Organic Chemistry
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
When finding hybridization always remember about what
resonance structures which determine hybridization
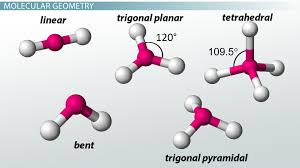
sp hybridization has what geometry and bond angle
linear and 180
sp2 hybridization has what geometry and bond angle
trigonal planar, 120
sp3 hybridization has what geometry and bond angle
tetrahedral, 109.5
Oxygen exists normally with how many bonds and lone pairs
2 bonds and 2 lone pair
Nitrogen exists normally with how many bonds and lone pairs
3 bonds and 1 lone pair
Phosphorous exists normally with how many bonds and lone pairs
3 bonds and 1 lone pair
Sulfur exists normally with how many bonds and lone pairs
2 bonds and 2 lone pairs
MEMORIZE
What are Lewis Acids
compounds that serve as electron acceptors
What are Lewis Base
compounds that serve as electron donors
What is Bronsted Lowry Base
are proton (H) acceptors
What is Bronsted Lowry Acid
proton (H) donors
The lower the pKa, the _acid
stronger
What are the 7 strong acids
HClO3,
HBr,
HCl,
HI,
HNO3,
HClO4,
H2SO4
High Ka means
high Ka means strong acid. High Ka means lower pKa value
High pKa means
low Ka value. Also means weakest acid
What happens to acidity as we go down a column in the periodic table
Acidity increases because atomic size increases
In an acid-based reaction, what kind of acid is favored for production?
the formation of a weaker acid is favored
For an amino acid, what happens to the amino and carboxyl group depending on how high or low pH is
If the pH is higher than functional group’s pKa, then carboxyl group will be in base form (COO-) no Hydrogen. If pH is lower than pKa, carboxyl group will gain proton COOH. If pH is greater than pKa, amino group will be the same (NH2). If pH is less than pKa, amino group will gain proton NH3+
If atoms like Fluorine group are closer to carboxylic acid what does this mean in terms of how strong the molecule is
Groups like Fluorine when close to carboxylic acid, are electron withdrawing groups so they will stabilize the negative charge of the conjugate base of the chosen molecule, resulting in stronger acid.
Which ones are stronger acid one with carboxylic acid or only alcohol group
Carboxylic acid because the conjugate base of carboxylic acids are MORE STABLE
A bigger atom does what to acidity
increases acidity
Electron withdrawing groups like F, Cl, Br, I do what to acidity
increase
What are some electron withdrawing groups resulting in increasing acidity
From Greatest to smallest:
nitro (NO2), (SO3H), cyano (CN), carbonyls (CHO, C=O, COOH, COOR), halogens,
When determining which molecule is the most acidic: Use CARDIO
C → carboxylic acid is strongest acid
A → atom size the bigger the size of atom the more acidic
R → resonance! must be able to be resonance stabilized or do resonance (SO no cyclic rings without double bonds inside)
DI → dipole induction. Which groups are the most electron withdrawing?
O → lok at orbitals. sp most acidic, then sp2, and sp3 is least acidic
Strong conjugate base means..
weak acid (low Ka, high pKa)
weak conjugate base means
strong acid (High Ka, low pKa)
strong conjugate acid means…
weak base
weak conjugate acid means
strong base
What makes a molecule more basic in terms of what kind of functional group must be there and characteristics?
electron donating groups like NH2, NHR, N, OH, OR are strongest, then NHCO-, OCO-, lastly weakest is Ch3, CH2CH3
Hydrogen bonding can happen intermolecularly between what kinds of molecules? Fundamental atoms in there that can allow this
Hydrogen (H) bonded to Nitrogen (N), Oxygen (O), or Fluorine (F).
IUPAC naming rules
try to number the carbons as long as you can (largest number of carbon) to incorporate as much as you can.
the functional group is named alphabetically in order which determines the lowest number assigned
assign the lowest number to the functional groups
Alkyne
triple bond. when naming for IUPAC want to assign this lowest number as possible. Numbering should go through it
-yne
Alkene
double bond. Try to assign lowest number. Counting should go through this.
-ene
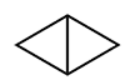
Name this
Bicyclo[1.1.0]butane
butane is parent name because there is 4 carbons
bicyclo in front of name

name this
spiro[3.4]octane
count all the carbons around the shape
establish parent name.
then count how many carbons are on opposite side of the vertices that both share.
label in brackets from lowest value to highest for spiro ones!!!
dont forget to put spiro in front
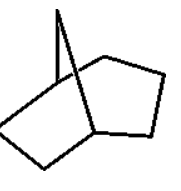
Name this
Bicyclo[3.2.1]octane
first name it bicyclo
then count how many total carbons and establish parent compound
then count how many carbons are NOT shared on either side separated by the bridge
then count how many carbons are shared in the bridge
label from descending value highest to lowest
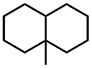
name this
1-methylbicyclo[4.4.0]decane
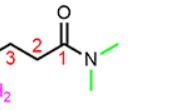
How do you name a molecule with functional group like this
We use “N,N-dimethyl” to indicate that there are two methyl groups bonded to the amide nitrogen.
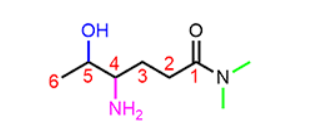
Where should you place carbon 1 on this molecule
On the carbonyl.
what functional group has the highest priority when the parent name ends in “-oic acid”
carboxylic acid
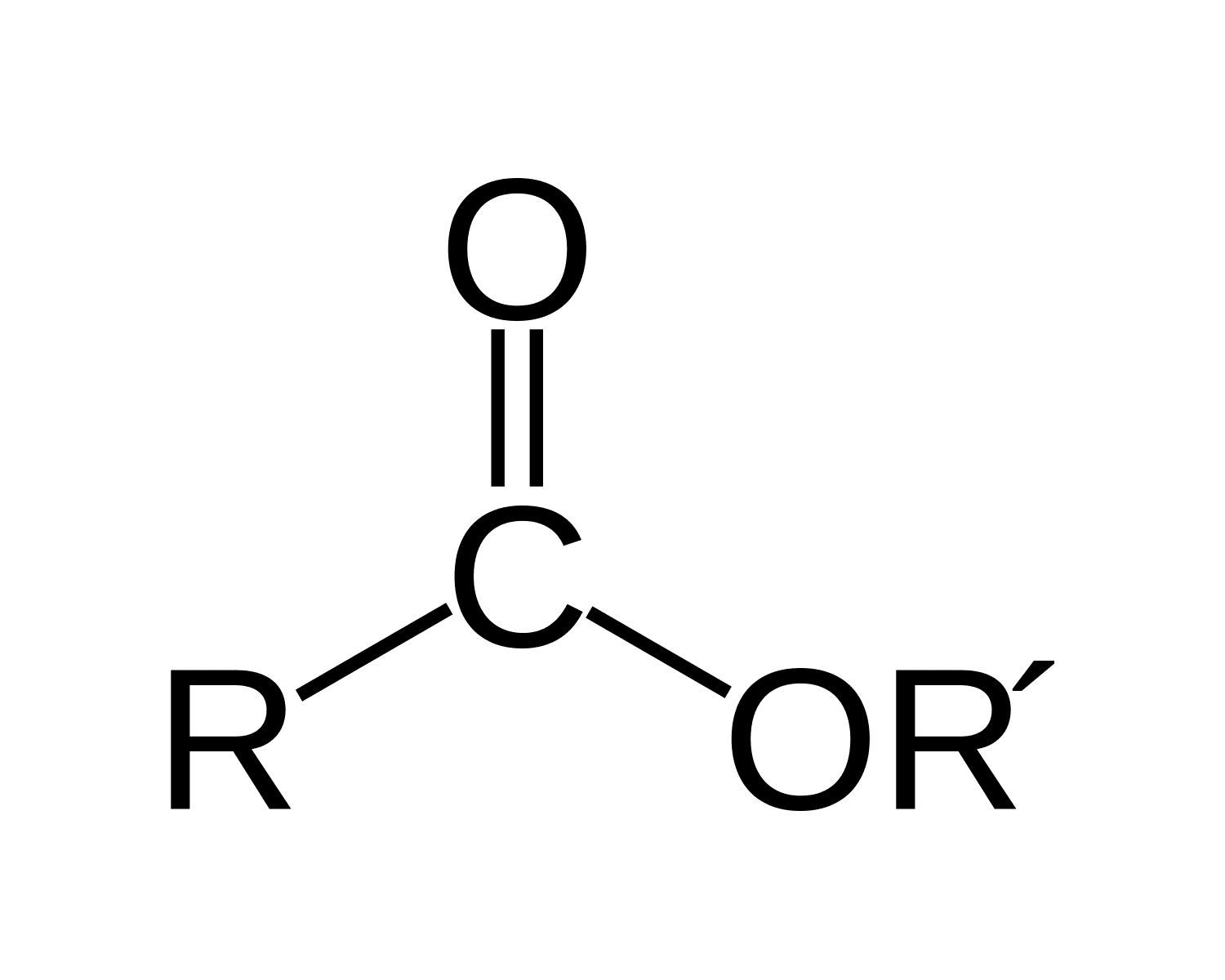
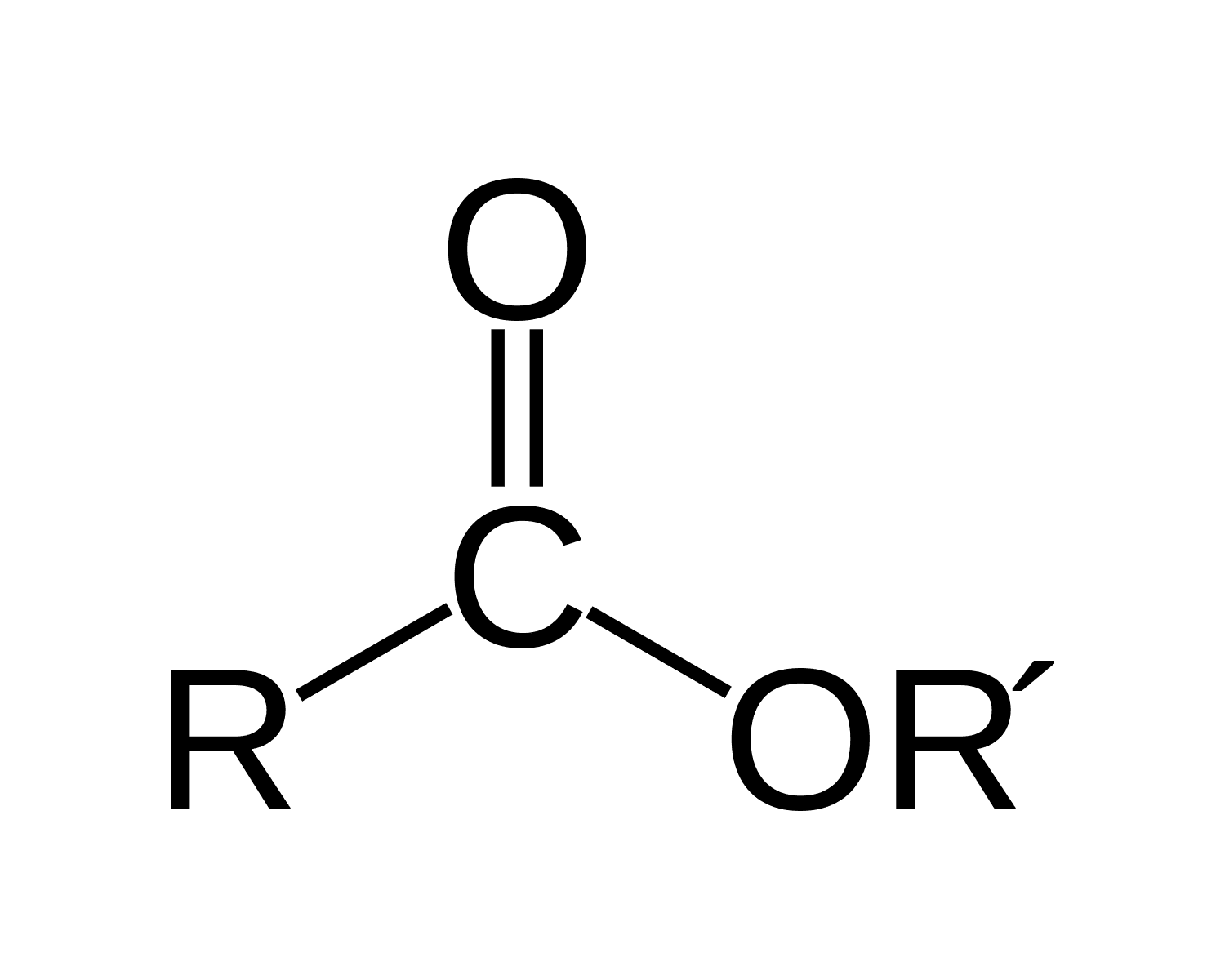
ester
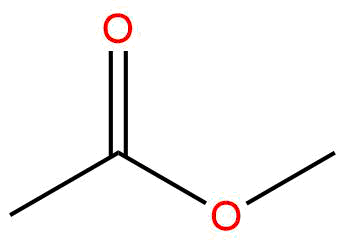
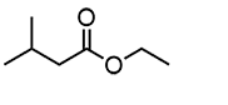
for esters IUPAC naming as the parent chain what do you attach
-oate
make sure that you also attach the name of the functional group bonded after the oxygen in the ester functional group in front of the entire name
for instance for this problem the full name is Ethyl 3-methylbutanoate
HNMR how to know if the signal will be singlet, doublet, triplet, quartet
n + 1 rule where n is the # of neighboring H’s around the designated proton and final answer will see if it is either one of those. The neighboring H must be from outside the carbon atom orignally attached to
Which compound will produce a large broad peak at 3200-3500 cm^-1 on IR spectrum
Alcohol group (-OH)
On an IR spectrum what are the characteristics of an Amine group (NH)
There will be a sharp peak at 3200-3500 cm^-1 and a secondary amine (-NR2H) peak and 2 primary amine peaks (-NRH2)
On an IR spectrum what are the characteristics of an Carboxylic acid group (-COOH)
2500-3500 cm^-1 and has broad jagged peak
On an IR spectrum what are the characteristics of an Nitrile group (-COOH)
2200-2250 cm^-1 and medium peak
On an IR spectrum what are the characteristics of an carbonyl group (C=O)
1700-1750 cm^-1 large sharp peak
On an IR spectrum what are the characteristics of an aromatic group
1450-1600 cm^-1 medium peak
Ethanol
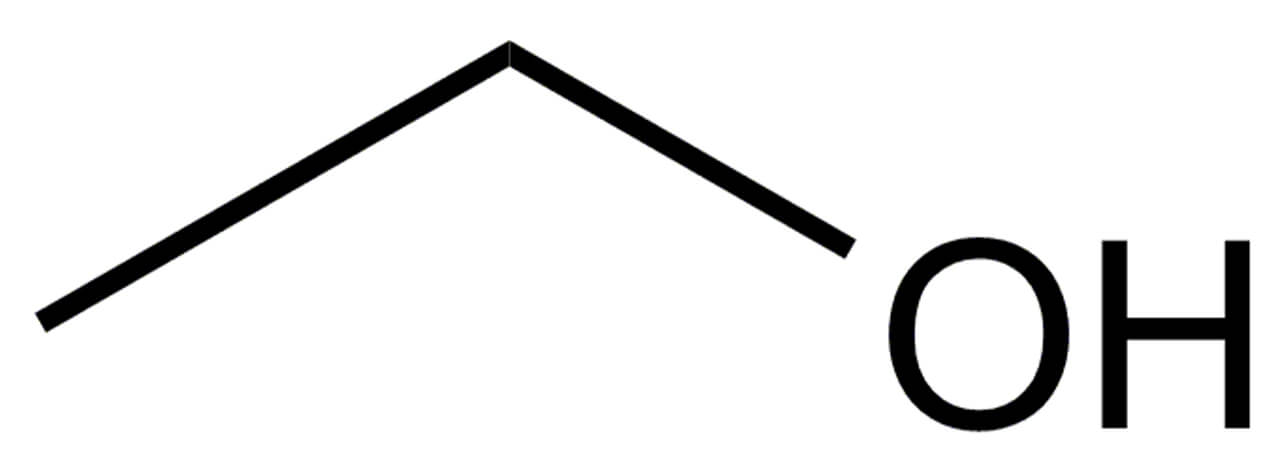
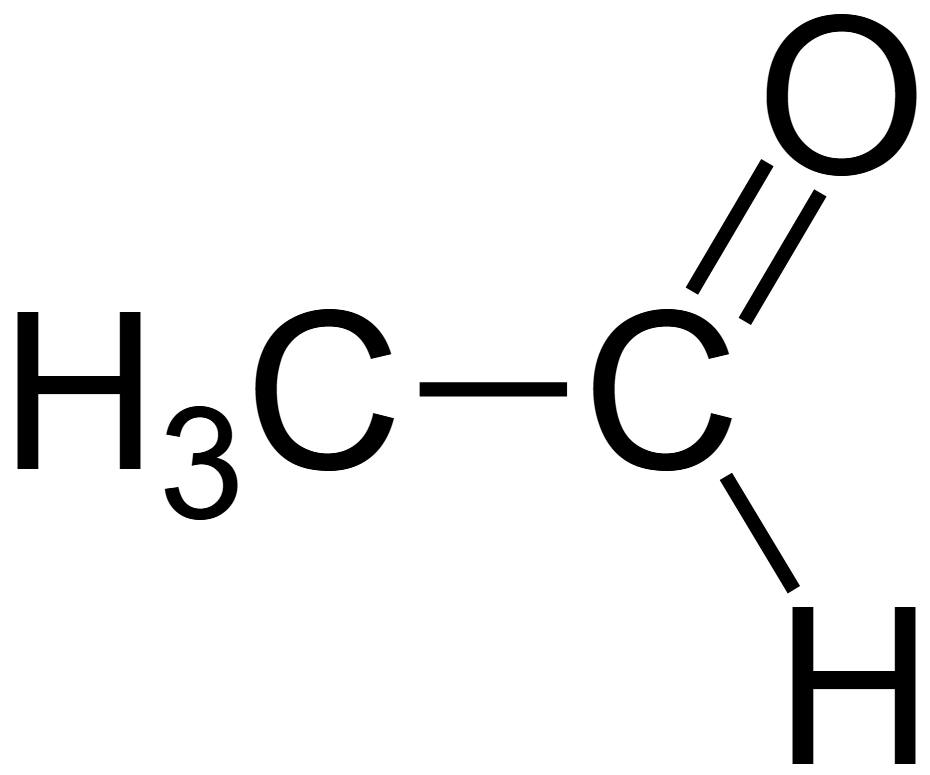
Ethanal
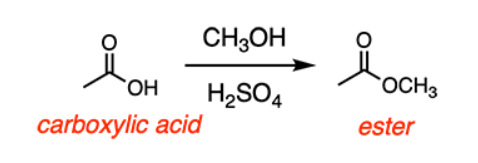
Ester