BIOC 4331 Lecture 26
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
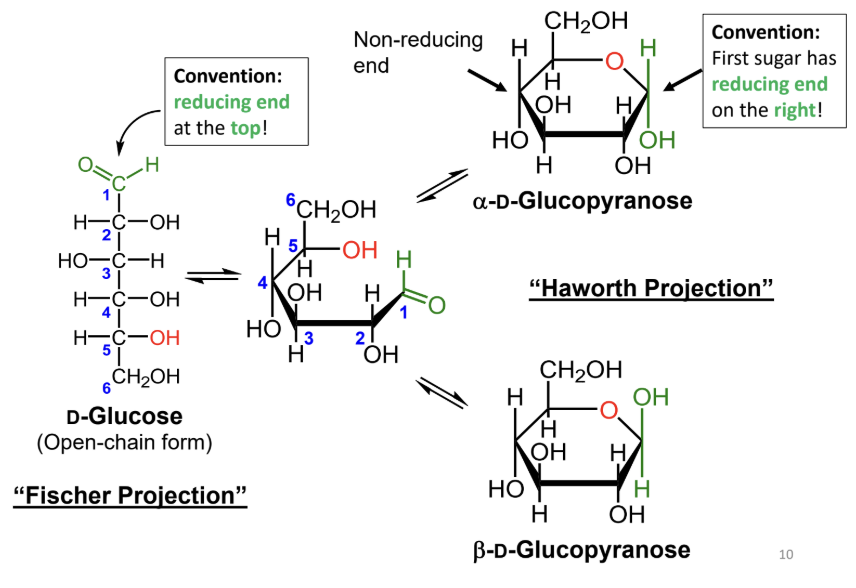
In a Fischer projection, where is the reducing end drawn by convention?
At the top.
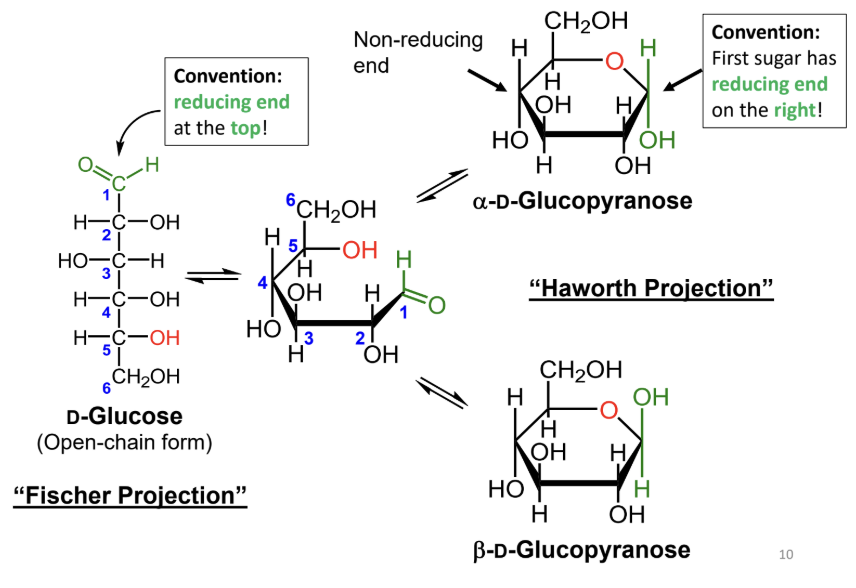
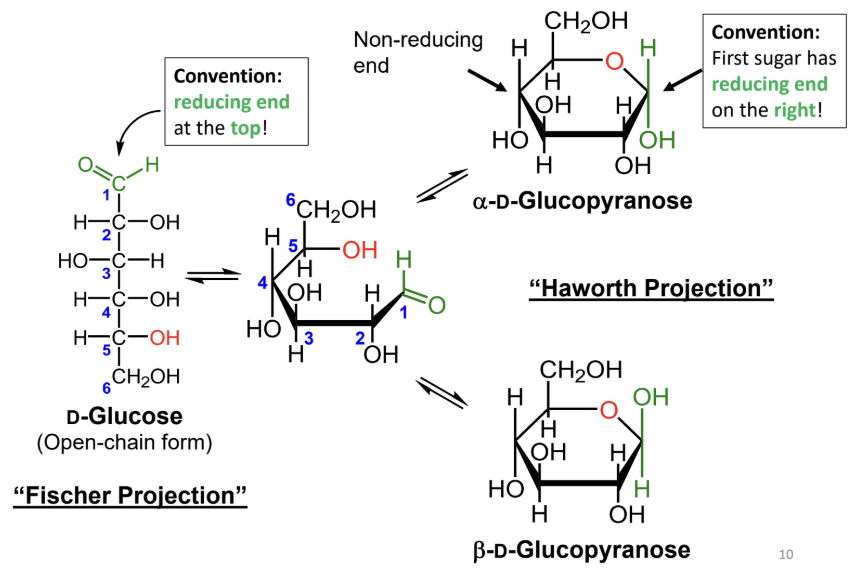
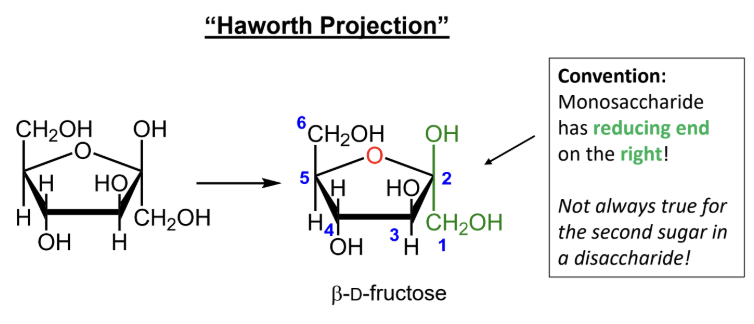
In a Haworth projection, where is the reducing end of the first sugar drawn by convention?
On the right.
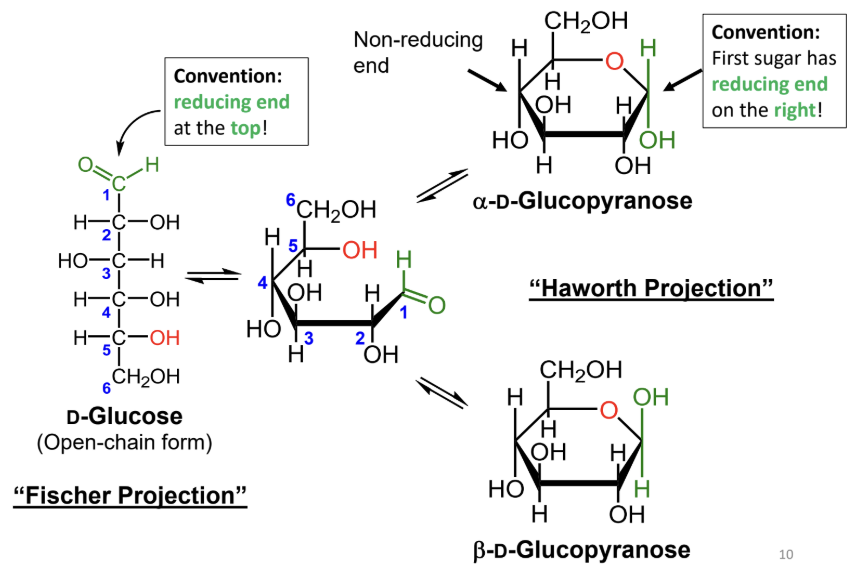
What reaction forms a cyclic sugar from a linear sugar?
An intramolecular reaction of an alcohol with an aldehyde or ketone to form a hemiacetal or hemiketal.
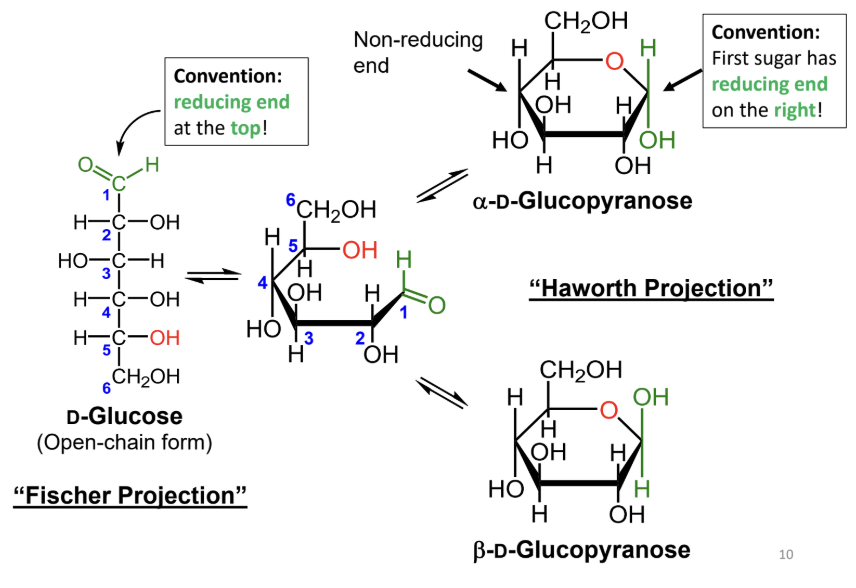
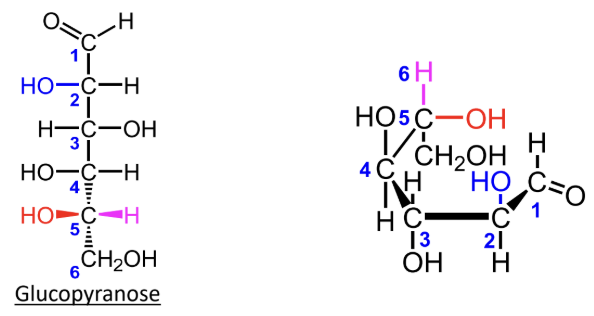
In a Fischer projection, how do you identify the anomeric carbon?
It is the carbonyl carbon (aldehyde or ketone carbon).
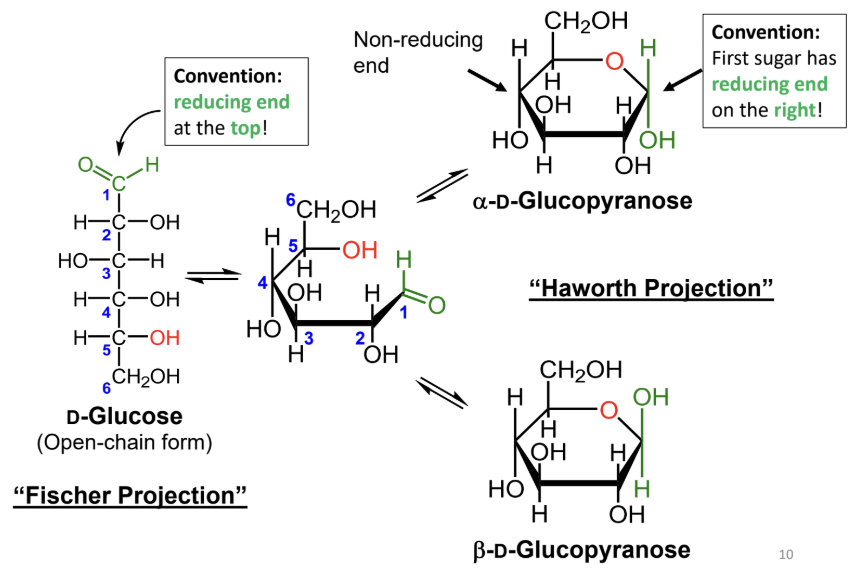
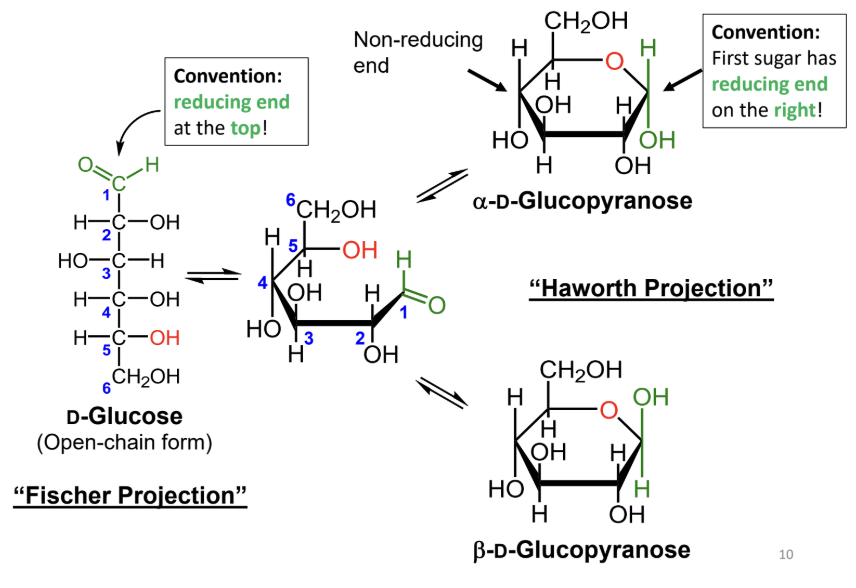
In a Haworth projection, how do you identify the anomeric carbon?
It is the carbon adjacent to the ring oxygen that is also bonded to another oxygen.
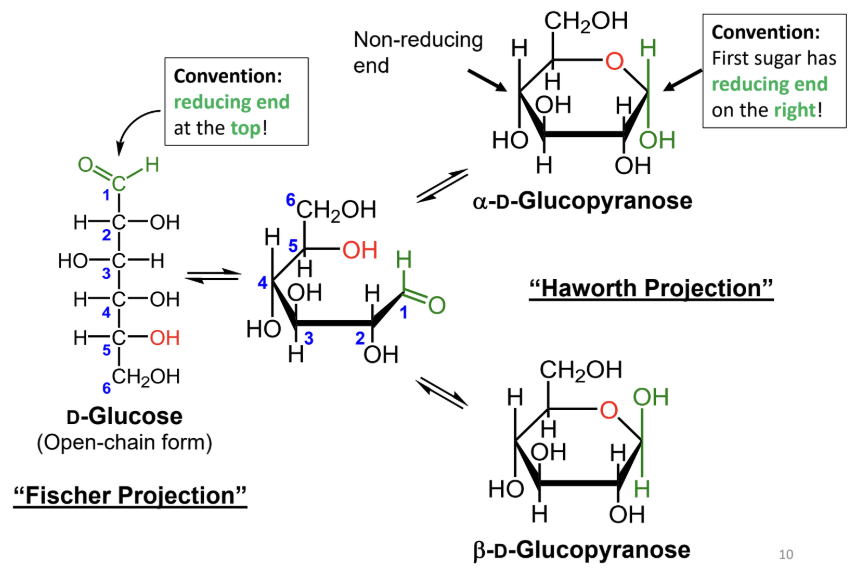
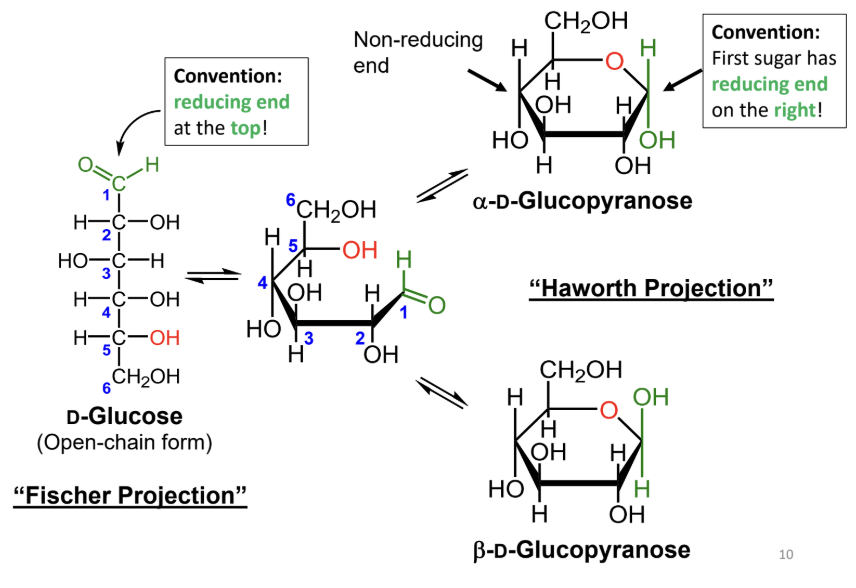
What numbering rule helps identify the anomeric carbon?
The anomeric carbon gets the lowest number possible when the carbons are counted in order.
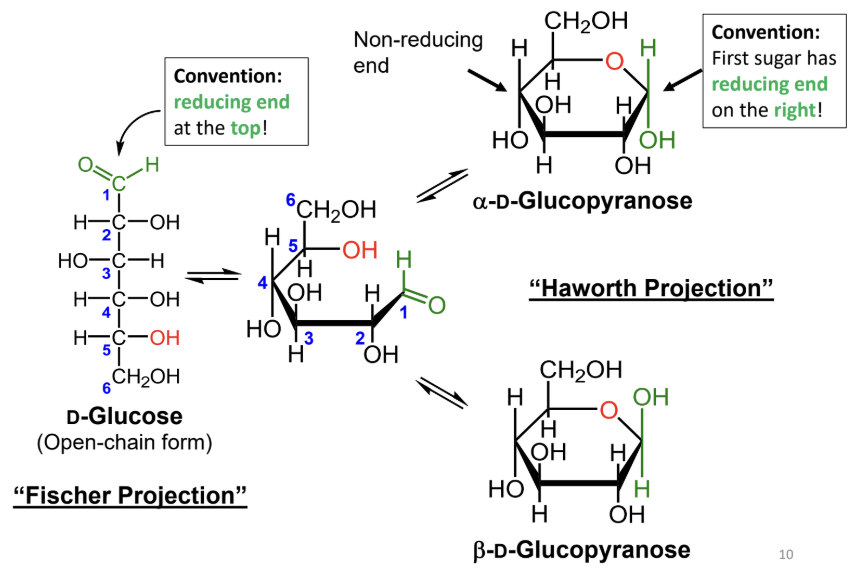
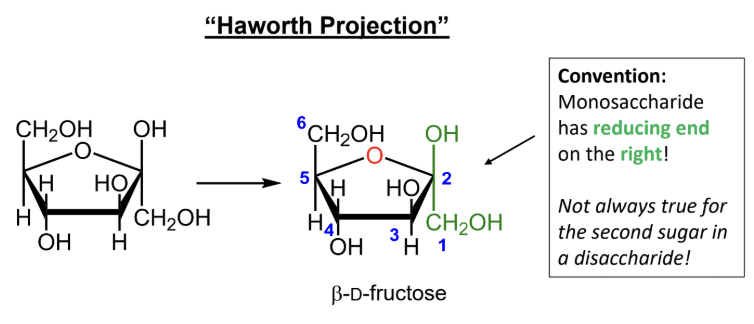
When numbering a Haworth projection, where do you start?
At the end of the carbon chain closest to the anomeric carbon, then number around the ring without passing through the ring oxygen.
What new stereochemical label appears for cyclized sugars?
Alpha or beta.
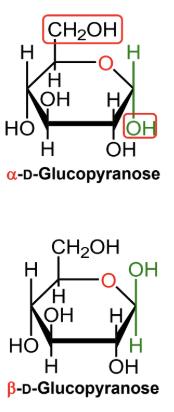
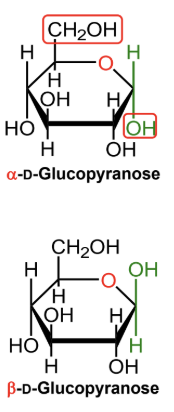
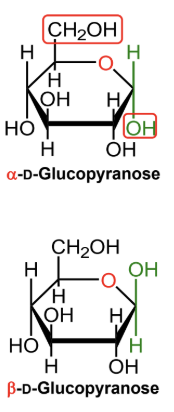
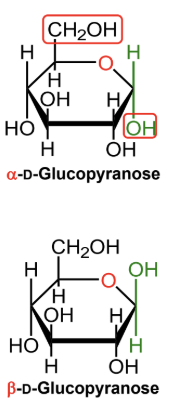
For a D-hexose in a Haworth projection, what does alpha mean?
The anomeric OH is on the opposite side from the exocyclic CH2OH group.
(-OH! It’s Hαw-pposite to the highest exocyclic carbon)
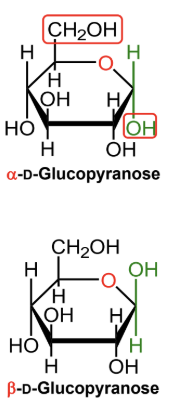
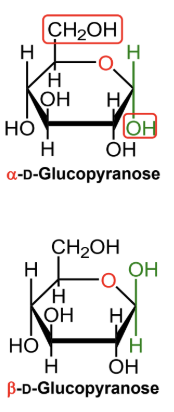
For a D-hexose in a Haworth projection, what does beta mean?
The anomeric OH is on the same side as the exocyclic CH2OH group.
The alpha/beta shortcut rule works for which sugars?
Aldoses and keto-furanoses.
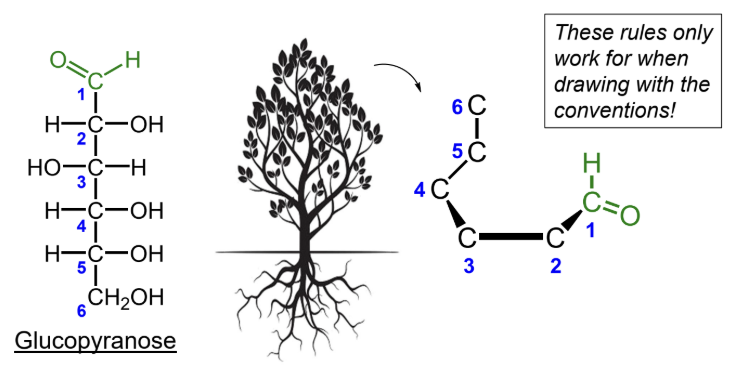
In converting Fischer to Haworth, what is the “tree” mnemonic?
The Fischer tree falls right down.
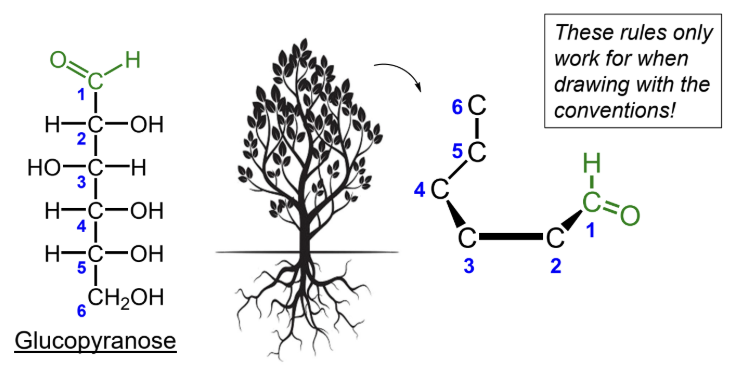
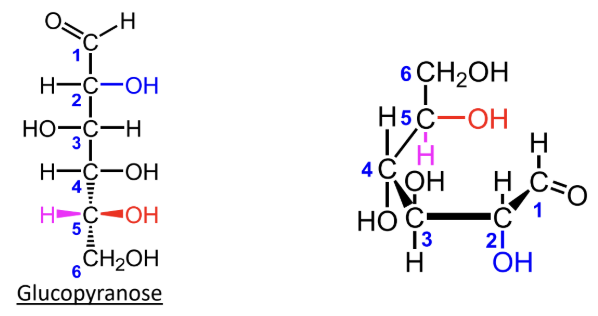
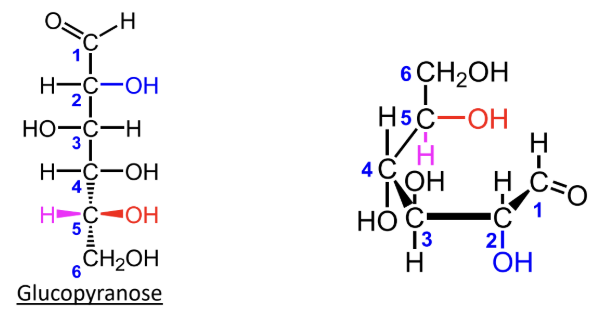
For D-monosaccharides, where is the highest-numbered CH2OH group placed in the Haworth ring?
Above the ring.
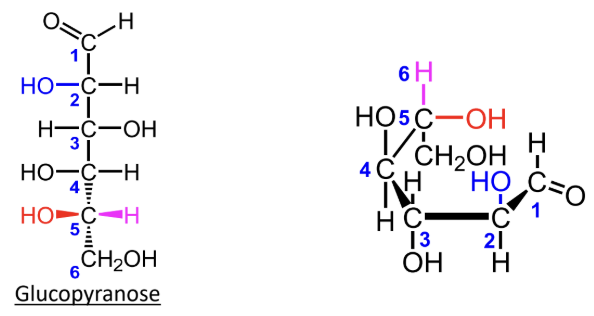
For L-monosaccharides, where is the highest-numbered CH2OH group placed in the Haworth ring?
Below the ring.
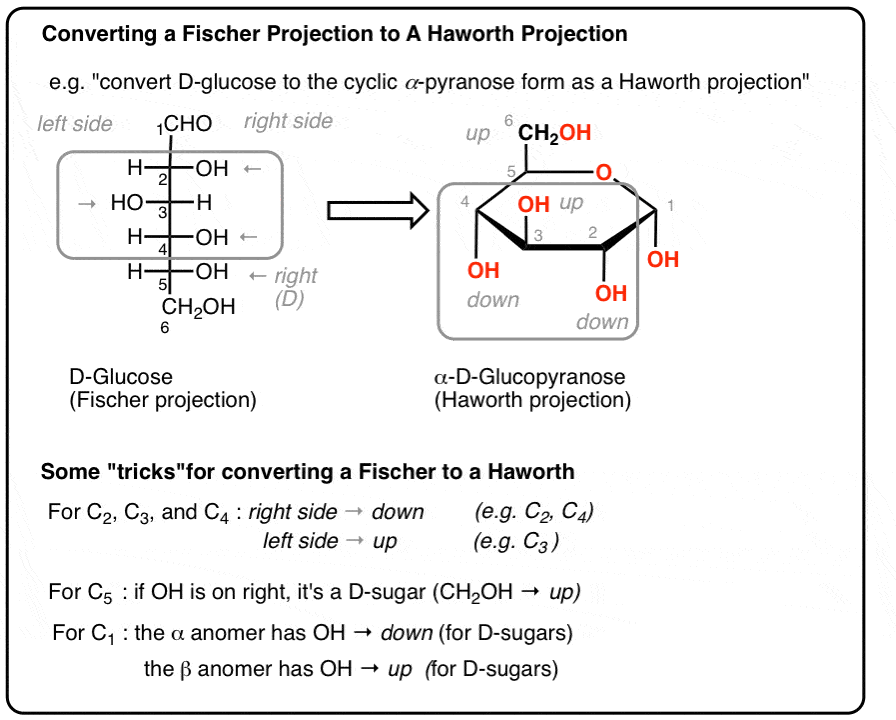
For all monosaccharides in Fischer to Haworth conversion, where do groups go if they are on the right side of the Fischer projection?
Below the ring.
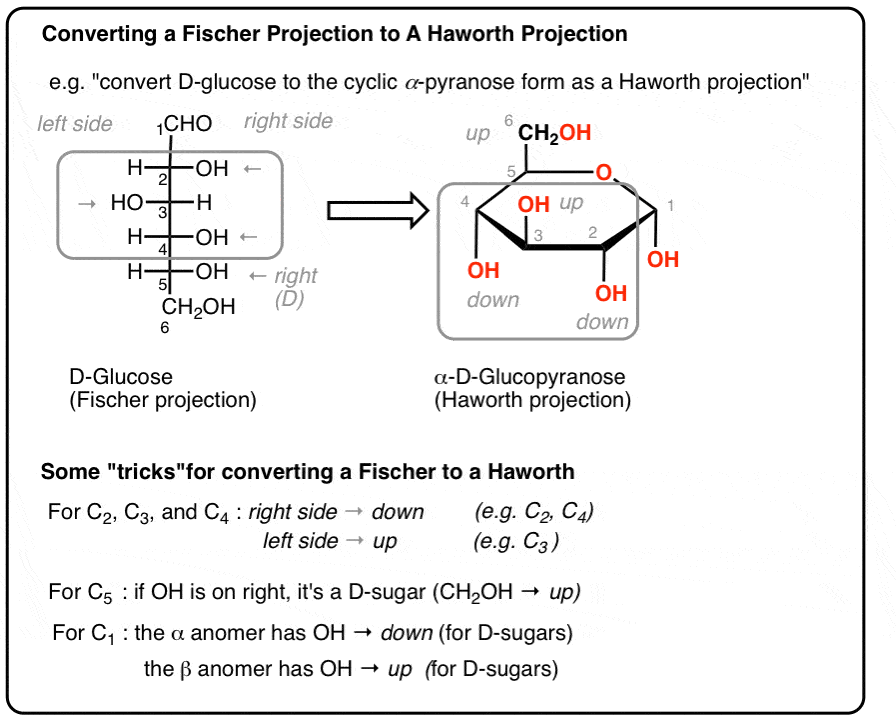
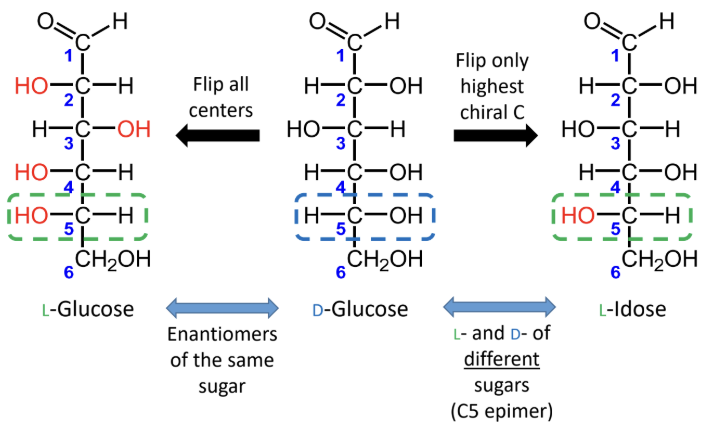
How do you tell whether a sugar is D or L in a Fischer projection?
Look at the highest-numbered chiral carbon: OH on the right = D, OH on the left = L.
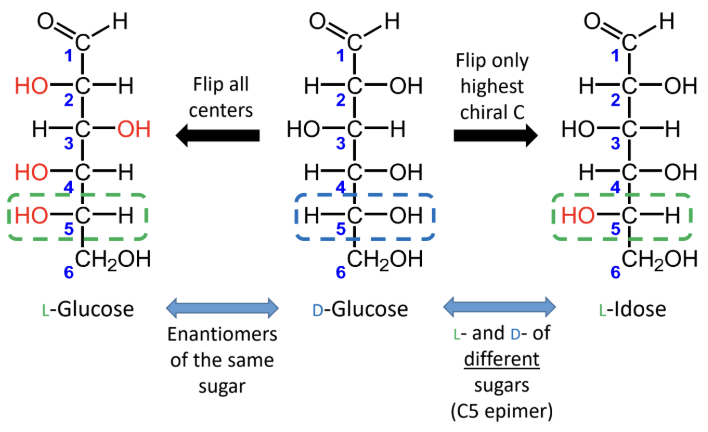
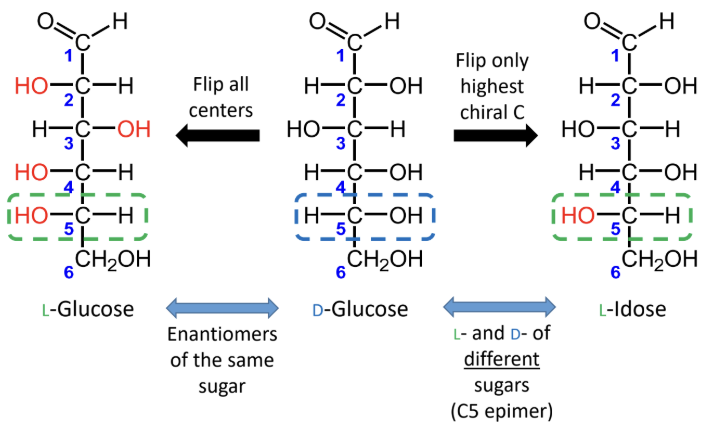
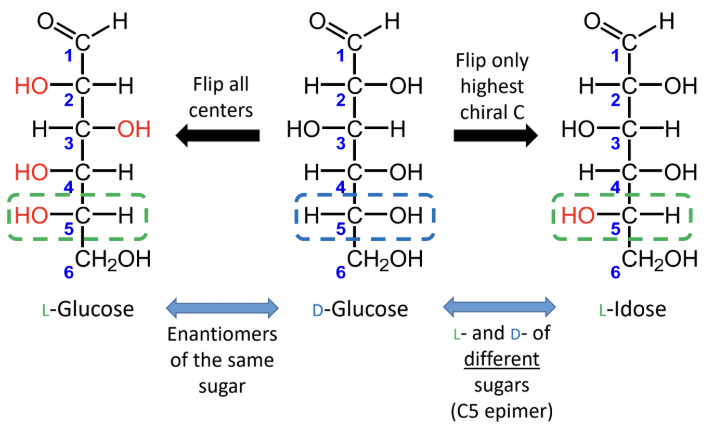
For sugars with the same name, what is the relationship between the D and L forms?
They are enantiomers (mirror images).
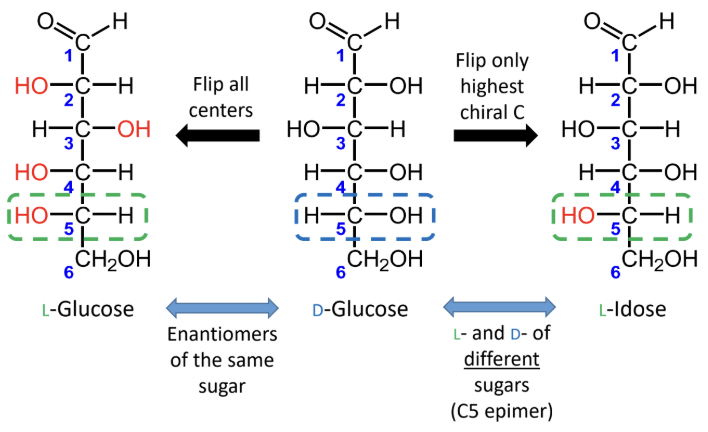
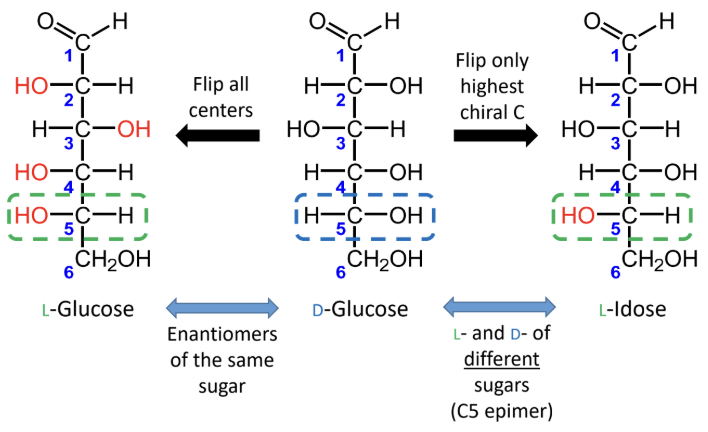
What happens if you flip only the highest chiral carbon instead of all chiral centers?
You get the L- and D- forms of different sugars, not enantiomers of the same sugar.
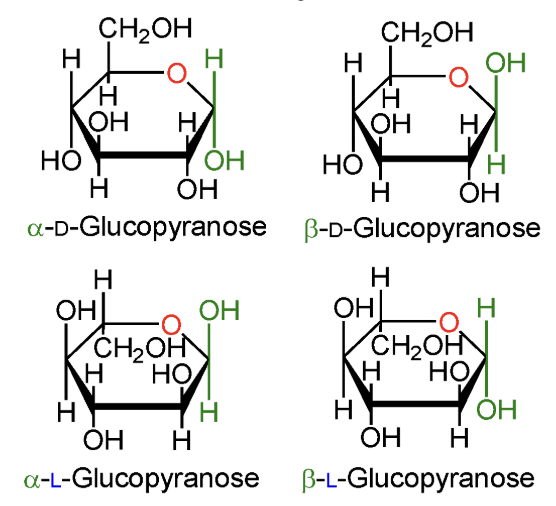
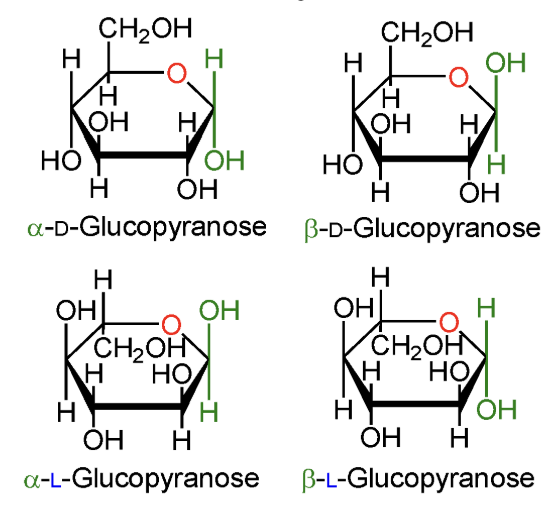
In α-D-glucopyranose, is the anomeric OH above or below the ring?
Below the ring.
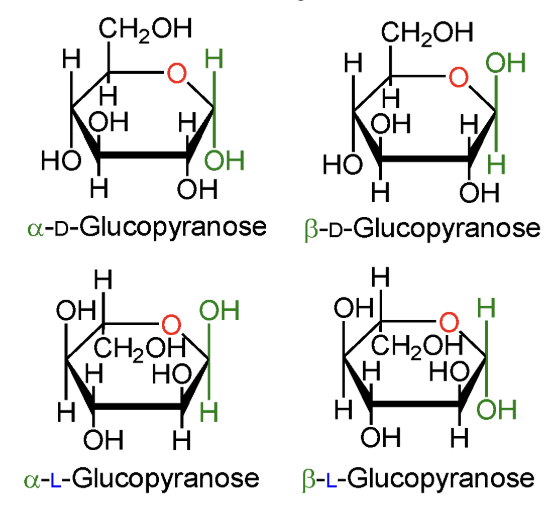
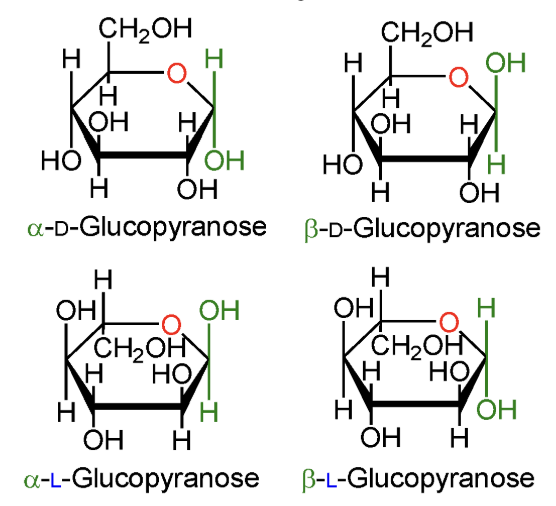
In β-D-glucopyranose, is the anomeric OH above or below the ring?
Above the ring.
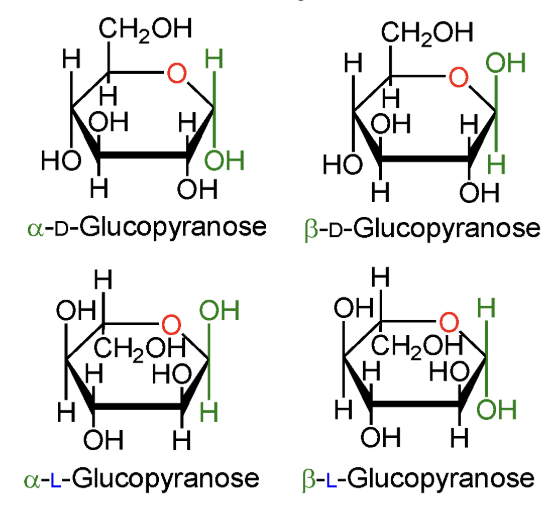
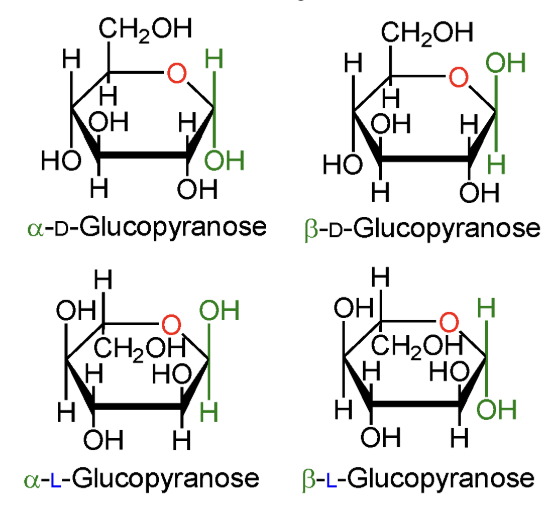
In α-L-glucopyranose, is the anomeric OH above or below the ring?
Above the ring.
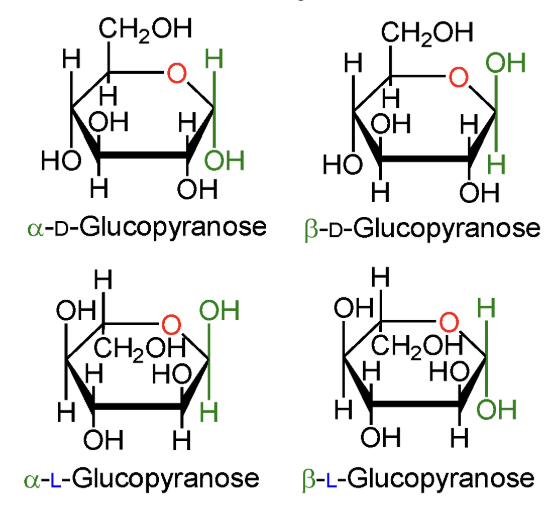
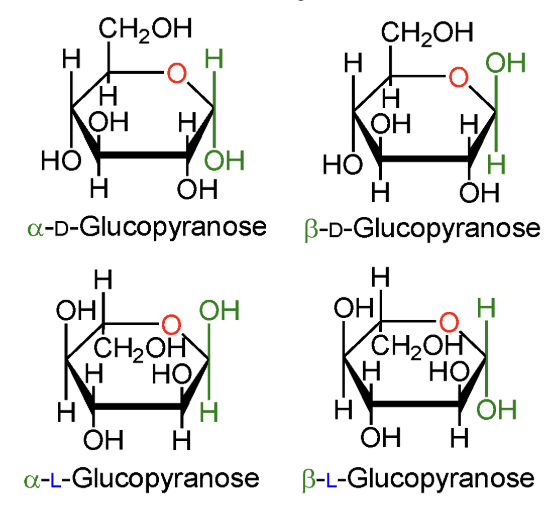
In β-L-glucopyranose, is the anomeric OH above or below the ring?
Below the ring.

What does a blue Fehling’s test result mean?
No reaction/no reduction.


What does a brown/red precipitate in Fehling’s test mean?
Reaction/reduction.

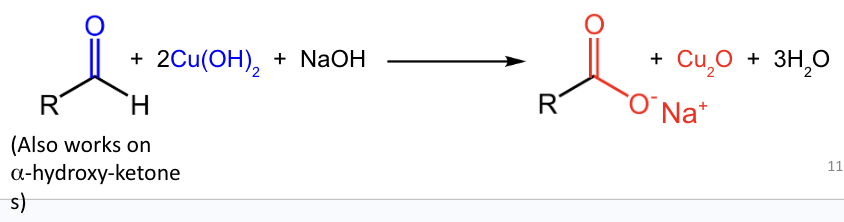
Fehling’s test detects reduction of what reagent to what product?
Cu2+ is reduced to Cu2O.
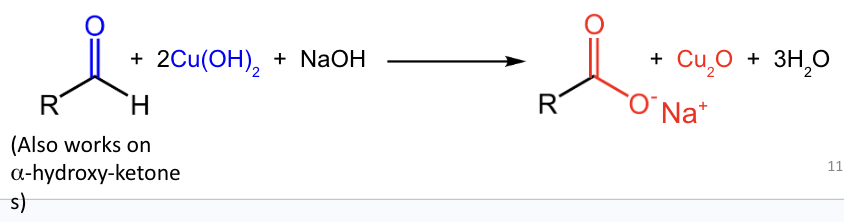
Fehling’s test also works on what besides aldehydes?
α-hydroxy ketones.
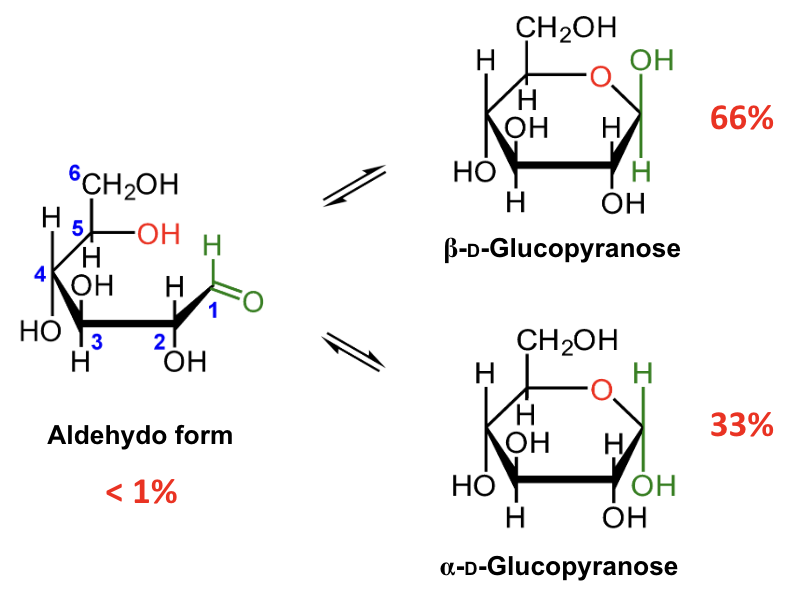
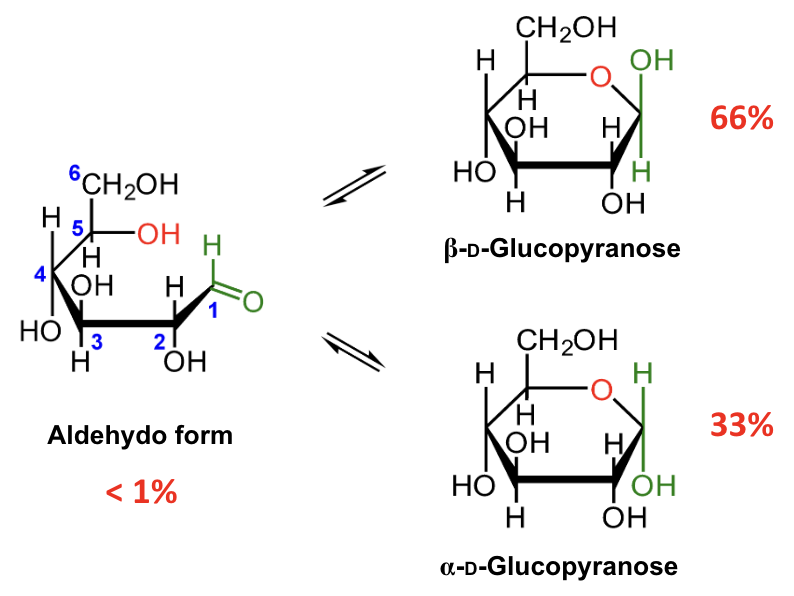
In the mutarotation equilibrium of D-glucose, which anomer is more abundant?
β-D-glucopyranose.
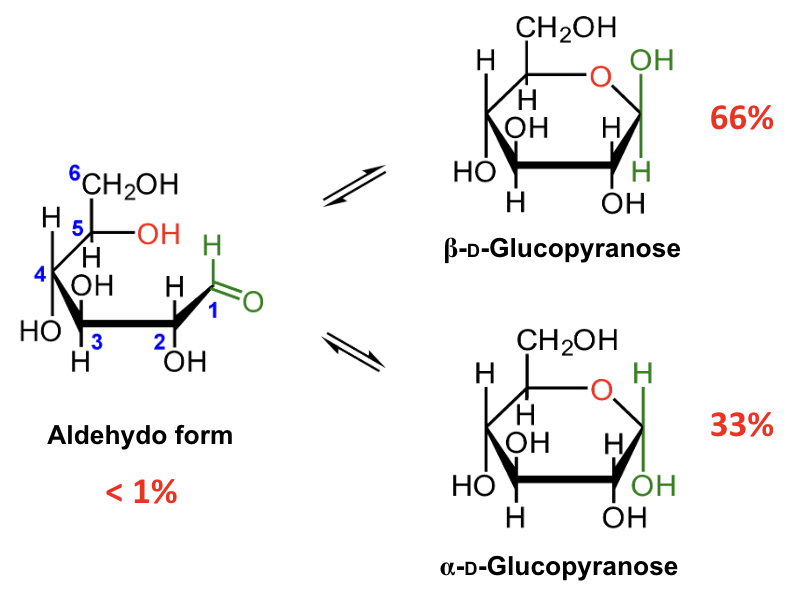
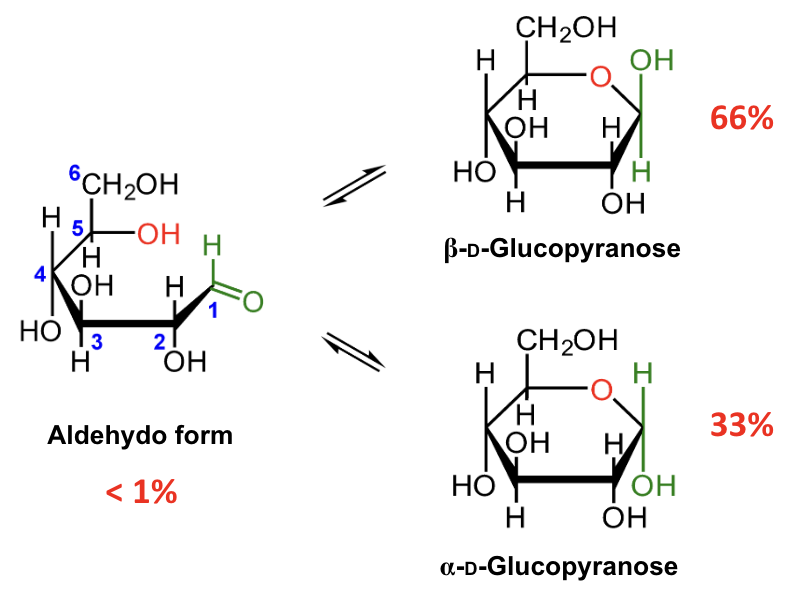
About what percentage of D-glucose at equilibrium is β-D-glucopyranose?
About 66%.
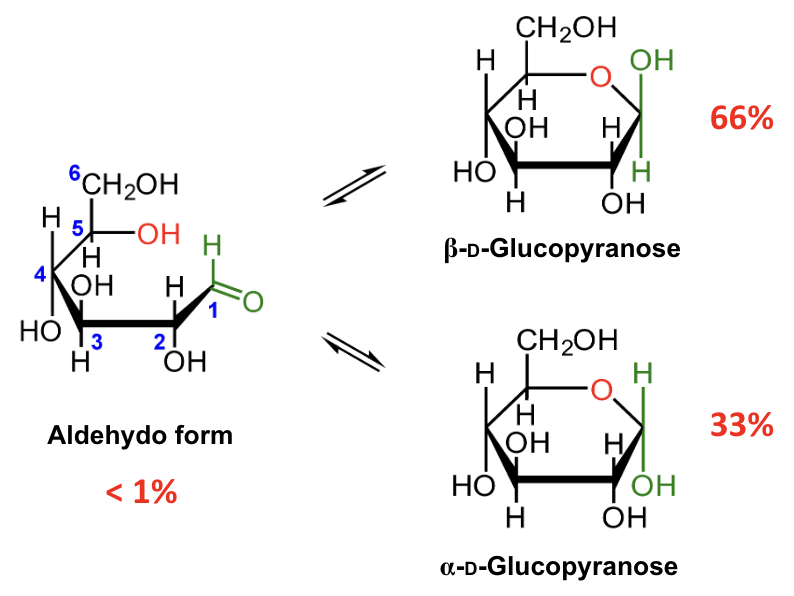
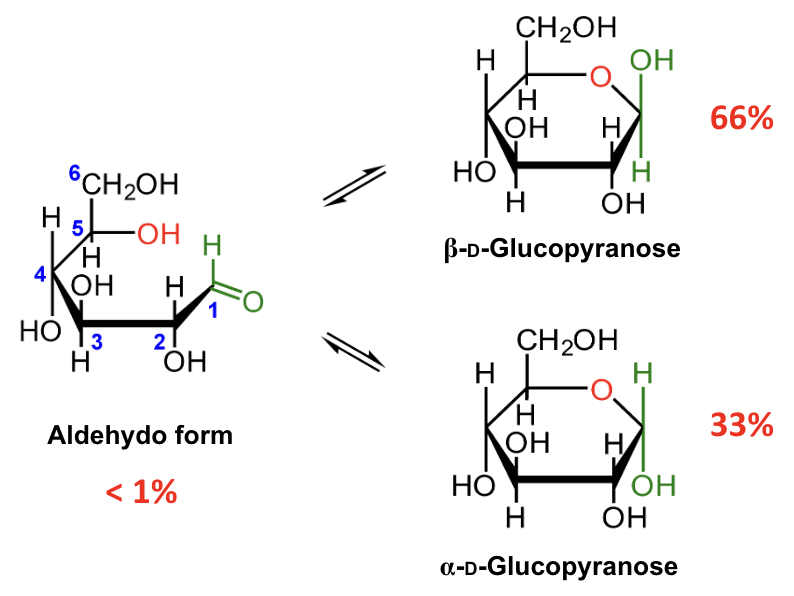
About what percentage of D-glucose at equilibrium is α-D-glucopyranose?
About 33%.
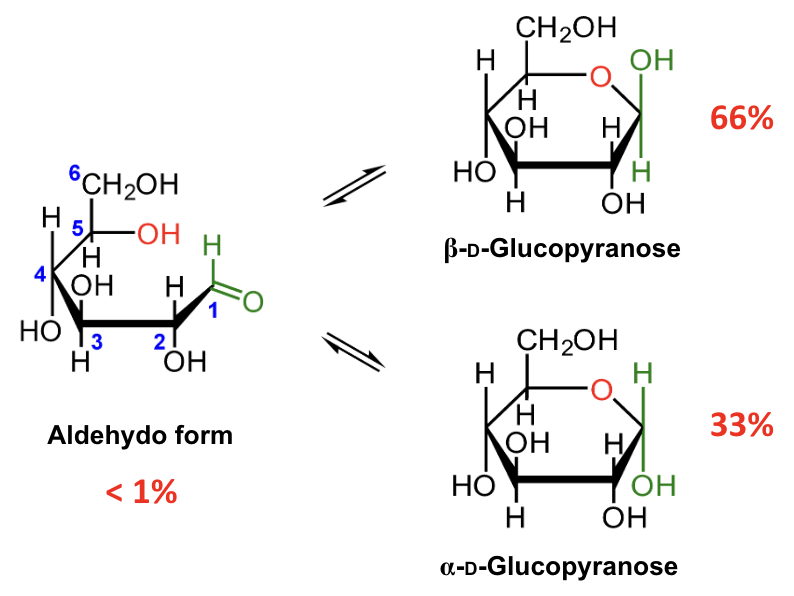
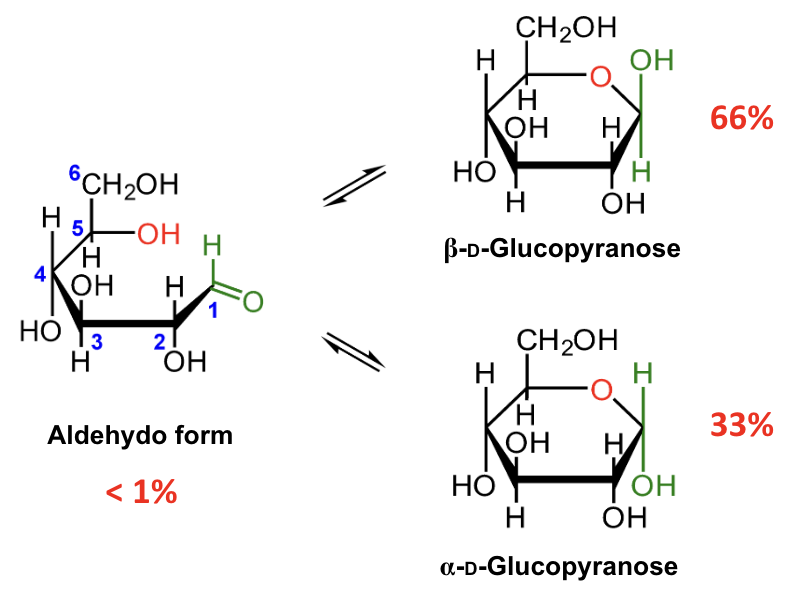
About what percentage of D-glucose at equilibrium is in the open-chain aldehydo form?
Less than 1%.
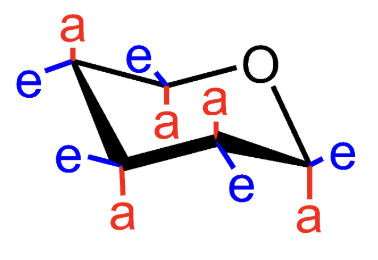
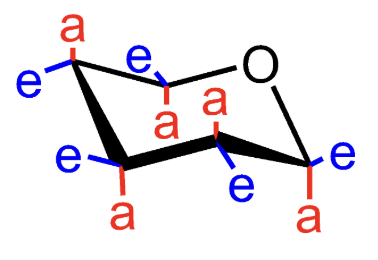
What does e mean in a chair conformation?
Equatorial substituent.
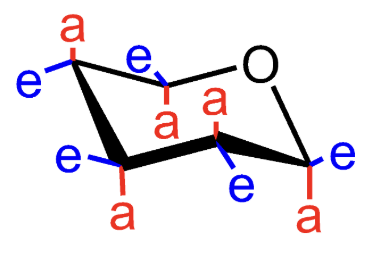
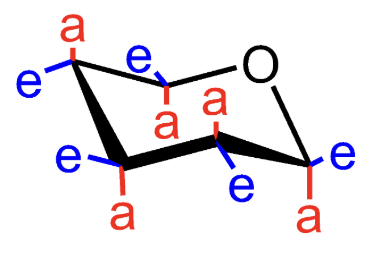
What does a mean in a chair conformation?
Axial substituent.
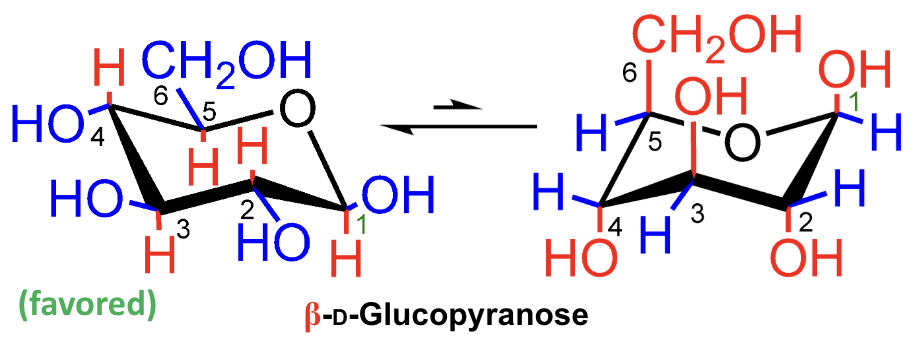
Which chair form of D-glucopyranose is favored?
β-D-glucopyranose.
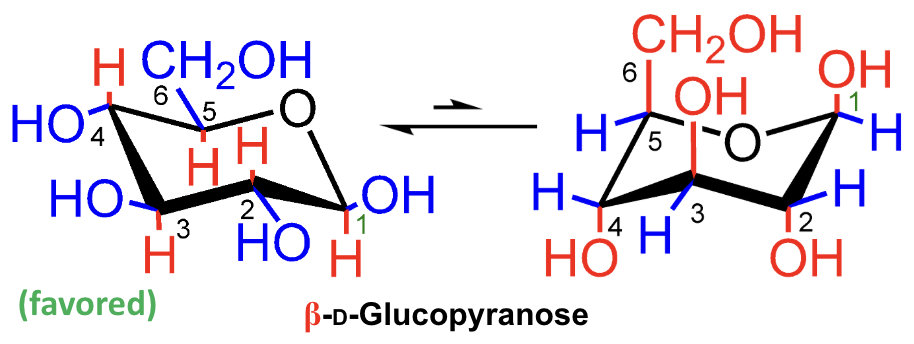
Why does stereochemistry matter so much in carbohydrates?
Because shape and stereochemistry determine enzyme recognition.
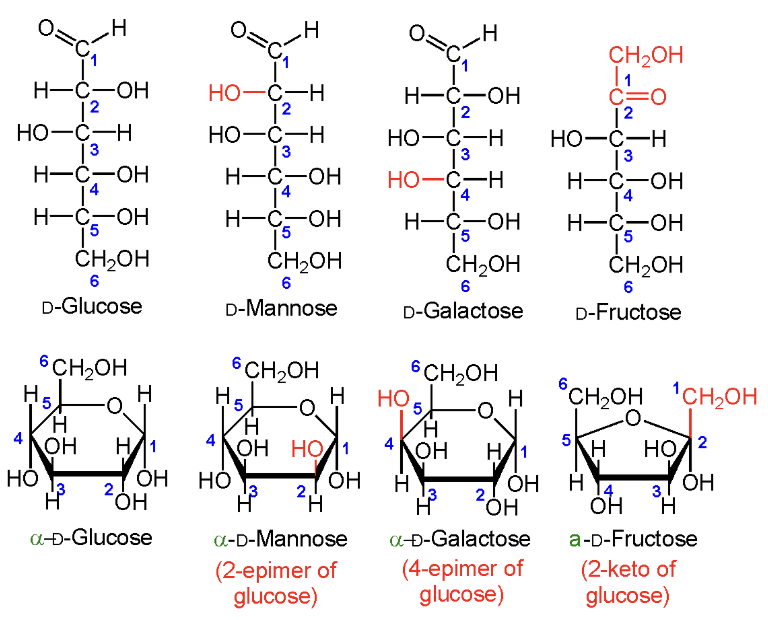
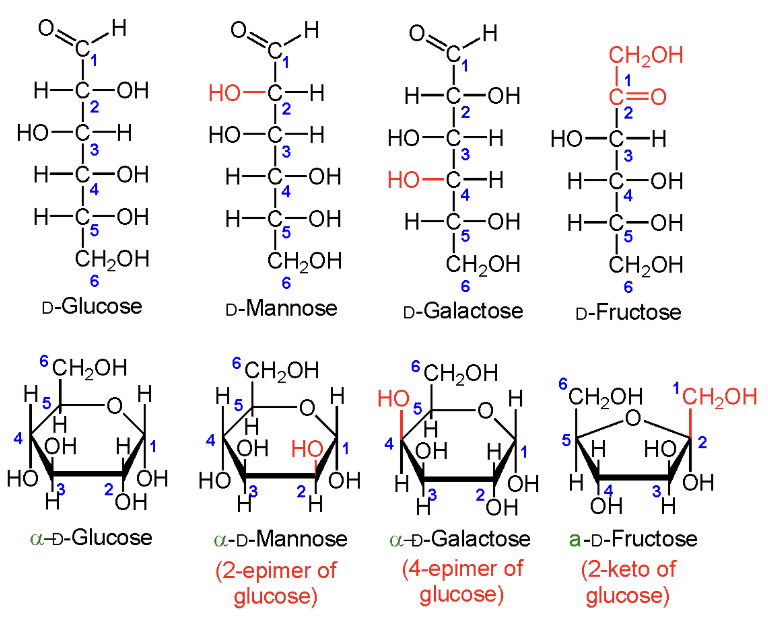
Which four sugars do you need to know?
D-Glucose
D-Mannose
D-Galactose
D-Fructose.