group of compounds commonly used as refigerants (CFC’s) and non-stick coating (Teflon)
- many organic halides are toxic and/or carcinogenic
properties of organic halides:
- the bonds between the carbon and halogens are more polar than those between carbon and hydrogen
- alkyl halides are more polar than their hydrocarbon parents
- they are more soluble in polar solvents than their hydrocarbon parents and have higher boiling points
- when a compound such as propane reacts with a halogen a mixture of compounds containing 1,2,3 or more halogens form
- the more halogenated a compound is the more polar it is
preparing organic halides:
- alkenes and alkynes readily add halogens or hydrogen halides to their double or triple bonds
- Markovnikov’s rule applies when hydrogen halides are reactants
- alkene/alkyne + hydrogen halide → organic halide
- compounds with benzene rings have a substitution reaction
- elimination reaction: a hydroxide ion is used to eliminate a hydrogen and halide ion from adjacent carbon atoms to form a double bond making an alkene
- alkane + OH → alkene + water + halogen
Alcohols and Ethers
alcohol: a water molecule with one of the hydrogen atoms replaced with an alkyl group (R - O - H)
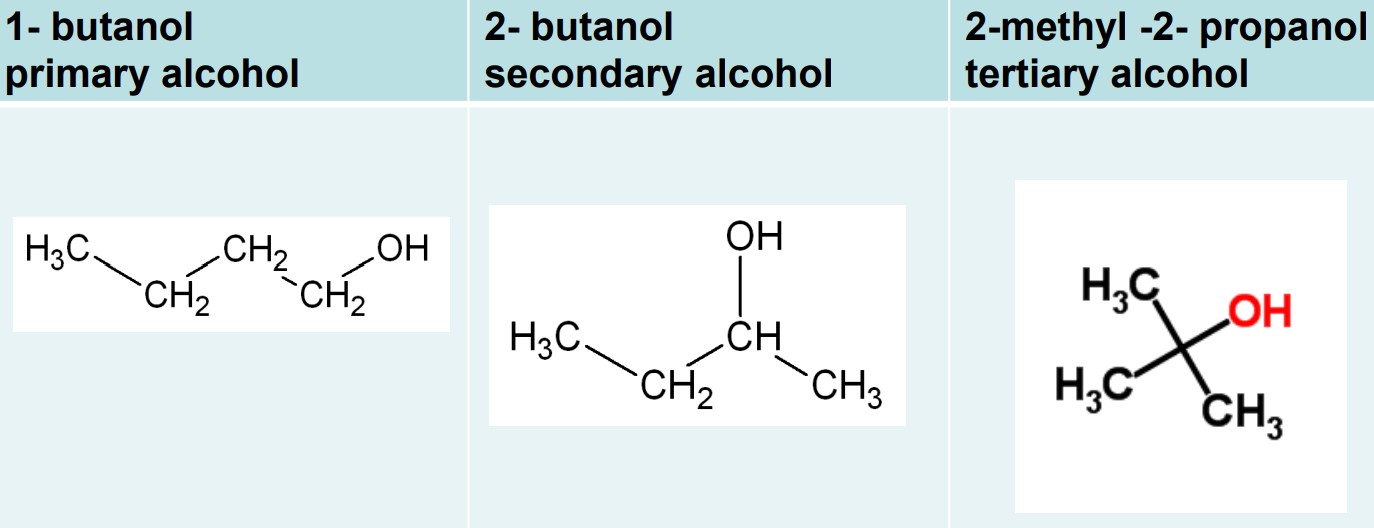
there are three classification alcohols:
primary alcohols: hydroxide is attached to an alkyl group attached to one other alkyl group
secondary alcohols: hydroxide is attached to an alkyl group attached to two other alkyl groups
tertiary alcohols: hydroxide is attached to an alkyl group attached to three other alkyl groups
cyclic alcohols: compounds containing cyclic alkanes or aromatic hydrocarbons attached to a hydroxyl group
properties of alcohols:
- much higher boiling points than parent alkanes
- more soluble than parent alkanes
- properties are due to H-bonding among molecules
reactions involving alcohols:
hydration: reacting alkenes with water in the presence of a catalyst results in an alcohol
- alkene + water → alcohol
- OH is found on the second carbon due to Markovnikov’s rule
combustion: alcohols undergo complete combustion to produce carbon dioxide and water
ethers: a water molecule with both of the hydrogen atoms replaced with alkyl groups (R - O - R) or (R - O - R’)
properties of ethers:
- don’t form hydrogen bonds since they lack an OH group
- more polar than hydrocarbons because of the dipole arising from their C-O-C bonds
naming ethers: add oxy to the prefix of the smaller hydrocarbon group and join it to the name of the larger hydrocarbon group
preparing ethers from alcohols:
- condensation: ethers are formed by the reaction of 2 alcohols and the elimination of a water molecule
- alcohol + alcohol → ether + water
Aldehydes and Ketones
aldehydes: consist of an alkyl group bonded to a carbonyl group with a hydrogen atom on the end
ketones: consist of two alkyl groups attached to a central carbonyl group
naming aldehydes and ketones:
- aldehyde: name ends with -al
- ketones: name ends with -one
- if the carbon chain has 5 or more carbon atoms a number is needed to indicate the location of the carbonyl group
properties of aldehydes and ketones:
- lower boiling points and less soluble than corresponding alcohols
- more soluble than corresponding alkanes
- aldehydes and ketones can mix with both polar and non-polar substances
- allows non-polar materials to be mixed with polar materials
preparing aldehydes and ketones from alcohols by oxidation reaction:
- oxidation: reactions involve a loss of electrons
- the element or compound that loses electrons is oxidized
- the element or compound that gains electrons is reduced
- controlled oxidation of alcohols results in aldehydes and ketones
- the reactive oxygen atoms are supplied by oxidizing agents (O)
- when primary alcohol is oxidized, an H atom remained on the C atom and an aldehyde is produced
- primary alcohol + (O) → aldehyde + water
- when secondary alcohol is oxidized, the carbonyl group that forms is attached to two alkyl groups forming a ketone
- secondary alcohol + (O) → ketone + water
- tertiary alcohols don’t react since there’s no hydrogen available for oxidation
- hydration: hydrogen can be added to the carbonyl group in aldehydes and ketones
- high temperatures and catalysts are required for this reaction
- this is the reverse of the controlled oxidation of alcohols
Carboxylic Acids and Esters
carboxylic acid: characterized by the presence of carboxyl functional groups (R-COOH)
naming carboxylic acids:
- the carboxyl group is made up of a hydroxyl (OH) bound to the C atom on a carbonyl group
- the main chain is the longest chain containing the carboxyl group
- ends with -oic acid
- when naming multiple carboxyl groups the suffix -dioic is used
- when more than two carboxyl groups are present, all COOH groups may be named as substituents on the parent chain
- the parent chain doesn’t include the carboxylic atoms
properties of carboxylic acids:
- highly polar molecules
- the polarity of the carboxyl group makes carboxylic acids soluble in water
- chains longer than ten carbons insoluble in water
- higher boiling points than their corresponding alkanes
- smaller members are soluble in water, larger carboxylic acids are relatively insoluble
- conduct electricity
- react with organic bases in neutralization reactions
- short chain carboxylic acids are liquids at standard temperature
- long chain carboxylic acids are waxy solids
preparing carboxylic acid:
- when alcohol is mildy oxidized, an aldehyde is produced
- if this aldehyde is oxidized further a carboxylic acid is produced
- alcohol + (O) → aldehyde
- aldehyde + (O) → carboxylic acid
ester: similar to carboxylic acids but th eH atom in the acid is replaced with another alkyl group (R - COO - R)
naming esters:
- the name of an ester has 2 parts
- first part: the name of the alkyl group used in the esterification process
- second part: the name of the acid
- the ending of the acid name change from -oic acid to -oate
properties of esters:
- the presence of the carbonyl group make esters somewhat polar
- esters are less polar than corresponding carboxylic acids because they lack an OH group capable of H bonding
- they are less soluble in water with lower melting and boiling points than corresponding alcohols and carboxylic acids
- smaller esters are liquid at standard temperatures while longer esters are insoluble
reactions of esters:
- esterification: carboxylic acid + alcohol → ester + water
- alcohol acts as an organic base and carboxylic acid acts as an acid
- the ester formed is considered an organic salt
- hydrolysis: reverses esterification by reacting the ester with an acid or base
- ester + acid/base → acid + alcohol
- a bond is broken by the addition of water resulting in two or more products
Amines and Amides
amines: ammonia with one to all of its hydrogens substituted by alkyl groups
there are three classifications of amines:
- primary amine: one hydrogen is substituted with an alkyl group
- prepared by reacting ammonia with an alkyl halide
- ammonia + alkyl halide → primary amine + hydrogen halide
- secondary amine: two hydrogens are substituted with alkyl groups
- prepared by reacting a primary amine further with an alkyl halide
- primary amine + alkyl halide → secondary amine + alkyl halide
- tertiary amine: all three hydrogens are substituted with alkyl groups
- prepared by reacting a secondary amine further with an alkyl halide
- secondary amine + alkyl halide → tertiary amine + alkyl halide
naming amines:
- amines are nitrogen derivatives of an alkane
- amines can also be named as alkyl derivatives of ammonia
- diamines: molecules with 2 amino groups
- for secondary and tertiary amines include the N-prefix to show the substituted groups on the amine
properties of amines:
- primary and secondary amines are very polar due to the N-H bond which allows them to H-bond with each other
- tertiary amines don’t have N-H bonds and can’t H-bond
- higher boiling and melting points than similar-sized ethers and alkanes
- smaller amines are soluble in water
- N-C and N-H bonds are more polar
- amines share a lower boiling point than alcohols of similar size because N-H bonds are less polar than O-H bonds
when amines are created using alkyl halides, a mixture of primary, secondary, and tertiary amines result
- can separate the different amines through boiling point
amides: hydrocarbon that contains a carbonyl group bonded to a nitrogen atom
- similar to esters but the N atom replaces the O atom in the chain of an ester
- they are the backbone of all protein molecules
- in proteins amide bonds are called peptides
properties of amides:
- amides have a polar carbonyl group and amides with a least one NH group can form strong hydrogen bonds among themselves
- higher boiling points that their corresponding hydrocarbon derivatives
- weak bases that are insoluble in water
- low molecular weight amides are slightly soluble
- can be hydrolyzed in acidic or basic conditions to produce a carboxylic acid and an amine
naming amides:
- the first part of an amide’s name comes from the amine
- the second part of the name comes from the acid
- ends with the suffix -amide
- if one or more alkyl groups is attached to the N atom the upper case N is used to clarify the location
preparing amides:
- condensation: carboxylic acids react with ammonia or primary or secondary amines to produce amides
- carboxylic acid + ammonia → amide + water
- tertiary amines don’t undergo condensation reactions since they lack the extra H atoms needed to make water
- amides can also be made with primary amines and carboxylic acids by condensation reactions
- carboxylic acid + primary amine → amide + water
Synthetic Addition Polymers
polymer: made up of a group of monomers (usually 10 or more)
- monomer: a hydrocarbon derived molecule
- polymers may contain thousands of individual monomers, the subscript n is used to indicate the number of repeating units
polyethylene: a polymer of ethene
- under certain conditions alkenes undergo addition reactions with other alkenes
- the double bond in each alkene changes into a single bond freeing an unbonded electron to form a single bond with other ethene monomers
addition polymerization: consists of three stages (initiation, propagation, and termination)
- an initiating molecule with an unpaired electron forms a bond with one of the carbonatoms in the double bonded monomer
- this yields an unpaired electron on the other end of the monomer
- this electron can then form covalen bonds with another group
- the reaction continues/propigates
- the chain grows until two unpaired electrons combine forming a covalent bond that links the growing chains together
properties of plastics:
- plastics are chemically unreactive because chemically active unsaturated alkenes have been converted into unreactive saturated carbon chains
- the strong covalent bonds make the structure very stable
- most intermolecular forces are Van der waals forces but because the molecules are so large these forces are very strong
strengthening polymers with cross-linking:
- alkenes with double bonds (-dienes) are monomers for several polymers
- these polymers often end in -ene
- cross-linking occurs between -dienes because the second double bond allows for the formation of covalent bonds between different chains of monomers
Synthetic Condensation Polymers
dimers: formed with molecules with functional groups react with other molecules
condemnation polymers: when monomers join end to end in ester or amide linkages they form polyesters and polyamides
polyesters: a polymer of carboxylic acids and alcohols
- when condensation reactions are repeatedly used to join carboxylic acids and alcohols, a polyester is formed
- uses a dicarboxylic acid and a diol
- the acids donates the OH, the alcohol donates the H
polyamides: a polymer of carboxylic acids and amines
- they are condensation polymers consisting of many amides
- the monomers must contain carboxyl groups, amine groups, or one of each