chem unit 2
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
arhennius acid
produces H ions when dissolved in aqueous solution; [HA]
Using ICE vs SRF tables
ICE: determining eq concentrations, will give Ka or Kb, use molarity; SRF: used mostly for buffer reactions, SA +SB reactions
Arrhenius base
produces hydroxide ions (OH-) when dissolved in aqueous solution; [A]
Equivalence point
Moles of titrant are EQUAL
bronsted lowry acid
proton (H+) donor (HCl)
bronstead Lowry base
proton (H+) acceptor (NH3)
conjugate acid
the species formed when a proton is transferred to the base; NH3→ NH4+
conjugate base
what remains of an acid molecule after a proton is lost; HCl → Cl-
hydronium ion
H3O+; the predominating form of the proton in an aqueous solution; can use H+ and H3O+ interchangeably
strong acid
complete dissociation of that ion to produce H+ and the conjugate base; lose H+ much easier; HA + H2O → A +H3O ; single arrow forward means complete dissociation
weak acid
partial dissociation; HA + H2O ←→ A+H3O ; arrow forward and back to show it has to move forward and back to reach equilibrium
strong base
a metal hydroxide salt that completely dissociates into its ions in water; holds onto H+ strongly; H+H2O → HB + OH; single arrow shows complete dissociation
weak base
a base that reacts with water to produce hydroxide ions to only a slight extent in aqueous solutions; BH2 + H2O ←→ BH3 +OH ; arrow forward and back to show it has to move forward and back to reach equilibrium
Ka
acid dissociation constant; numerical measure of an acids strength in aqueous solution; higher Ka means stronger acid; [A][H3O]/[HA]
autoinization
the transfer of a proton from one molecule to another of the same substance; OH to H2O or vice versa; water is amphoteric- can be acid or base
Kw
ion product constant; equilibrium constant for the autoionization of water; = 1.0×10^-14; Kw=[H3O][OH]; pKw=pH + pOH=14
significant figures and pH: characteristic and mantissa
pH=7.00; characteristic: related to the exponent of the power of 10 (7); mantissa: indicates the number of sig figs (00)
calculating the pH of weak acids
weak acids will dissociate and form an equilibrium with their conjugate bases; set up Ka expression; set up ice table; solve for x and determine concentrations and then convert to pH
calculating pH of weak bases
weak bases will dissociate and form an equilibrium with their CA; set up Kb expression and use ice table
salt
ionic compound
predicting whether a salt solution will be acidic, basic, or neutral
1) strong base and strong acid: salt has no hydrolyzable ions and form a neutral salt solution; 2)strong base and weak acid: produces basic salt; 3)weak base and strong acid: produces acidic salt
common ion effect
the shift in equilibrium position caused by the addition or presence of an ion involved in the equilibrium reaction
buffer solution
resists change in pH when OH ions or H protons are added; consists of a weak acid and its salt (conjugate base) or consists of a weak base and its salt (conjugate acid)
Henderson hasselbach equation (provided)
used to calculate buffer pH or design buffers; relate the pH of a buffer solution to the pKa of a weak acid and its conjugate base
buffering capacity
[A]/[HA]; the ability of a buffered solution to absorb protons or hydroxide ions without a significant change in pH ; determined by magnitudes of [HA] and [A] in solution; the most effective buffer is one where the ratio is close or equal to 1, meaning when pH is close to pKa
titration
the process of reaction a solution of unknown concentration with one of known concentration (standard solution)
titration curve
graph of the pH as a function of the volume of added titrant
strong acid-strong base titration
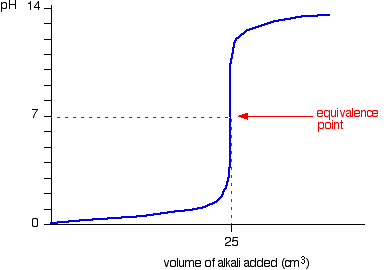
equivalence point
weak acid strong base titration
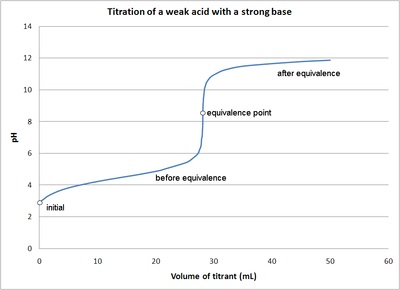
initial pH, buffer region: between initial and before equivalence; equivalence point; half equivalence point right in the middle of the buffer region: where pH=pKa; pH at equivalence point is above 7
strong acid weak base titration
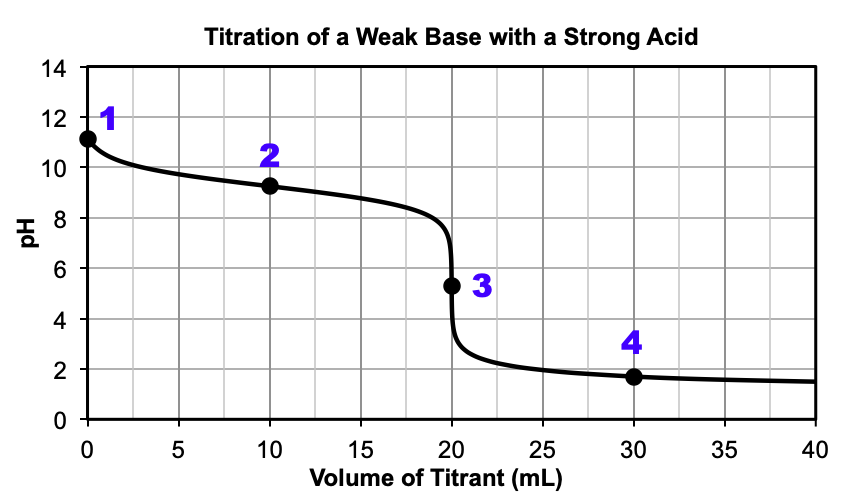
2: half equivalence point and buffer region, pH=pKa; 3= equivalence point; equivalence point is below 7
determining equivalence point experimentally
use an acid-base indicator which marks the endpoint of titration by changing color
how to choose which indicator to use; you will have the color chart
select one whose pH color change range overlaps the steep vertical section of your titration curve, usually centered near the equivalence point
percent dissociation
concentration dissoicated/original concentration